Q: The work function of Au is 5.1 V. For an uncharged Au electrode, what is the electrochemical potenti...
A: Work function of Au = 5.1V We have been given an uncharged electrode. We have been asked to compare ...
Q: Given that, in our bodies, when we metabolize molecules to derive energy from them, we are oxidizing...
A: Note : Energy is released when electron moves from low electron affinity center moves to high electr...
Q: 2. A marble ball placed on an analytical balance giving a mass of 12.5640 g. This marble ball is pla...
A:
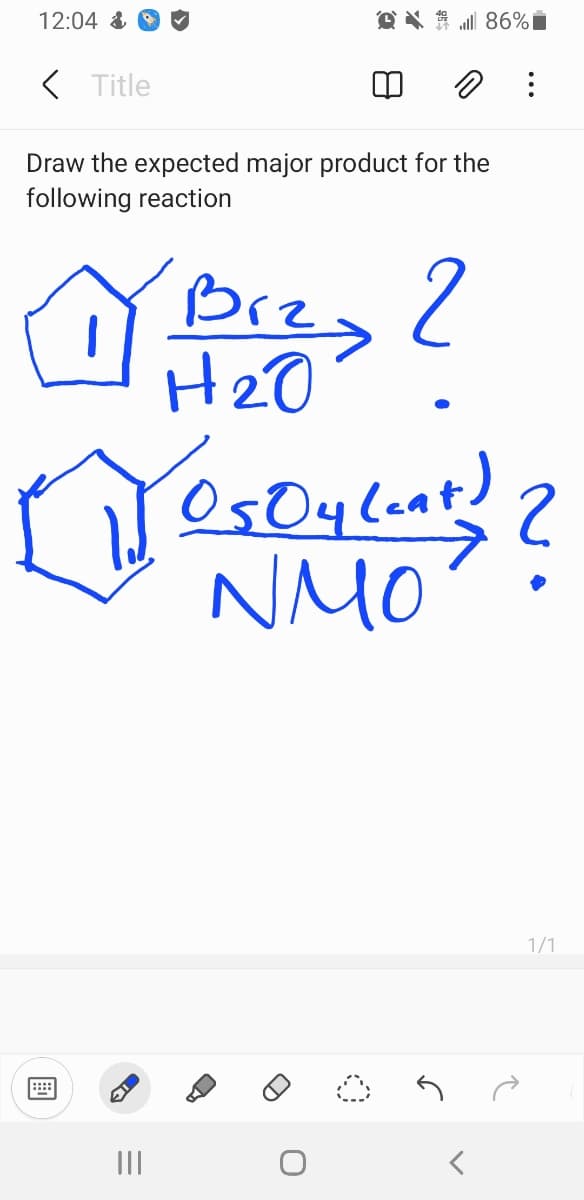
Q: H-
A: Given,
Q: Write the MBE, PBE and CBE for the following: a. 0.15M H2CrO4 b. 0.35M KClO4 c. 0.50M NaHSO4
A: Charge Balance Equation (CBE): It is based on electroneutrality principle. The molar concentration o...
Q: Millon’s test: Put 5 drops of Millon’s reagent to phenol. Notice the color formed. What is the mol...
A:
Q: b. ion-dipole forces d. pole-dipole forces 7. Which of the following intermolecular forces is presen...
A: Intermolecular forces of attraction: As per our guideline we have to answer first question only.
Q: If the reactants on the left side of a chemical equation are CsHa +50, what could be the products in...
A: Combustion Reaction: When a hydrocarbon reacts with Oxygen, it undergoes combustion, usually produci...
Q: Provide the reagents required.
A: Answer of the question given below,
Q: Identify the bonds formed between the following pairs of atoms as either covalent or ionic: zinc and...
A: Bond's present between the given compounds.
Q: In a calorimeter experiment, NaOH and HNO3 underwent a neutralization reaction and produced 0.075mol...
A: Neutralization reactions are generally exothermic and thus ΔH is negative.
Q: Calculation the pH of the following systematically. Make sure to include the pertinent reactions and...
A:
Q: What is paper chromatography please include the importance of using it
A: Introduction : Chromatography can be defined as a technique for separating different component of s...
Q: A 1.00-m solution of acetic acid, CH3COOH, in benzene has a freezing point of 2.96°C. Use the data i...
A:
Q: Determine the equilibrium constant for the following reaction at 25 deg. C: 2I- (aq) + Br2(l) --> I...
A: The equilibrium constant for the above reaction is given below
Q: A quantity of O2 initially at 300K is compressed reversibly and adiabatically from 50 L to 10 L. Ass...
A: Relation between CP and CV can be given as: CP-CV=R …(1) For an adiabatic process, the r...
Q: Mo(PMes)3(C6HS)2
A: The given complex is of the molybdenum which is a a d-group metal (transition metal) and formed comp...
Q: Он
A:
Q: Draw the condensed stuctural diagram for the molecule: 4-amino-5-isopropoxyhex-trans-2-enal
A: Given molecule is : 4-amino-5-isopropoxyhex-trans-2-enal Draw the condensed structural diagram for ...
Q: a) Propose a synthesis route to prepare the following product from the given starting material. Note...
A: Here we have to convert 1-phenylprop-2-en-1-one to 1-methyl-1,3-diphenylpropane-1,3-diol in a multis...
Q: When a 49.0-g sample of an unknown compound is dissolved in 500.g of benzene, the freezing point of ...
A: Given, Mass of unknown sample = 49.0 g Mass of benzene solvent = 500.0 g = 0.5 Kg Freezing point of...
Q: A solution contains 1 mole of liquid A and 3 moles of liquid B. This solution has a vapor pressure o...
A: 1st option is correct that is This solution exhibits a positive deviation from Raoult's Law.
Q: What would be the best reagents to carry out the following Fischer esterification? OCHS он ethanol e...
A:
Q: Calculate the weight in grams of ethylene glycol (antifreeze) that would be needed for an automobile...
A:
Q: The peroxydisulfate ion (S2O8-2) reacts with the iodide ion in aqueous solution via the reaction: S2...
A: (a) Answer -
Q: Which of the following statements is false? * The percent yield = x 100% The limiting reactant is co...
A: In a chemical reaction, the limiting reactant is the reactant that gets consumed first and the other...
Q: Which is the summary of a large amount of scientific information?
A: Scientific Law or say laws of science is based on the behalf of repeated experiments and observation...
Q: The researchers are requested to prepare a 5.23 mol dm-3 of concentration of Sodium Thiosulphate for...
A: sodium thiosulfate solution that has to be prepared its concentration is 5.23 mol/dm3 or 5.23 mol/L....
Q: e limiting reactant.
A:
Q: Pre-lab Exercise I: 1. Calculate the theoretical number of moles of hydrogen gas produced. 2. What w...
A: 1. From the given reaction ,it is given that 2 mole of H2O2 produced 1 mole of H2 gas .so theoretic...
Q: The determination of copper in a sca water sample gave a mean value of 77.81 ug/L and a standard dev...
A:
Q: Consider the following data on some weak acids and weak bases: acid base Ka K, name formula name for...
A: We have to find the pH according to Handerson Hasselbatch equation. pH = pKa + log [Salt]/[Weak Aci...
Q: How many grams of Mgla are produced by the reaction of 25.0 g of Mg with 25.0 g of l:? *
A:
Q: Provide the proper IUPAC or common name for the following compound. [do not use italics in Canvas]
A: IUPAC nomenclature is used for naming the organic compound as recommended by international union of ...
Q: Which of the following are sets of quantum numbers for orbitals are possible? a) n = 1, l = 1, m...
A: The correct option is:
Q: Postassium dichromate oxidizes potassium iodide in acid solution liberating iodine use half reaction...
A:
Q: At 906.4 °C, the equilibrium constant K, is 4.81 × 10-5. What is the value of K?
A: Given, Kp = 4.81x10-5 Temperature, T = 906.4 °C
Q: Write the equilibrium constant expression Kofor the following reaction. 2H,0(g) O2(g) + 2H, (g)
A: The given reaction is, 2H2O(g) ---> O2(g) + 2H2(g)
Q: For a particular reaction, AH° is 20.1 kJ/mol and AS is 45.9 J/(mol-K). Assuming these values change...
A: Given: ∆H° = 20.1kj/mol = 20100 j/mol ∆S° = 45.9 j/k-mol For reaction to be spontaneous, ∆G should...
Q: A nitrogen sample occupies 246 mL at STP. If the same sample were collected over water at 25°C and 6...
