Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter22: Surfaces
Section: Chapter Questions
Problem 22.24E
Related questions
Question
100%
Choose the best answer among the choices under each question/ statement. Thank you
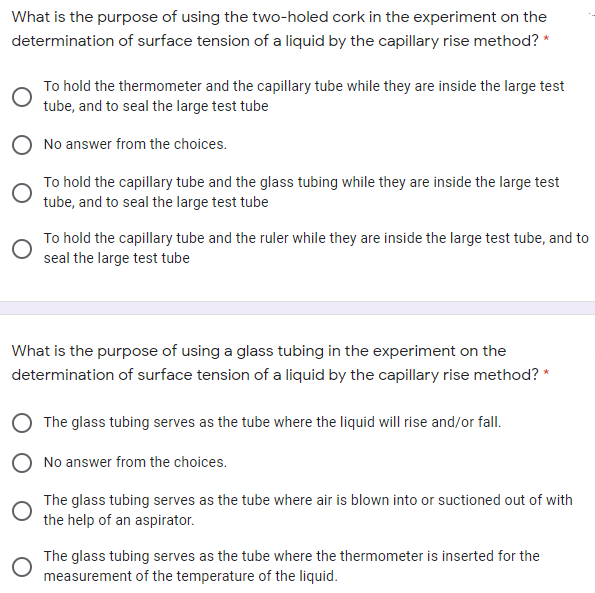
Transcribed Image Text:What is the purpose of using the two-holed cork in the experiment on the
determination of surface tension of a liquid by the capillary rise method?
To hold the thermometer and the capillary tube while they are inside the large test
tube, and to seal the large test tube
No answer from the choices.
To hold the capillary tube and the glass tubing while they are inside the large test
tube, and to seal the large test tube
To hold the capillary tube and the ruler while they are inside the large test tube, and to
seal the large test tube
What is the purpose of using a glass tubing in the experiment on the
determination of surface tension of a liquid by the capillary rise method? *
The glass tubing serves as the tube where the liquid will rise and/or fall.
No answer from the choices.
The glass tubing serves as the tube where air is blown into or suctioned out of with
the help of an aspirator.
The glass tubing serves as the tube where the thermometer is inserted for the
measurement of the temperature of the liquid.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning