Chapter22: Carbonyl Alpha-substitution Reactions
Section22.SE: Something Extra
Problem 64AP: The key step in a reported laboratory synthesis of sativene, a hydrocarbon isolated from the mold...
Related questions
Question
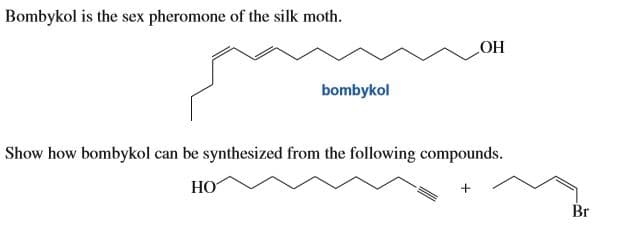
Transcribed Image Text:Bombykol is the sex pheromone of the silk moth.
НО
bombykol
Show how bombykol can be synthesized from the following compounds.
НО
Br
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning