Q: The combustion of gasoline produces carbon dioxide andwater. Assume gasoline to be pure octane…
A: A combustion reaction occurs when fuel is oxidized by reacting with an oxidizing agent and energy is…
Q: How many grams Of water is produced when 0.45 moles of oxygen is burned ? Balance C6H12 + O2 --> CO2…
A: In a balanced chemical reaction, stoichiometry gives the relationship between the amount of…
Q: how many moles of ammonia can be oxidized by 303 grams of oxygen?
A:
Q: Nitric acid is often manufactured from the atmospheric gases nitrogen and oxygen, and hydrogen…
A: The balanced reaction of the formation of 9 kg HNO3 and theoretical yield - NH3g +2O2g →HNO3g…
Q: How much heat in kilojoules is evolved in the reaction of 1.00g of Na with H2O? 2Na(s) + 2H20(1) →…
A: Given :- 2Na(s) + 2H2O(l) ----> 2NaOH(aq) +H2(g) Delta Ho = -368.4 kJ To calculate :- Heat…
Q: How much oxygen is needed to produce 105.2 kJ via the following reaction? 2C2H6(g) + 7O2(g) →…
A: ∆H = -3120 KJ/mol How many moles of O2 will produce 105.2KJ Molar mass of oxygen = 32.0 g/mol
Q: In the combustion of heptane, CO2 is produced Assume that you want to produce 600kg of dry ice per…
A:
Q: 4) What is the heat change when 18.6 g of Hydrogen reacts with excess O2 according to the following…
A: If a reaction releases more energy than it absorbs, the reaction is exothermic and enthalpy will be…
Q: Deterioration of buildings, bridges, and other structures through the rusting of iron costs millions…
A:
Q: Given the following, how much heat is evolved when 0.025 m3 of CH6N2 is combusted at STP?
A: Combustion reaction is the reaction of any fuel or substance in presence of oxygen to form carbon…
Q: Determine the amount of heat (in kJ) associated with the production of 1.76 × 104 g of NO2 according…
A: Molar mass of nitrogen dioxide is 46 g/mol
Q: Given the following reaction, how much energy would be produced if 3.59 L of oxygen gas reacted with…
A: STP is standard pressure and temperature. At STP , 1 mol of gas is equivalent to 22.4 L of its…
Q: The major source of aluminum in the world is bauxite. Its thermal decomposition can be written as:…
A: Thermodynamic is the branch of chemistry that mainly deals with the heat transfer between system and…
Q: The fat stored in a camel’s hump is a source of both energy andwater. Calculate the mass of H2O…
A: The reaction taking place is, Number of moles of fat can be calculated as follows, 1 kg = 1000 g
Q: How many grams of CO2CO2 are produced when C3H8C3H8 is combusted in oxygen and 298.0 kJ of heat are…
A: When an atom combines with oxygen which produce carbon dioxide and water is known as combustion…
Q: How many grams of CoHe must decompose according to the following chemical equation to transfer 430|…
A: Heat of reaction is defined as difference of energy of product and energy of Reactant. If heat of…
Q: When 40.2 g of HgO (MM = 216.6 g/mol) decompose, 16.9 kJ of energy is absorbed. Calculate ΔHrxn in…
A:
Q: 7. Calculate the number of grams of CO2 that are produced by heating 116.4 grams of magnesium…
A: 60.72 gram of CO2 is produced from 116.4 gram of MgCO3.
