Q: If ammonium chloride was mixed into the sand in a sandbox, then would this be of any concern?
A: Ammonium chloride- AlCl3 Aluminium chloride and sand are mixed in physical forms. They form a mixtur...
Q: 2. Propose an efficient synthetic route for each the following multi-step reactions after demonstrat...
A: Since you have asked multiple questions, we will solve the first one for you. For remaining ones, ki...
Q: Detemine the empirical formula of the hydrated salt from question 1 (A 5 gram sample of hydrated ber...
A:
Q: If an electron from a hydrogen atom drops from n=7 quantum state down to n= 3 state, what named seri...
A:
Q: 1. How many MOLECULES of sulfur tetrafluoride are present in 1.23 moles of this compound ? |molecule...
A: 1. We have to calculate the number of molecules. 2. We have to calculate the number of moles.
Q: 1.5. The resistance of a platinum wire is found to be 11.0002 at the ice point, 15.247 2 at the stea...
A:
Q: An astronomer measures the distance from Earth to a newly-discovered asteroid. The distance is 2.38 ...
A: • The values provided in the question are:- i) Distance between earth and newly discovered astero...
Q: Energy absorbed when an electron is added to an isolated atom in the gaseous state „A O Energy relea...
A: Given that, Energy absorbed when an electron is added to an isolated atom in the gaseous state.
Q: Find the molecular formula for a compound which contains 7.5 g potassium, 6.8 g chlorine, and 9 g ox...
A:
Q: What is the correct formula of Lead(II) phosphate? O Pb3(PO4)2 O Pb(PO4)2 O PBPO4 O Pb3P2 O Pb2PO4
A: From given Intially lead and phosphate formula is given and after criss crossing with their respect...
Q: Part C How many grams of the excess reactant remain after the limiting reactant is completely consum...
A: limiting reagent is the reactant, that reacts completely in the reaction. The other reactant remains...
Q: Hydronium Ion Computation of Lewis Structure: (see the picture)
A: A question based on atomic structure, which is to be accomplished.
Q: For the reaction: 2 NO(g) + Cl2(g) = 2 NOCI(g), Keg = 2500 %3D If the initial pressures are: P(NO) =...
A: The given equilibrium reaction is as follows: 2NOg + Cl2g ↔ 2NOClg Keq = 2500 The initial pressure o...
Q: When [A] is doubled, the rate doubles; when [B] is doubled, the rate quadruples; when [C] is doubled...
A: We have to predict the change of rate.
Q: If [OH" ] = 5.2 x 10-4 M, what is the pH ? pH РОН [H* ] [OH ] ? 5.2 x 10-4 M O -3.3 O 10.7 O -10.7 O...
A: given, [OH-] = 5.2 ×10-4 M
Q: How do you know if you should add more sodium sulfate when you are drying your ether layer containin...
A:
Q: Please convert this name into the structure: (2R,3R,4S,5S,6R)-2-(((2S,3S,4S,5R)-3,4-dihydroxy-2,5-bi...
A:
Q: Carbon disulfide, CS2, is a colorless, highly flammable liquid used in the manufacture of rayon and ...
A: We will convert number of moles into mass by using mole concept.
Q: Protein A has a binding site for ligand X with a dissociation constant, Ka, of 3.0 × 10-' M. Protein...
A: We have to predict the protein that has great affinity for the ligand.
Q: Use the References to access important values if needed for this question. Then the following molecu...
A: Balanced equation :- A chemical equation is said to be balanced if number of atoms of different ele...
Q: Carbon disulfide, CS2, is a colorless, highly flammable liquid used in the manufacture of rayon and ...
A:
Q: an unknown element is a mixture of isotopes 120X and 122X. the average atomic mass of X is 121.03amu...
A:
Q: Place the following cations in order from the lowest to the highest hydration enthalpy
A: Hydration energy is directly proportional to charge and inversely proportional to the size of the io...
Q: A chemistry student needs 70.0 g of dimethyl sulfoxide for an experiment. By consulting the CRC Hand...
A: Given Mass of dimethyl sulfoxide = 70 gram density = 1.10 gram.cm-3 or 1.10 gram/cm3 Volume = ?
Q: a) reaction of 1-butene with Cl2/H2O b) reaction of 1-butene with H+/H2O c) reaction of 2-methyl-2-b...
A:
Q: piece of copper with a mass of 6 g is placed in a solution of silver (I) nitrate containing excess A...
A:
Q: Calculate the pH of the following acid-base buffers a. 100 mL of 0.025 M formic acid and 0.015 M sod...
A:
Q: OH HCI 1 Br HBr CI HC1 3 H20 2.
A:
Q: most t ONE () of the following, then (a) draw twn possible resonance hybrids, and (b) circle the sta...
A: Resonance structures: structure which have least separation of charges is the best contributing stru...
Q: Hydronium Ion Moleculary Polarity: Formal Charges: Generic Formula: (ex. AX3E) Molecular shape: Ele...
A: Hydronium is the simplest form of oxonium ions. All the acidic aqueous solutions contain protonated ...
Q: Follow these steps: Calculate the % chloride in each sample (heated and unheated) and then use the p...
A: Formula of the coordination complex is given to be: [Cox(NH3)yClz1]Clz2
Q: Suppose for the following reaction the initial pressures are: 2 SO3 (g) = 2 SO2 (g) + O2 (g) Initial...
A:
Q: Aqueous hydrobromic acid (HBr) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium b...
A: Calculate the % yield of water in the given question----
Q: Q3: Calculate (a) the standard-state potential is (b) the equilibrium constant, and (c) the potentia...
A:
Q: (True/False) 1. If the name of a polyatomic ion ends in -ate and is added with an acid, the resulti...
A:
Q: MASS SPECTRUM COMPOUND C Abundance Scan 473 (4.445 min): 311DEHJ.Dldata.ms 2000000 57.1 1900000 1800...
A: Molecular ion peak: Peak of highest mass that corresponds to the MW of molecule Base peak: Peak of h...
Q: Compound H2O NH3 Conjugate Acid Conjugate Base HS CH;OH HSO4 H2PO4
A:
Q: When conducting a recrystallization what should generally be true when comparing the compounds melti...
A: Recrystallization is an experimental technique of separating pure compound from the solution contain...
Q: In class questions: Q97: - Use the provided equation to calculate the following: KMNO, (aq) + HCI (a...
A:
Q: chlorine trifluoride 1. what is the formal charge?
A: Chlorine trifluoride consists of 1 chlorine atom and 3 fluorine atoms so the chemical formula become...
Q: Which of the following statement/s is/are true for steady state approximation? Select all the correc...
A: In chemical kinetics, the steady-state approximation is a method used for the determination of rate ...
Q: Draw the structure of this cyclic disulfide: CH;CHCH,CH2CHCH3 SH SH 2,5-hexanedithiol (Hint: Draw th...
A: 6 membered cyclic ring form..
Q: Record the measurement using the correct number of significant figures: 8. -7 mL
A: For a concave meniscus, the correct volume will be read at the lowest level of the curve where for a...
Q: For the same molecule, which of the images corresponds to the correct Newman projection along the in...
A:
Q: What would be the osmotic pressure at 17°C of an aqueous solution containing 1.85g of sucrose (C12H2...
A:
Q: You have a sample of granite with density 2.8 g/cm3. The density of water is 1.0 g/cm3. What is the ...
A: Density of granite = 2.8g/cm3 Density of water = 1.0g/cm3 Density is mass÷volume Specific gravity is...
Q: Write the empirical formula of at least four binary ionic compounds that could be formed from the fo...
A: Write the empirical formula of ionic compound formed by given ions----
Q: Which among the given statement is NOT true about precipitation? ++ Pb Ва ++ and Sr ++ Most sulfates...
A: There are some solubility rules by which solubility of ionic compounds in water can be determined. F...
Q: What is the wavelength of light emitted by a hydrogen electron that transitions from energy level n=...
A:
Draw two chair conformation and project which configuration is stable
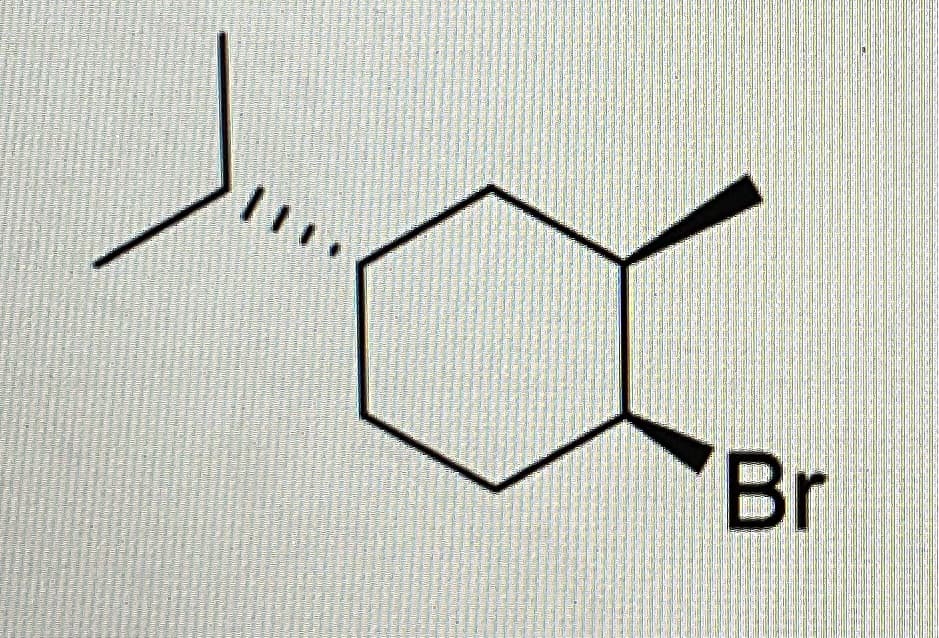
Step by step
Solved in 2 steps with 2 images


