Chapter20: Carboxylic Acids And Nitriles
Section20.SE: Something Extra
Problem 27MP: Naturally occurring compounds called cyanogenic glycosides, such as lotaustralin, release hydrogen...
Related questions
Question
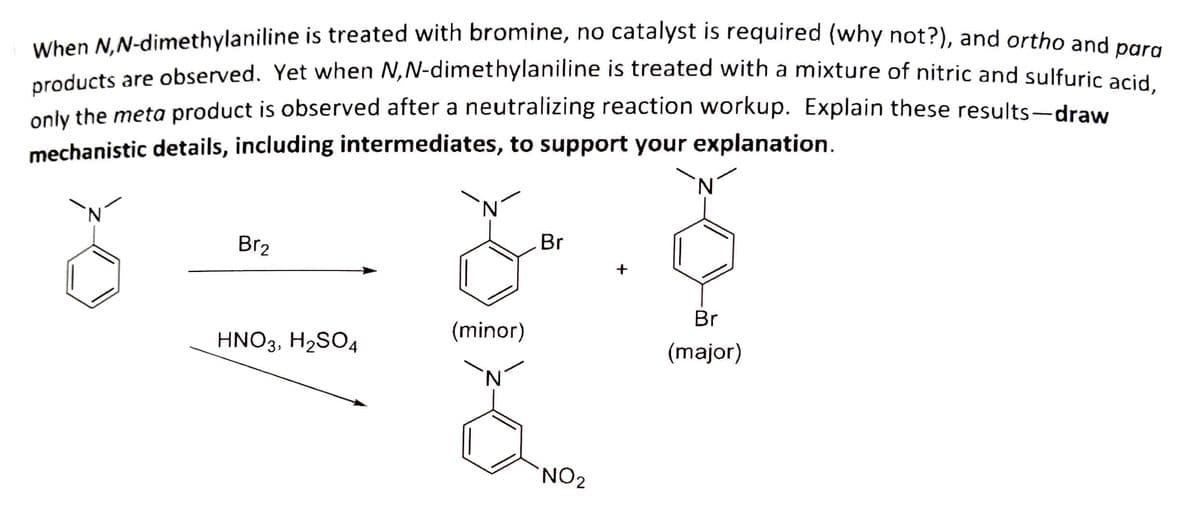
Transcribed Image Text:When N.N-dimethylaniline is treated with bromine, no catalyst is required (why not?), and ortho and nara
educts are observed. Yet when N,N-dimethylaniline is treated with a mixture of nitric and sulfuric acid
only the meta product is observed after a neutralizing reaction workup. Explain these results-draw
mechanistic details, including intermediates, to support your explanation.
Br2
Br
+
Br
HNO3, H2SO4
(minor)
(major)
NO2
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning