Q: Using the Bohr model, determine the lowest possible energy, in joules, for the electron in the Li2+ ...
A: The equation for the calculation of energy for the electron is given below, ...
Q: What is the density of a substance if 5.000 mL has a mass of 5.025 grams? Select one: O A. 1.005 g/m...
A: given the volume = 5.000 ml, and mass = 5.025 grams
Q: Give the structure corresponding to following name. (Z)-3-isopropylhept-2-ene
A: Z isomer means highest priority groups are in same side of the double bond. The corresponding struct...
Q: Among the following compoundsthe compounds that will NOT form enamine with R2NH is ? Give reasons to...
A: Aldehydes and ketones are carbonyl compounds that undergo characteristic, nucleophilic addition reac...
Q: Chemistry Question
A: INTRODUCTION: GC-MS also called as gas chromatography-Mass spectrometry is a simple technique used t...
Q: When 1.0 g of fructose, C6H12O6(s), a sugar commonly found in fruits, is burned in oxygen in a bomb ...
A: Given: Mass of fructose =1.0g Rise in temperature of calorimeter = 1.58 °C heat capacit...
Q: Calculate the molar solubility of XA2 (s) (K.p - 9,5×10) in 0.15M XNO3 (aq). HINT - What is the Comm...
A:
Q: If a solid ionic compound AB dissolves in water, what is found in the solution besides water molecul...
A: If an ionic solid compound dissolves in water, then it dissociates into cations and anions and these...
Q: For the following reaction of A -> 2B. The 1/[A] plot is linear with y=0.045x+200. Find the conce...
A:
Q: Sketch the general shapes of the s, p, and d orbitals.
A: An atom consists of certain sub-atomic particles which are arranged in atom in such a way that overa...
Q: Chemistry Question
A: Constitutional isomers are the isomers that have the same molecular formula but different connectivi...
Q: Determine the concentration of H3O+ to the correct number of significant figures in a solution with ...
A: INTRODUCTION: pH is defined as the negative logorithm of concentration of H+ ion.
Q: Draw the structure of benzonitrile :
A: Cyclic organic compounds possess a cyclic ring having 3 or additional C-atoms. These can be simple u...
Q: Can someone please help with my homework and be clear for answer box thank you
A: The equation for the Heisenberg uncertainty principle is given below, ...
Q: Place the methyl groups by drawing the axial and equatorial positions on the cyclohexane structure i...
A:
Q: What volume in mL of 0.1152 M NaOH is required to titrate 15.00 mL of 0.1073 M acetic acid (HAc)?
A:
Q: Does it matter how much water is used to dissolve the unknown monoprotic acid? Explain your answer. ...
A: Yes , It matter how much water is used to dissolve the unknown monoprotic acid to make it into solut...
Q: Determine the kinds of intermolecular forces that are present in each element or compound.a. N2 b. N...
A: Intermolecular force: The attractive force that withholds two molecules is called as intermolecular ...
Q: The blood serum acetone for a person is determined to be 18.7 mg of acetone per 100 mL of serum. Ex...
A:
Q: Does it matter if you use cis or trans stilbene as a starting material? Would this give you the same...
A: Cis- and trans-isomerisation of stilbene can take place as photo-chemical isomerisation.
Q: what is the correct total charge for each of the following molecules: 1. Lysine 2. Arginine 3. The d...
A: lysine +1 arginine +1 The dipeptide composed of arginine & threonine +1 The tripeptide Glu...
Q: Which of the following react with ammonia to form a black precipitate? AgCl PbCl...
A: Here the following is Hg2Cl2 which will give black precipitate with ammonia.
Q: A pH change of 0.2 units requires the addition of 0.05 mL of NaOH. You are probably at what point in...
A: For the acid-base titration, the possible curve is shown below.
Q: In a GCMS experiment, a compound shows a 1:1 height ratio of its M peak at 122 mass units and its M+...
A:
Q: How many mL of 0.115 M NaOH will react with exactly with 28.7 mL of a 0.210 M solution of H2SO4 (3 S...
