Q: 9. If 6.2 mL of 0.0160 M KOH solution must be added to 5.0 mL of 0.240 M CaCl₂ solution to just form…
A: Solubility product is the multiplication of molar solubility of the constituent ions of the…
Q: A 10.00 g charcoal brick from a barbeque measures a C-14 decay rate of 5050 disintegrations per gram…
A: 1 gram of C-14 sample has a disintegration rate of 921 disintegrations per hour initially. Initial…
Q: s-Balance Equations Skeleton
A: Since you have asked multiparts, we will solve the first three subparts for you. If you want any…
Q: SEATWORK: Calculate the hydroxide ion concentration of a 0.0750r NH₂ solution. The Kb is 1.75 x…
A: Given data,Molarity of NH3=0.075 MKb of NH3=1.75×10-5
Q: The equilibrium constant, Ke, for the following reaction is 1.80x10-2 at 698K. 2HI(g) =H₂(g) + I2(g)…
A: The equilibrium reaction given is 2 HI (g) ⇌ H2 (g) + I2 (g) The equilibrium constant, Kc =…
Q: What reagent(s) need to be added to benzene to synthesize the following product? O OCH3MgBr OCH3OCH…
A:
Q: Which of the following statements best explains why the titanium complex, [Ti(H₂O)614+, is…
A: Titanium, Ti is a d-block element with an atomic number of 22. The below electronic configuration.…
Q: Draw a structural formula for 2-cyclopenten-1-ol. • Show stereochemistry only if given in the name.…
A: The structural formula for 2-cyclopenten-1-ol is:
Q: + Water "softeners" remove metal ions such as Ca²* and Fe³* by replacing them with enough Na ions to…
A: Given data,Volume of solution=7.96×102LMolarity of Ca2+=0.0357 MMolarity of Fe3+=0.00151 M
Q: Molecule CI P. cl H H H CI + CI-Pd-CI CI H. H 2- H Highest symmetry rotation Any LC₂ ? Any oh? Any…
A:
Q: activation energy (kJ mol-1) for th
A:
Q: Consider the following reaction: 2HI(g) H₂(g) + 1₂(g) If 3.53 moles of HI(g), 0.621 moles of H₂, and…
A: Kc is the ratio of the equilibrium concentrations of product over equilibrium concentrations of…
Q: How many moles of NH4Cl must be added to 1.0 L of a 0.050-M solution of NH3 to give a buffer…
A:
Q: 10 A 3.40-g sample of a mixture of naphthalene (C, H.) and pyrene (CH) is dissolved in 35.0 g…
A: Given: The mass of the mixture of naphthalene and pyrene = 3.40 g The mass of benzene (solvent) =…
Q: 3. Write down the all products for the following reaction. Also write a detail step by step…
A: Note : Esters react with Grignard reagent produces tertiary alcohols. ( see below).
Q: Select the correct reaction to represent the dissolution of MgCO3(s) (magnesium carbonate, a salt)…
A:
Q: the shape of the model
A:
Q: This question may contain multiple correct answers. Make sure to select all correct answers. Which…
A: The nature of salta is determined from the nature of acid and base from which they form. It is found…
Q: When ionic bromination of cyclohexene is carried out, the product is trans-1,2-dibromocyclohexane.…
A: When ionic bromination of cyclohexene is carried out, the product is trans-1,2-dibromocyclohexane.…
Q: Describe the evaporation of a liquid in terms of kinetic molecular theory. Criteria: I can explain…
A: To explain: Evaporation of a liquid in terms of KMT.
Q: For the titration curve below, select one option from the list below for each of these three…
A:
Q: Write the balanced equation for the reaction in which Fe(s) is placed in HClO4(aq). Do not include…
A: Here, we have to write the balanced equation for the reaction between Fe and HClO4.
Q: The following reaction can be carried out in two steps. Choose the correct reagents i) and ii) to…
A:
Q: If more Brions were added to a saturated HgBr2 solution, then the reaction below: HgBr₂(s) Hg2+ (aq)…
A: The answer is given below
Q: A protein is Multiple Choice O O O a polysaccharide. one of the units making up a nucleic acid. an…
A: Proteins are high molecular weight complex organic containing C, H, O, N, S etc atoms.
Q: What reagent(s) need to be added to benzene to synthesize the following product? CH3 Br CH3OCH 3…
A:
Q: Using the Heisenberg uncertainty principle, calculate the uncertainty in position, Ax, for: (a) an…
A: According to Heisenberg uncertainty principle, ∆x·∆p≥h4π where, ∆p=m∆vx
Q: which is most thermodynamically stable menthol or neomenthol
A: The stability of a cyclic hexane compound is determined based on the substituent present on the…
Q: How does the solubility in water give you information about the bonding in a substance? Criteria: I…
A: To explain: The nature of bonding in the substance based on its solubility in water.
