Q: After 51.0 min, 16.0% of a compound has decomposed. What is the half-life of this reaction assuming ...
A:
Q: Stomach acids (HCL) has a pH of about 1.5. For individuals who suffer from acid refulx, antacids or ...
A: We have to predict the balance chemical reaction.
Q: Draw the structure of the major organic product(s) of the reaction. H20 CI • You do not have to cons...
A: Answer of the question given below,
Q: A solution contains 10.0 g pentane, C5H12, 10.0 g hexane, C,H14 and 10.0 g benzene, C6H6. What is th...
A:
Q: There are 20 common, naturally occurring amino acids from which all proteins are derived (as will be...
A:
Q: The following compounds are of interest in pharmaceutical chemistry, since they are part of the synt...
A: Here we have to synthesize the given target compound by 2-chloromethylthiophene by multistep reactio...
Q: e) Discuss whether the following reaction will be reversible or irreversible. OH cP Cl
A: Since you have asked multiple questions, as per guidelines we will solve only the first one for you....
Q: 2. Given the reactions: Reaction Kc at 25°C 6.8 x 104 HF(aq) H*(aq) + F'(aq) H2C2O4(aq) = 2H*(aq) + ...
A:
Q: Which of the following IS NOTa structural isomer of hex-1-ene (C&H12)? 04-methylpent-2-ene hex-3-ene...
A: isomers are molecules with identical molecular formulae that is, same number of atoms of each eleme...
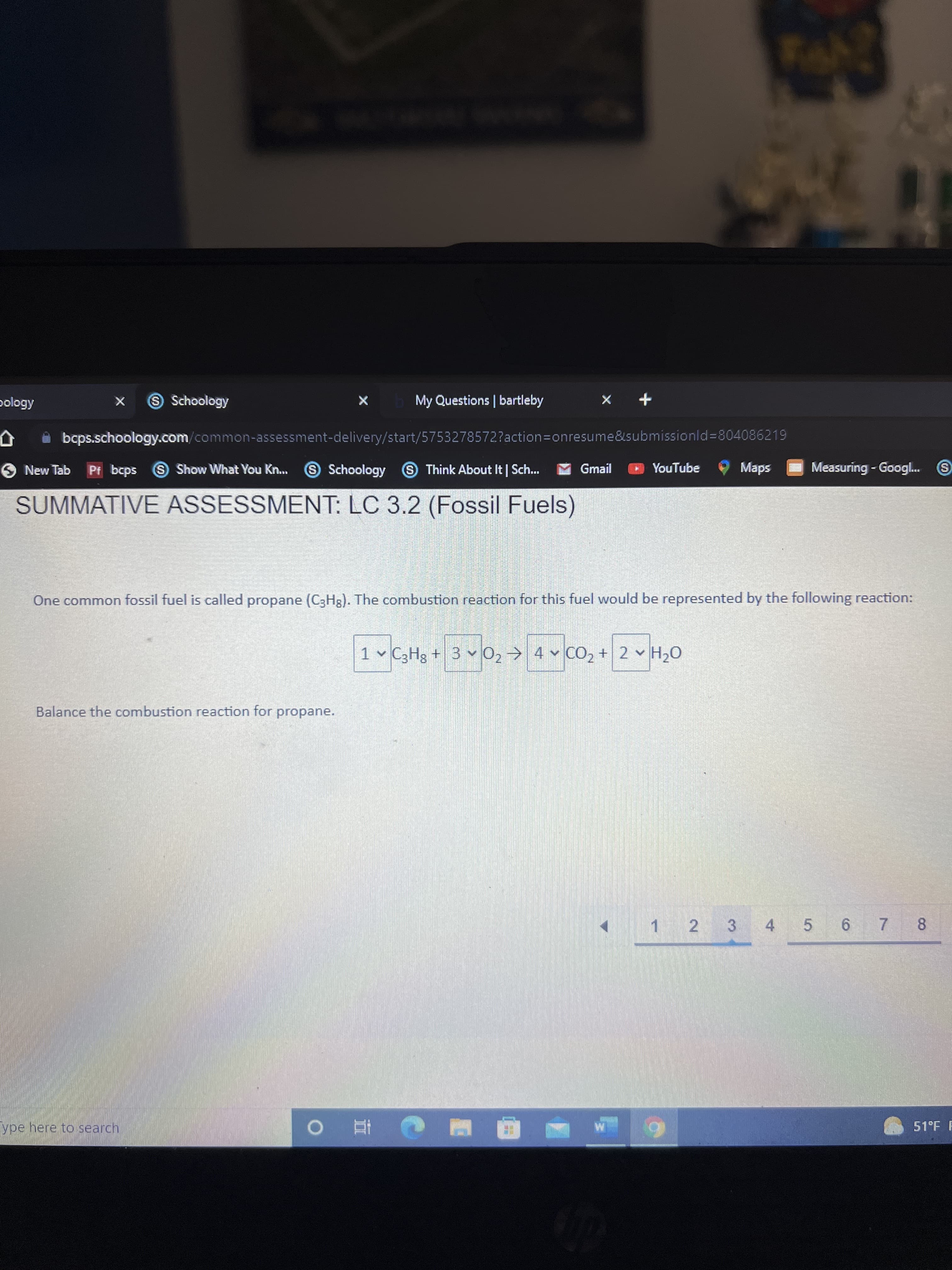
Q: In the combustion of methane, how many moles of O, are required if 6.75 mol of CH, is to be complete...