Q: How much heat is absorbed when 20.00 g of Cij reacts in the presence of excess SO to produce CS and…
A: Enthalpy is a property of a thermodynamic system, and is defined as the sum of the system's internal…
Q: Write a balanced chemical equation for the standard formation reactic of gaseous dinitrogen…
A: We have to give balanced chemical equation for the standard formation reaction of gaseous nitrogen…
Q: 7. How many grams of carbon dioxide would be produced from the following reaction, if 175 kJ of…
A: 2CO +O2 → 2CO2 , ∆H = -566 kJ2 mol CO⇔1 mol O2 ⇔ 2 mol CO2
Q: 3. Syngas can be burned directly or converted to methanol. Calculate AH° for the conversion…
A:
Q: How many kilojoules are required when 1.2 moles of SO3 reacts? Express your answer to two…
A: The balanced equation for the decomposition of SO3 is as follows. 2SO3g→2Ss+3O2g ∆H=790KJ…
Q: Ethylene (C₂H₄) is the starting material for the preparationof polyethylene. Although typically made…
A: a) The balanced thermochemical equation for the combustion of C2H4 is - C2H4(g)+ 3O2(g) →2CO2(g) +…
Q: how much heat will be given off when 18.5 g of C2H4 is burned in oxygen to form CO2(g) and H2O(g)?…
A: Heat released and absorbed during a reaction by change in bonding is defined as heat of reaction or…
Q: N2(g) + O2(g) + 180.5 kJ/mol NO 2NO(3) side 13. On what side of the equation do we find AH ? The The…
A: Since you have posted a question with multiple sub parts, we will solve first three sub parts for…
Q: How many grams of C6H, must decompose according to the following chemical equation to transfer 430…
A: The balanced equation is :- C6H6(l) ------> 3C2H2(g) Given, ∆H = 430 KJ
Q: Acetylene can be produced by forcing calcium carbide to react with water, producing calcium…
A:
Q: Determine the amount of heat (in kJ) given off when 2.48×104g of NO2 is produced according to the…
A: Thermodynamic is the branch of chemistry that mainly deals with the heat transfer between system and…
Q: 14. Give the products of the combustion reaction of cyclohexane and balance the equation. + 0₂
A: Combustion reaction of hydrocarbons is the reaction of hydrocarbons in sufficient amount of oxygen…
Q: Use the set of three reactions shown below to answer the questions that follow.2NO(g) + O2(g) →…
A: The number of moles for a given compound or a molecule can be defined as the ratio of the mass to…
Q: What is the mole ratio for O2 gas to water for the balanced equation for the complete combustion of…
A: What is the mole ratio for O2 gas to water for the balanced equation for the complete combustion of…
Q: A 27 g aluminum foil pan is used to roast vegetables. The pan is put into a cold oven at 22 oC .…
A: The heat released or absorbed (q) by a substance when the temperature of that substance is raised is…
Q: Ammonium nitrate is an explosive and a fertilizer. Once a reaction is sparked (by some other means)…
A: Ammonium nitrate is a nitric acid salt of ammonium. It has a function as a fertilizer, an oxidizing…
Q: how did you get the numbers beside the balanced equations. such as 4 beside H2O and 2 beside N2.
A: The unbalanced equation is NH4NO2 ---------> N2 + O2 + H2O General rule is that the number of…