Q: What reagent(s) needs to be added to cause the following transformation? ? OH₂, Pt Br2 O NaOCH 3…
A:
Q: For each of the designated spaces in the table below (green letters), match the sign of the…
A:
Q: What is the solubility of Cr(OH)₃ at a pH of 9.80? (Ksp Cr(OH)₃ is 6.70 × 10⁻³
A: Given - - > pH = 9.80 Ksp of Cr(OH)₃ = 6.70 × 10-31 We know that, 14 - pH = - log[OH-] 14 - 9.8…
Q: A student prepared three nickel samples in the lab but forgot to label them. The samples that were…
A:
Q: in the blanks poorest leaving group among the hali because it is the and the most
A: Leaving ground is the substance which easily leaves in substitution reaction. Weak bases are good…
Q: Study the following sketch of a molecular orbital (MO) in a homonuclear diatomic molecule. This MO…
A: The explanation is given below-
Q: 10. Draw the line structure for the organic compound given as a Lewis structure below. Is this…
A: Ester: An ester is a group where one oxygen atom is doubly bonded to a carbon atom which is singly…
Q: mine the pH of a 0.52 M lithium nitrite solution. (Ka for nitrous acid = 4.5 x 10-4 7.00 12.18 8.53…
A: Given, the Ka for nitrous acid = 4.5×10-4 We have to calculate the pH of 0.52 M lithium nitrite…
Q: What are the E/Z configurations of the following compounds? CI CH₂Br H3CO CH=CHNH2 HO -OH CC13 HO…
A:
Q: 10. Draw the line structure for the organic compound given as a Lewis structure below. Is this…
A:
Q: is –2.32 °C, then what is the molar mass of the solute (in g/mole)?
A: If the freezing point of an aqueous solution containing 7.162 g of solute dissolved in 72.297 g…
Q: The only true hydrocarbon polymer found in nature is Multiple Choice O O O rubber. polystyrene.…
A:
Q: Rank the following complexes from smallest to largest d-orbital splitting i) [Fe(CN)6]³- ii)…
A: The d-orbital splitting energy can be calculated by the following relation: Splitting…
Q: What will its volume
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Which of the following is a strong acid? O acetic acid hydrochloric acid hydrofluoric acid O…
A: Multiple choice questions are given here.
Q: The diprotic acid H₂A has Ka1 = 1.58*10-5 and Ka2 = 1.47*10-6. If the initial concentration of H₂A…
A:
Q: Calculate the amount of heat needed to melt 13.2 g of ice (H₂O) and bring it to a temperature of…
A:
Q: Fill in the blanks: The E1 mechanism is most favored by a alkyl halide and a solvent.
A: E1 means elemination is Unimolecular and in this mechanism is most favourable in 3o type halocarbon…
Q: What volume of a 4.00 × 10-4 M FeSCN2+ solution would be needed to prepare 50.0 mL of a 7.20 × 10-5…
A: Here, we have to find the volume of a 4.00×10-4 M FeSCN2+ solution that would be required to prepare…
Q: Polystyrene results from the polymerization of H H H A. CH-C dad B. CH₂-C C. CH=C H CH3 CN H D. CHẮC…
A:
Q: B C O NH3 COO NH3 COO -N Ninhydrin Aq. HCI & Heat ? + CO₂ + purple compound ?+ ?
A:
4
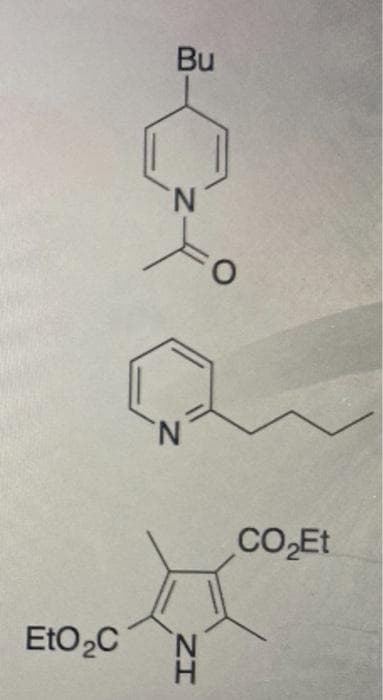
Step by step
Solved in 4 steps with 3 images


