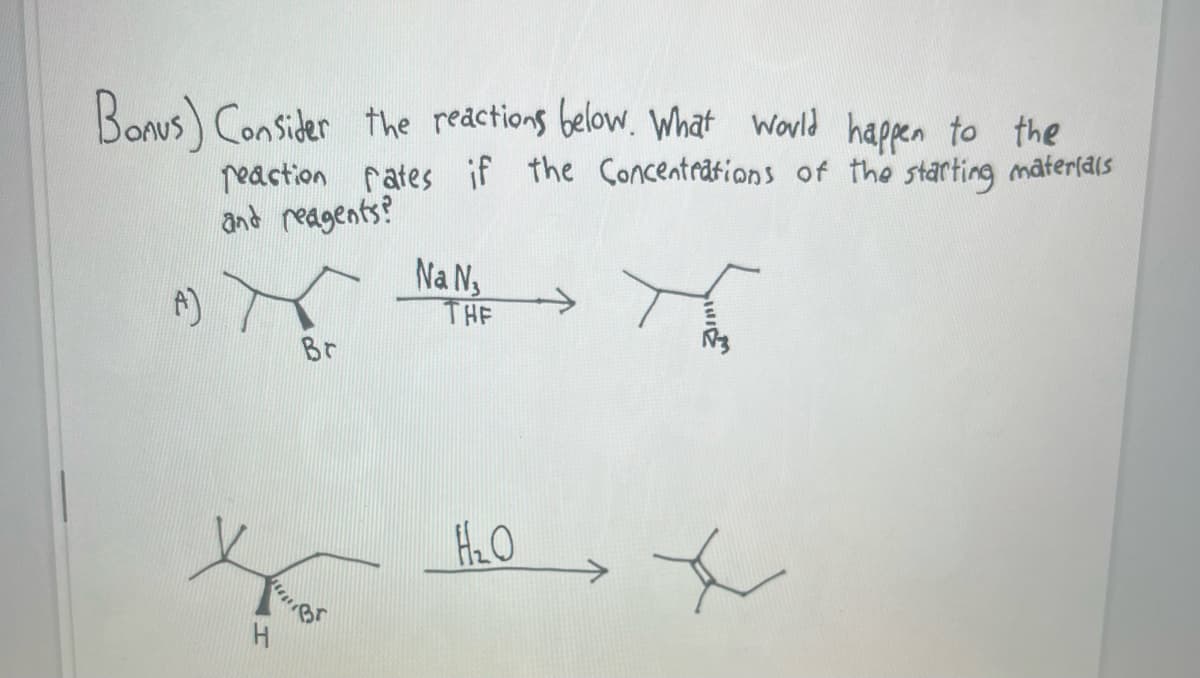
Bonus) Consider the reactions below. What would happen to the reaction rates if the Concentrations of the starting materials and reagents? (A) H Br Na N₁ THE H₂O +
Q: Draw a Lewis structure for HC C-CHO. Draw the Lewis dot structure for the given compound. Include…
A: propiolaldehyde Chemical Formula: C3H2O Exact Mass: 54.01 Molecular Weight: 54.05 m/z: 54.01…
Q: Answer the following questions for the reaction scheme below: phenol pka-10.0 OH CEN cyanida a. The…
A: The equilibrium reaction given is
Q: We react 2-propanone with 1º NaCN, H+, 2º LiAlH4 and 3º H3O+. Indicate the final product. Comment…
A: The given reaction scheme is shown belowWe have to draw the final product of the reaction.Also, we…
Q: An exothermic reaction is taking place in a constant volume reactor that contains a cooling coil.…
A: k1feed concentration CA= 4 mol/LConversion =50%Volumetric flow rate = 2L/hrInitial Temperature=…
Q: d) 6.63 x 10" 18. The chemical shift of CDC13 appear at 2130 Hz by using 300-MHz spectrometer. This…
A: Formula: Chemical shift ( in ppm ) = Nnn
Q: Please do not give solution in image format thanku The bond between two hydrogen atoms (a covalent…
A: Hydrogen atom has only one electron. It condition of a nucleus and one electron nucleus is…
Q: 2. The net equation for a given voltaic cell is: Sn (s) + 2 Agt → Sn²+ + 2 Ag (s) Write the two…
A: The question is based on the concept of electrochemistry.We need to identify half cell reactions and…
Q: Write the two pertinent balanced reactions (standardization and sample analysis) 2. Calculate the…
A: Standardization Reaction:5Na2C2O4 + 2KMnO4 + 8H2SO4 → 2MnSO4 + 10CO2 + 5Na2SO4 + K2SO4 + 8H2OSample…
Q: 1) Which of the following elements is a metal? A) nitrogen 2) Which of the following elements is a…
A: 1.nitrogen is a non metalic element Which belongs to group 15.2.fluorine is also a non metal which…
Q: (A) Which of the following is the strongest base? NH (B) OO (C) (D)
A: Answer:-This question is answered by using the simple concept of determination of strength of the…
Q: If a tree dies and the trunk remains undisturbed for 1.160 × 10⁴ years, what percentage of the…
A:
Q: a. How many grams of iron(III) nitrite, Fe(NO2)3, are present in 1.82 moles of this compound? grams…
A:
Q: d) Acetone and oxygen can react in a combustion reaction with the equation below. Balance the…
A: Part dMoles of acetone = 2.5Balancing of reaction and moles of CO2 produce ?Parr eComparison of bond…
Q: SHOW WORK) Calculate ΔH when 652 kg of H2O2 decomposes, in the following reaction: 2 H2O2(l) ⟶ 2…
A: Given data 2 H2O2(l) ⟶ 2 H2O(l) + O2(g) ΔH = -196.1 kJThe change in enthalpy of the reaction…