A:
Q: Determine whether each of the following S,2 reactions is reversible or irreversible. (a) Br (b) (c) ...
A: a) The given reaction is; It is mentioned that the reaction would follow the SN1 mechanism pathway....
Q: Complete the table: Solute Mass of solute Moles of solute Volume of solution Molarity of solution...
A: Molarity (M) of a solution is defined as the number of moles of solute dissolved per volume of the s...
Q: How many non-equivalent carbons exist in the following compound? (Give a number) Ph
A:
Q: How many grams of NH3 can be obtained from 12.0 g N2 using thẻ TölloWIng synthesis of ammonia? N2 + ...
A: According to the reaction, 3H2(g) + N2(g) ------------------:>2NH3(g) 12.0 gm...
Q: I need help with this question about Factor
A: Molarity is a concentration term, which is can be calculated as number of moles of solute in volume ...
Q: Identify the conjugate acid and base pairs in the following reaction. NH3 + H2S ® HS– + NH4+...
A: Hey, since you have asked multiple question, we will solve the first question for you. If you want a...
Q: I have a hard time with this factor
A:
Q: Match the functional group to its name. If an alcohol or amine determine whether these are primary, ...
A: A functional group is a single atom or group of atoms within an organic compound that is responsible...
Q: How many different signals would we expect in the 13C NMR spectrum of the following compound? (Give ...
A: In 13C NMR equivalent carbons will show one signal. So we have to find the no of equivalent carbon a...
Q: Need help with this both questions
A: Since you have posted 2 questions in a single session we are entitled to answer first question only....
Q: How many C4H10 molecules are contained in 9.213 g of this compound? How many hydrogen atoms?
A: Formula for calculation of number of molecules is:
Q: A compound has a molecular formula of C5H802 and exhibits the following 13C NMR spectrum. 8 171.48, ...
A: SOLUTION: Step 1: The given compound contains 5 C13 NMR signals, among the given structures B and C ...
Q: How many miles of O atoms are in 18.1 mol if CH2O
A: Solution stoichiometry involves the calculation of concentration of solutions in the given condition...
Q: A solution containing 1.50 g of iron (III) chloride reacts with a solution containing 3.00 g sodiu...
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and s...
Q: A young man recently passed away from unknown causes. The newly hired forensic officer, Citlali, rec...
A: Given information: Mass of sample = 9.002 g Moles of carbon dioxide = 0.450 mol Moles of water = 0.2...
Q: 6. a) What is the pH of pure water at 35.0 °C? At this temperature Kw = 2.042 x 10-14. b) Based on t...
A: (a) The chemical equation for self-ionization of water is: Thus, At 25oC, Kw = 1.0 * 10-14 and [H...
Q: What is the pH of a 0.10 M H2C2O4 solution that has the stepwise dissociation constants Ka1 = 5.9 × ...
A:
Q: A metal ion with 2+ charge has 23 electrons and forms a compound with a halogen ion that contains 17...
A:
Q: Consider the titration of a 40.0 mL of 0.189 M weak acid HA (Ka = 2.7 x 10⁻⁸) with 0.100 M LiOH. a. ...
A: “Since you have posted a question with multiple sub-parts, we will solve the first three sub-parts f...
Q: During an electrolysis experiment, the following species are present in solution: H+, CH3COOH, CH3CO...
A: Concept: Reducing Agent- Species that reduce the other substance and gets oxidized itself in the rea...
Q: Draw the mechanism for how y-curcumene is biosynthesized from farnesyl pyrophosphate. y-Curcumene
A: The hydrocarbon part of farnesylpyrophosphate is made up of three isoprene units, and γ-curcumene ha...
Q: What mass of NaCl is present in 50.0 mL of 1.05 M NaCl solution?
A: given : volume = 50.0 ml = 0.05 L Molarity = 1.05 M we have to find mass of NaCl ? Moles = Molarity ...
Q: Nanotechnology, the field of building ultrasmall structures one atom at a time, has progressed in re...
A:
What reagents and reaction conditions are needed for the attached conversions?
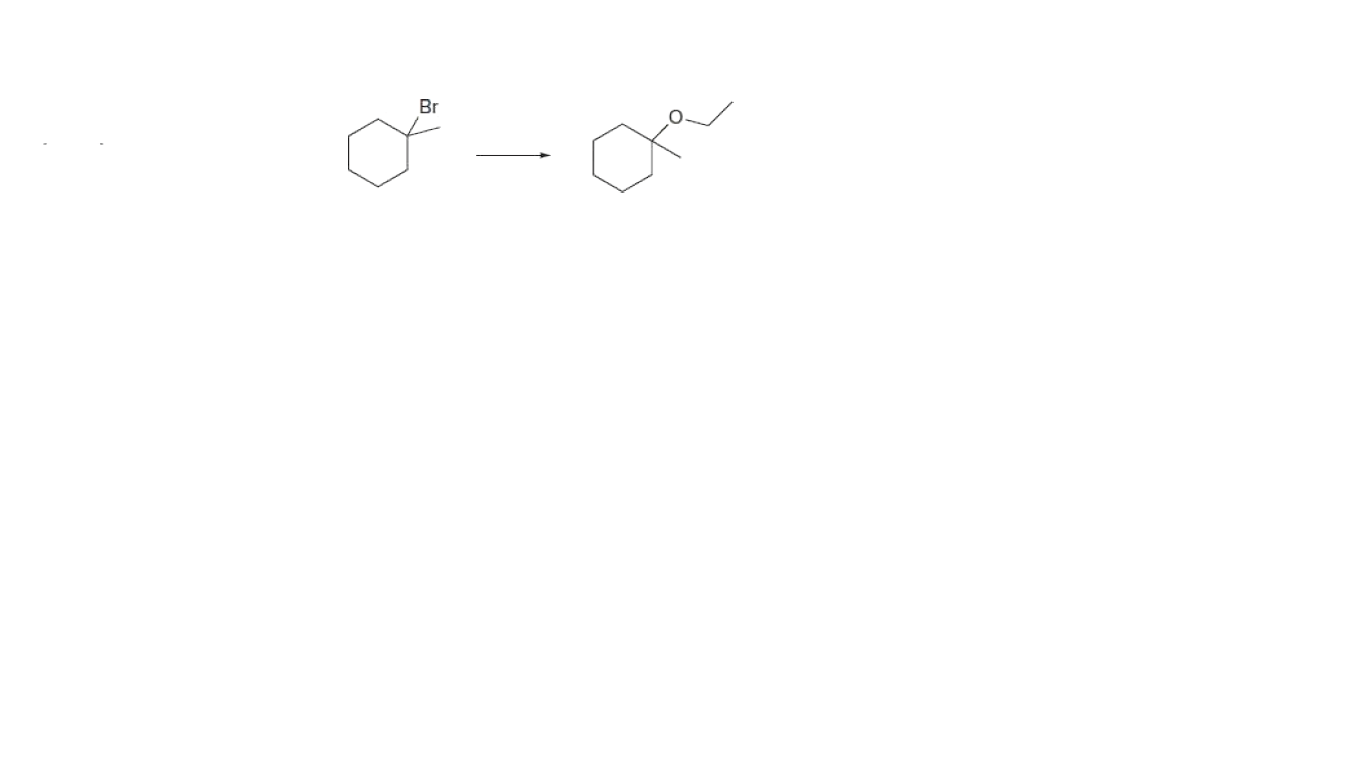
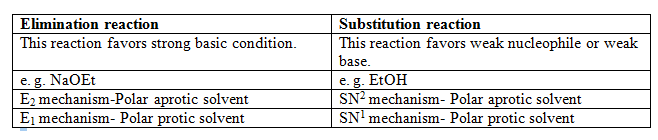
The difference between Elimination reaction and Substitution reaction is tabulated as follows-
Step by step
Solved in 2 steps with 2 images