A: Complete combustion of methane produce CO2 and H2O CH4 + 2O2 ---------- > CO2 + 2H2O
Q: 1. What is so important about the fatty acids in oil paint and what does that contribute to the dryi...
A: Answer: 1) a) Important about the fatty acids in oil paint- b) Fatty acids in drying process. 2) Ch...
Q: How many grams of nitrogen are required to react completely with 6.5 liters of hydrogen in forming a...
A:
Q: When UV radiation of wavelength 58.4 nm from a helium lamp is directed on to a sample of Xe, electro...
A:
Q: reaction with 4 moles of C2H60 and 6 moles of 02. The Limiting Reactant is O2 Reaction Table C2H60 3...
A:
Q: Consider the following reaction: 2NH3 (g) N2(g) + 3H2(g) If at 298K, 3.55 mol of NH3 are added t...
A: Given, 2NH3 (g) <----> N2(g) + 3H2(g) If at 298K, 3.55 mol of NH3 are added to a 1.00L contain...
Q: 7-43. Supply the missing data in the table below. Molar Analytical Concentration, cT Acid (cr = cHa ...
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for you...
Q: Same reaction as previous question. Which statement(s) is(are) true concerning the rate-limiting ste...
A: We have to predict the correct statement regarding rate limiting step.
Q: A sodium hydroxide solution has a pOH of 3.11. Determine the [H"] concentration in this solution.
A: Given, pOH of sodium hydroxide (NaOH) solution = 3.11 Concentration of H+ ion = [H+] = ? The formula...
Q: How doyou complete this reaction pathway H3O+
A:
Q: KMnO4 according to the following chemical equation: K2C2O4 (aq) + 2 KMnO4 (aq) + 8 H2O →10 CO2 ...
A: Based on the balanced chemical equation for the given reaction and following simple unitry method we...
Q: 400 450 500 550 600 650 700 wavelength (nm) : Wavclength of maximum absorbance for base form ph show...
A:
Q: What mass of H2SO4 would be required to prepare 750. mL of a 0.150 M H2SO4 solution?
A: Given, Molarity of H2SO4 solution = 0.150 M = 0.150 mol/L Volume of H2SO4 solution = 750. mL = 0.750...
Q: Propose an efficient synthesis for the following transformation: он ?. OH The transformation above c...
A:
Q: What is the solubility of PbSO 4 in 0.1868 M Na 2SO 4? K sp(PbSO 4) = 6.300e-7
A: The solubility of PbSO4 can be calculated as follows
Q: How many liters of amımonia will be produced from 15 grams of hydrogen?
A: To solve this problem the first thing you need to know is the equation for the reaction of hydrogen ...
Q: The colorless solution made from an unknown white solid turned blue litmus red. The flame test showe...
A:
Q: temperature
A:
Q: Calculate the concentration in meq/L in a sample that contains 50.8 mg/L of calcium .
A:
Q: In a coffee cup calorimeter, 35 ml of 0.100 M AgNO3(aq) and 69 mL of 0.100 M NaCI (aq) are mixed. Th...
A:
Q: Using activity coefficients, calculate the concentration of Co" present in a 0.1 M solution of KNO3 ...
A:
Q: The limiting reactant determines how much product can be formed. Select one: True O False
A: Given statement: The limiting reactant determines how much product can be formed. We have to find wh...
Q: Solid zinc hydroxide and solid aluminum hydroxide are in equilibrium with a solution containing 9.61...
A: Solution contains Zinc nitrate. Concentration of zinc nitrate = 9.61 x 10-3 M The solid precipitates...
Q: 1. In order to make 1.5 L of 0.5 M CUSO4 (MM:159.6g/mol), 79.8 g of CuSO4 is needed. II. One hundred...
A:
Q: Calculate the pH of a buffer solution made from 0.30M hydrofluoric acid and 0.70 sodium fluoride aft...
A:
Q: b. Describe two properties you would predict for propanone. Justify your answer using your understan...
A: Answer - According to the question Two properties of propanone - 1.Propanone (acetone) is one of the...
Q: The molarity of 90.0% (w/w) H2SO4 solution (density = 1.80 g/mL) is [Select ] . The normality of the...
A: 28) Given, mass % of H2SO4 solution = 90.0% Density of the solution = 1.80 g/mL Molarity of the sol...
Q: First one is my data and second is what i need help with, please include calculations. a. name of co...
A: Given, Concentration of stock solution ,M1=200×10-6M
Q: Predict the probable structure of a compound with formula Cl,Mn,CgOg, where the mass spectrum includ...