Q: Calculate the solubility of CuCO3 in water at 25 °C. You'll find Kp data in the ALEKS Data tab.…
A: From standard data, Ksp for CuCO3 = 1.4 × 10-10
Q: Complete a seperation scheme for biphenyl, sand, and salicylic acid
A: A mixture of biphenyl, sand and salicylic acid is to be separated.
Q: 1. The region of electromagnetic spectrum for nuclear magnetic resonance is: a) Microwave b) UV-rays…
A: IR spectroscopy is a technique used to identify chemical compounds based on how infrared radiations…
Q: One of these reactions to prepare a carboxylic acid from an alkyl chloride fails. Which? O O Ya CI…
A: Tertiary alkyl halide not give SN2 reaction.
Q: Question A10 Complete the missing blank in the following question: Rf value is (a) (c) (d) Distance…
A: A question based on analytical processing. 4 options statements about Rf value are given, from which…
Q: Draw a Lewis structure for the molecular formula: COBr2 This molecule only contains carbon,…
A: COBr2C is central atom.
Q: describe a specific nanotechnology that exploits spintronics. Explain why for that technology that…
A: Spin-based magnetic memory, commonly referred to as spintronics memory or spin memory, is one…
Q: Step 1 Step 2 What is the rate law for the proposed mechanism? H₂(g) + 2NO(g) N₂O(g) + H₂O(g) (slow)…
A: The rate law of a reaction is determined by the slow step of a reaction. Since slow step is the rate…
Q: Calculate the concentration of the products of the following equilibrium when the equilibrium…
A: Equilibrium constant= 12.3 Equilibrium concentration of NOBr2 = 2.17
Q: he following could enter into H-bonding interactions with water? (Select all that apply.) -…
A: When a hydrogen atom is bound to a strongly electronegative atom (such oxygen, nitrogen, or…
Q: Sulfur atoms combine to form molecules that have different numbers of atoms depending on the…
A: given reaction S8 (g) ⇌ 4 S2 (g) at equilibrium positionThe equilibrium constant (KC ) =…
Q: (a) Define the terms 'functional group' and 'unsaturated hydrocarbon'. (b) Name the following…
A: Define Functional group and unsaturated hydrocarbons. Naming of organic compounds Skeletal structure…
Q: Aqueous Pb(ClO3)2 and aqueous Nal are mixed together. What mass of precipitate, in grams, will form…
A: The question is based on the concept of reaction stoichiometry.we need to calculate mass of…
Q: Which substance is not readily oxidized by acidified potassium A. propan-1-ol propan-2-ol propanal…
A: -> potassium dicromate can oxidize 1° alcohol to Carboxylic acid.-> potassium dicromate can…
Q: Which of the following molecules is cyclopentyldiethylamine?