A: For the phase change of the ideal gas the temperature, pressure, and volume of the initial state and...
Q: 5.31. Generate a curve for the titration of 50.00 mL of a solution in which the analytical concentra...
A: In the given problem, the titration curve for the titration of 50.00 mL mixture of 0.1000 M HClO4 an...
Q: Lodu.edu/ultra/courses/_393452 1/cl/outline v Question Completion Status: A student wants to determi...
A: Ethyl acetate and water are colourless while copper (l) bromide is green in colour.
Q: The vapor pressure of water at 20.0 °C is 17.5 torr. At that temperature, what would be the vapor pr...
A: In your case, you know that the vapor pressure of pure water at 20℃ is 17.5 torr. This means that a...
Q: Part A A,H° = 121 kJ molA,S = - 262 J K-' mol-' :T = 292 K Express your answer as an integer. Templa...
A:
Q: An important industrial process for synthesizing the ammonia used in agricultural fertilizers involv...
A:
Q: Which of the following is NOT true about "yield"? * The percent yield is the ratio of the actual yie...
A: Quantitative analysis is branch of chemistry that deals with the determination of the amount of cons...
Q: Give the IUPAC name of the compound below.
A: The IUPAC name of the compound can be written on the basis of the main carbon chain, functional grou...
Q: Why are Be and Mg metals used as deoxidizers?
A: Be and Mg metals are used as deoxidizers.
Q: d). pОH %3D12.3 [oH]= 5.011823e-13 110-14 =.99니068구e-15
A:
Step by step
Solved in 2 steps with 2 images

- In light of your answer to Problem 30-40, explain why a mixture of products occurs in the following reaction:predict the stating materials, reagents, or products of the followinf reactionscmplete the reacting by adding necessrt reagents. write in thapce provided. make sure to write regents in chemical formula . n separate regents with comma & space (ex. h2so6, h202)
- I’m currently trying to write a lab report for the synthesis of dimolybdenum tetraacetate [Mo2(O2CCH3)4] from the reaction of molybdenum hexacarbonyl, Mo(CO)6 in glacial acetic acid and acetic anhydride under a nitrogen atmosphere, involving the difficult formation of a quadruple bond and requires high heat and long reaction time (approximately 20 hours). But these are the question I’m stumped on: 1. Why need the reaction be done under nitrogen? We also added dichlorobenzene and hexanes during the reaction. 2. Explain the purpose of dichlorobenzene and hexanes. 3. Why does the reaction take 20 hrs?Predict the product(s) of the reaction below: Al₂O3 → which is it below O2 AI AI + O AI +O2Phenols (ArOH) are relatively acidic, and the presence of a substituent group on the aromatic ring has a large effect. The pKa of unsubstituted phenol, for example, is 9.89, while that of p-nitrophenol is 7.15. Draw resonance structures of the corresponding phenoxide anions and explain the data.
- Predict the product(s) and provide the mechanism for each reaction below.Use your knowledge of directing effects, along with the following data, to deduce the directions of the dipole moments in aniline and bromobenzene.Given: Mass spec for 4-acetylbiphenyl. Knowing that 196 m/z is the major product, how would you assign 181 m/z and 152 m/z? (i've also attached a picture of the reaction scheme)
- As reaction of (CH3)2CO with LIC≡CH followed by H2O affords compound D, which has a molecular ion in its mass spectrum at 84 and prominent absorptions in its IR spectrum at 3600−3200, 3303, 2938, and 2120 cm−1. D shows the following 1H NMR spectral data: 1.53 (singlet, 6 H), 2.37 (singlet, 1 H), and 2.43 (singlet, 1 H) ppm. What is the structure of D?In the following cases rearrange the compounds as directed : (Delhi 2010)(i) In an increasing order of basic strength :C6H5NH2, C6H5 N(CH3)2, (C2H5)2NH and CH3NH2(ii) In a decreasing order of basic strength :Aniline, p-nitroaniline and p-toluidine(iii) In an increasing order of pKb values :C2H5NH2, C6H5 NHCH3, (C2H5)2NH and C6H5NH2Review the mechanism of oxymercuration shown in Figure 8-3 on page 230, and then unite the mechanism of the alkoxymercuration reaction of 1-methyl- cyclopentene with ethanol. Use curved arrows to show the electron flow in each step.