A:
Q: For the chemical reaction: 2 Mg (s) + O2 (g) – → 2 Mg0 (s), if you start the reaction with 2 moles o...
A:
Q: A 1.0 g sample of a molecular compound having a molecular weight of 100,000 g/mol is dissolved in 10...
A:
Q: Discuss all the principles that can be used to facilitate acylation reactions. b) Why do aldehydes a...
A: (a) Answer - According to the question - Given - Acylation is the process of adding an acyl group t...
Q: What is the pH of a solution with 10.2mL 0.10M NH3 and 0.15mL 1.0M NaOH ? Kb = 1.8x10^-5
A:
Q: The protein catalase catalyzes the reaction 2H,0, (aq) – 2 H,O(0) + 0,(g) and has a Michaelis-Menten...
A:
Q: Explain why 1-ethylpropene is the incorrect name for the following molecule. Provide the correct nam...
A:
Q: What must be the concentration of SO 4 2− to just start precipitation of SrSO 4 from a solution whic...
A: Given, the Ksp of SrSO4 = 3.200×10-7 We have to calculate the concentration of SO42− to just start p...
Q: In the synthesis of B, two consecutive reactions are required that start from the furan to obtain A ...
A:
Q: Consider the reaction: 2 S(s) + 3 O2(g)2 SO3(g) Write the equilibrium constant for this reaction in...
A: We have to predict the equilibrium constant for given reaction.
Q: The pH of a water sample is found to be 6.3. It is titrated with 4.5mL of 0.02 N NaOH to a pH of 8.3...
A:
Q: The pH of a water sample is found to be 6.3. It is titrated with 4.5mL of 0.02 N NaOH to a pH of 8.3...
A:
Q: Calculate initial concentrations added to the test tube, the dilution factor must be taken into cons...
A:
Step by step
Solved in 2 steps with 2 images

- Show all steps leading to the final answer po. Here’s a pdf file in accordance with the topic po: https://drive.google.com/file/d/1_FnDtXCrFKSol3RNWIG_9tNQ7IxgxD6t/view?usp=drivesdkcan you answer, please? the experimental data are Reference: https://www.youtube.com/watch?v=OOXRkycKEOc&feature=youtu.be and https://www.youtube.com/watch?v=3wLJLm0QLpg&feature=youtu.bePlease answer atleast 4 sub-parts. Thank you.
- How to set up for problem 2100 g of soil is leached with a strong solution of Calcium chloride such that all the exchange sites are occupied by Ca2+. The soil is subsequently leached again with a strong solution of magnesium chloride. It is determined that the resulting 100 mL leachate contains 5000 mg of Ca2+. What is the soil CEC (cmolc/kg)? The atomic wt. of Ca is 40 g/mol.please answer in word don't image upload thank you.Chemistry An aquifer contaminated with petroleum is found to have the following component concentrations at a particular site: benzene158 ppm toluene124 ppm ethylbenzene91 ppm xylene45 ppm n-heptadecane161 ppm pristane 84 ppm Provide an estimate for the age of the spill at this site using (a) BTEX ratio and (b) nC17:Pr ratio. Show your calculations and use units throughout. Give proper s.f. for the answer.
- A 100 g soil containing 20% smectitie mineral matter was mixed with 150 mL solution containing 0.10 M Pb. CEC of pure smectitie is 110 emol(+)/kg. CEC of the soil (cmol(+)/kg) is:The %purity of a powdered crude sample of Na2CO3 containing only inert impurities is to be determined by reacting 225.0 mg of the crude sample to 10.0 mL of 3.00 M HCl solution, and bubbling the resulting CO2(g) product in water that is at exactly 29 °C. After the reaction has completed, the level of the liquid inside the eudiometer rests 4.30 cm above the water level in the beaker. The graduation on the eudiometer indicates that the trapped gas is 44.37 mL. The experiment was done under a barometric pressure of 755.2 torr. a. How many moles of CO2 were collected? b. What is the percent purity of the sample? Round off to the nearest whole numberThe %purity of a powdered crude sample of Na2CO3 containing only inert impurities is to be determined by reacting 225.0 mg of the crude sample to 10.0 mL of 3.00 M HCl solution, and bubbling the resulting CO2(g) product in water that is at exactly 29 °C. After the reaction has completed, the level of the liquid inside the eudiometer rests 4.30 cm above the water level in the beaker. The graduation on the eudiometer indicates that the trapped gas is 44.37 mL. The experiment was done under a barometric pressure of 755.2 torr. What CASE does this experiment satisfy?
- Please show steps and states (aq),(s).. etc thank youProvide the remaining data missingShow ALL calculations pls. Don't reject if you dont know how to answer I need this. a. What are the average values for vph and vmo? Include calculation of net volume and errorsb. What is/are the component/s of the soda ash sample? c. Calculate the percent composition of the component/s. No need to include error propagation, but you still need to apply Significant Figure rules.