Q: How many moles of KCIO3 must be reacted according to the following balanced chemical reaction to…
A: The given balanced chemical equation is as follows --- 2 KClO3 (s) → 2 KCl (s) + 3 O2 (g)…
Q: Calcium carbide reacts with water to produce acetylene, C2H2. Production of 13g of acetylene…
A: Mass of acetylene is converted to moles:
Q: of CsHs is combusted to produce enough heat to raise the emperature of 150.0 g of water by 7.632 °C.
A: We are given heat produced = -4790 J And moles = 0.001561 We have to find enthalpy change (∆H ) in…
Q: 2) What is the heat change when 1.48 g of Chlorine reacts with excess phosphorus according to the…
A:
Q: of
A: Given: 2KClO₃(s)→2KCl(s)+3O₂(g) ∆H= -89.4 kJ Moles of KClO₃ to Librte -34.2 kJ of heat =?
Q: How many moles of Hz are required to produce-3751 kJ of heat in the following reaction? N: (g) +3 H2…
A: For the given reaction: N2(g)+3H2(g)→2NH3(g) ∆H°=-91.8 kJ/mol That is 3 moles of H2 are required to…
Q: How many moles of KCIO, must be reacted according to the following balanced chemical reaction to…
A: 1) Write the balanced chemical equation. 2) Compare the number of moles of reactant with the amount…
Q: Combustion reactions take place between a fuel and oxygen and produce carbon dioxide and water.…
A: In simple words combustion is burning. Combustion reaction is chemical process in which substance…
Q: 2. Green plants utilize sunlight to convert CO2 and H2O to glucose (C,H1206) and O2. All of the…
A: Photosynthesis is a process by which CO2 is absorbed by plant to convert it into glucose using solar…
Q: How much heat will be released when 1.48g of chlorine reacts with excess phosphorus according to the…
A:
Q: Calculate AG°for the combustion of propane. C3H8(g) + 502(g)→ 3CO2(g) + 4H2O(g)
A: The balanced reaction taking place is given as, => C3H8 (g) + 5 O2 (g) -------> 3 CO2 (g) + 4…
Q: The following data were obtained in a test on coal fired steam generator. The ultimate analysis of…
A:
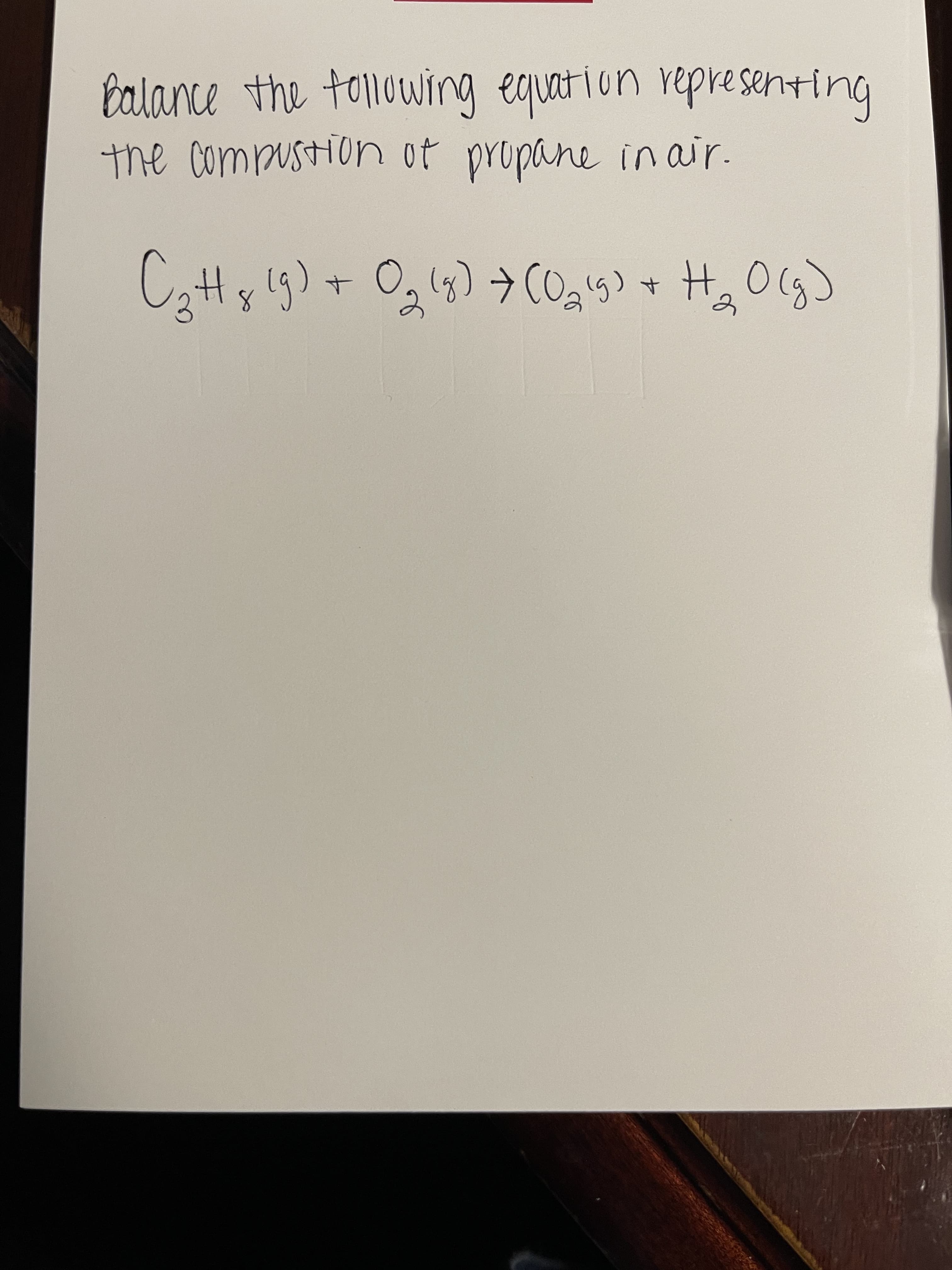
Step by step
Solved in 2 steps

- Chlorine dioxide, ClO2, is a reddish yellow gas used in bleaching paper pulp. The average speed of a ClO2 molecule at 25C is 306 m/s. What is the kinetic energy (in joules) of a ClO2 molecule moving at this speed?62 Ammonium dinitramide (ADN), NH4N(NO2)2, was considered as a possible replacement for aluminium chloride as the oxidizer in the solid fuel booster rockets used to launch the space shuttle. When detonated by a spark, AND rapidly decomposes to produce a gaseous mixture of N2,O2, and H2O. (This is not a combustion reaction. The ADN is the only reactant.) The reaction releases a lot of heat, so the gases are initially formed at high temperature and pressure. The thrust of the rocket results mainly from the expansion of this gas mixture. Suppose a 2.3-kg sample of ADN is denoted and decomposes completely to give N2,O2, and H2O. If the resulting gas mixture expands until it reaches a temperature of 100°C and a pressure of 1.00 atm, what volume will it occupy? Is your answer consistent with the proposed use of ADN as a rocket fuel?When one mole of ethylene gas, C2H4, reacts with fluorine gas, hydrogen fluoride and carbon tetrafluoride gases are formed and 2496.7 kJ of heat are given off. What is Hf for CF4(g)?
- The equation for the oxidation of phosphorus in air is P4(s) + 5 O2(g) P4O10(s). Identify the reactants and products and the stoichiometric coefficients. To what do the designations s and g refer?A Boron and hydrogen form an extensive family of compounds, and the diagram below shows how they are related by reaction. The following table gives the weight percent of boron in each of the compounds. Derive the empirical and molecular formulas of compounds A-E.In the early days of automobiles, illumination at night was provided by burning acetylene, C2H2. Though no longer used as auto headlamps, acetylene is still used as a source of light by some cave explorers. The acetylene is (was) prepared in the lamp by the reaction of water with calcium carbide, CaC2 : CaC2(s)+2H2O(l)Ca(OH)2(s)+C2H2(g). Calculate the standard enthalpy of the reaction. The Hf of CaC2 is 15.14 kcal/mol.
- In the presence of nitric acid, UO2+ undergoes a redox process. It is converted to UO22+ and nitric oxide (NO) gas is produced according to the following unbalanced equation: H+(aq)+NO3(aq)+UO2+(aq)NO(g)+UO22+(aq)+H2O(l) If 2.55 102 mL NO(g) is isolated at 29C and 1.5 atm, what amount (moles) of UO2+ was used in the reaction? (Hint: Balance the reaction by the oxidation states method.)9.65 When 0.0157 g of a compound with a heat of combustion of —37.6 kJ/mol is burned in a calorimeter, 18.5 j of heat is released. What is the molar mass of the compound?A power plant is driven by the combustion of a complex fossil fuel having the formula C11H7S. Assume the air supply is composed of only N2 and O2 with a molar ratio of 3.76:1.00, and the N2 remains unreacted. In addition to the water produced, the fuels C is completely combusted to CO2 and its sulfur content is converted to SO2. In order to evaluate gases emitted at the exhaust stacks for environmental regulation purposes, the nitrogen supplied with the air must also be included in the balanced reactions. a Including the N2 supplied m the air, write a balanced combustion equation for the complex fuel assuming 100% stoichiometric combustion (i.e., when there is no excess oxygen in the products and the only C-containing product is CO2). Except in the case of N2, use only integer coefficients. b Including N2 supplied in the air, write a balanced combustion equation for the complex fuel assuming 120% stoichiometric combustion (i.e., when excess oxygen is present in the products and the only C-containing product is CO2). Except in the case of use only integer coefficients c Calculate the minimum mass (in kg) of air required to completely combust 1700 kg of C11H7S. d Calculate the air/fuel mass ratio, assuming 100% stoichiometric combustion. e Calculate the air/fuel mass ratio, assuming 120% stoichiometric combustion.
- The heat of neutralization, Hneut, can be defined as the amount of heat released (or absorbed), q, per mole of acid (or base) neutralized. Hneut for nitric acid is -52 kJ/mol HNO3. At 27.3C, 50.00 mL of 0.743M HNO3 is neutralized by 1.00 M Sr(OH)2 in a coffee-cup calorimeter. (a) How many mL of Sr(OH)2 were used in the neutralization? (b) What is the final temperature of the resulting solution? (Use the assumptions in Question 11.)9.89 A sample of gas is 80.0% CH4 and 20.0% C2H6 by mass. What is the heat from the combustion of 1.00 g of this mixture? Assume the products are CO2 (g) and H2O (l).Write a balanced equation for the reaction between (a) dihydrogen sulfide and sulfur dioxide gases to form sulfur solid and steam. (b) methane, ammonia, and oxygen gases to form hydrogen cyanide gas and steam. (c) iron(lll) oxide and hydrogen gas to form molten iron and steam. (d) uranium(IV) oxide and hydrogen fluoride gas to form uranium(IV) fluoride and steam. (e) the combustion of ethyl alcohol (C2H5OH) to give carbon dioxide and water.