A: The given name of the molecule is cyclopentyldiethylamine
Q: 13. Curium - 245 is an alpha emitter. Write the equation for the nuclear reaction and identify the…
A: Equation for alpha emission for CuriumParticle X in the Equation Equation for beta emission for…
Q: What is the net ionic equation for the reaction of aqueous acetic acid and aqueous potassium…
A: For writing the net ionic +equation for the reaction between acetic acid and potassium hydroxide, we…
Q: Provide IUPAC names for the following structuras. b. Br
A:
Q: 2. Give the resonance structures for the species given below. HO NH Which atom is more basic in the…
A:
Q: If y=B*e(A*x) then ln(y)=
A: Mathematical functions are used in physical chemistry to calculate a number of measurements. Natural…
Q: Which reason best describes why the magnitude of dipole of bond A would be less than the magnitude…
A: The reason why the magnitude of the dipole of bond A (Ca-O) would be less than the magnitude of the…
Q: SO3H 0+6+6+6 OA. X: HCl, Y: NaOH, Z: H₂SO4 X B. X: H₂SO4, Y: NaCl, Z: HC1 ONa O C. X: H₂SO4, Y:…
A:
Q: A solution containing 1.21 g of AgNO, was mixed with a solution containing 1.19 g of NaCl: c) Write…
A: c) The balanced chemical equation for the reaction of AgNO3 and NaCl is:AgNO3 + NaCl → AgCl +…
Q: a. Name the following molecules: Br CI b. On the structures above, put an asterisk (*) on any…
A: a). 1. 5-isopropyl-3-methyloctane Chemical Formula: C12H26 Exact Mass: 170.20 Molecular Weight:…
Q: EXERCISE A one-particle, one-dimensional system has the potential energy function V = Vo for () ≤ x…
A:
Q: We measure the absorbance of a protein solution with the concentration 0.006M, which absorbs at…
A: Concentration (c) of protein solution = 0.006 MPath length (l) of cuvette = 1 cm molar absorptivity…
Q: B. Draw the Lewis electron-dot structure for each ionic compound that will be formed by the…
A:
Q: Aqueous hydrobromic acid (HBr) will react with solid sodium hydroxide (NaOH) to produce aqueous…
A:
Q: Determine the oxidation state and d configuration of the iridium metals and the bond order for the…
A: Iridium belongs to group-9 of the d-block in the periodic table. Electron configuration : [Xe] 4f14…
Q: 1. Which of the following solutions will be the best buffer at a pH of 9.26? (Ka for HC2H302 is 1.8…
A: Buffer solutions are solutions that resist pH change when small amount of acid or base is added to…
Q: Classify the ionic compounds as soluble or insoluble. (a) zinc chloride Solubble (b) chromium(III)…
A: Based on general solubility rules, we can classify the following ionic compounds as soluble or…
Q: Isn't the answer 0.038 atm?
A: Since, beginning measure of H2 gas isn't given in atm, so balance of the response will move towards…
Q: Compare and contrast the physical and chemical properties of: • propan-1-ol and propan-2-ol; •…
A: Propan-1-ol and propan-2-ol, also known as n-propanol and isopropanol, respectively, are two isomers…
Q: Structure A H H3C H3C H H CI H H- H CH3 CH3 O ¹.H CH3 CO₂H Br Br Structure B H3C H H H3C Br H H CH3…
A: Constitutional isomers :- The isomers of a compound which have same molecular formula but differ in…
Q: Order the compounds by rate of reaction against electrophilic aromatic substitution:…
A: To order the compounds by rate of reaction against electrophilic aromatic substitution (EAS), we…
Q: Eto &a -CO₂Et || OEt 1) NaOEt, EtOH 2) H3O+ III IV CO₂Et
A: Dieckmann condensation is a reaction where in a cyclic β-ketoester is formed from diesters. This is…
Step by step
Solved in 3 steps with 1 images

- For the reaction in part "b", please explain which products are the major and minor products. Also explain which are the kinetic and thermodynamic products.Define mechanism of haloform reaction ?The phenyl group, C6H5, is known to be an ortho/para-directing group. (a) With that in mind, predict theproduct of the reaction shown here. (b) Justify why it is an ortho/para director by examining the ortho, meta,and para arenium ion intermediates that would beformed during the course of the reaction.
- can someone help me with this? Make a comparison table between the reaction mechanism SN1 and SN2, take into account thereaction order, nature of the substrates, solvents used, nature of the nucleophile, stereochemistry,kinetics, and other characteristics that you consider relevant.Make the mechanism of the following reactions, remember to be detailed in the steps and write all the necessary intermediaries in each case.Explain the Summary of Factors That Determine Whether the SN1 or SN2 Mechanism Occurs ?
- Define mechanism Acid-Catalyzed Halogenation at the α Carbon ?Provide the mechanism of the reaction between the molecules above under acidic conditions as well as the structure of the product. Assume a large excess of the first molecule (one on top) If 2.08 g of the molecule on the bottom are used, what is the minimummass of the molecule on top that is needed to make the productthat you drew in question 3 above?Give the clear handwritten mechanism of the reaction !!!!!!
- Help! Explain in detail please. Thank you! (A) For the following set of reaction conditions use the table to identify which mechanism is favored by each factor and which is favored overall (B) Give the major product(s) of the reactionWhat is the mechanism of the reaction below.What are the relevant transition states for this reaction?

