Physical Chemistry
2nd Edition
ISBN:9781133958437
Author:Ball, David W. (david Warren), BAER, Tomas
Publisher:Ball, David W. (david Warren), BAER, Tomas
Chapter6: Equilibria In Single-component Systems
Section: Chapter Questions
Problem 6.60E
Related questions
Question
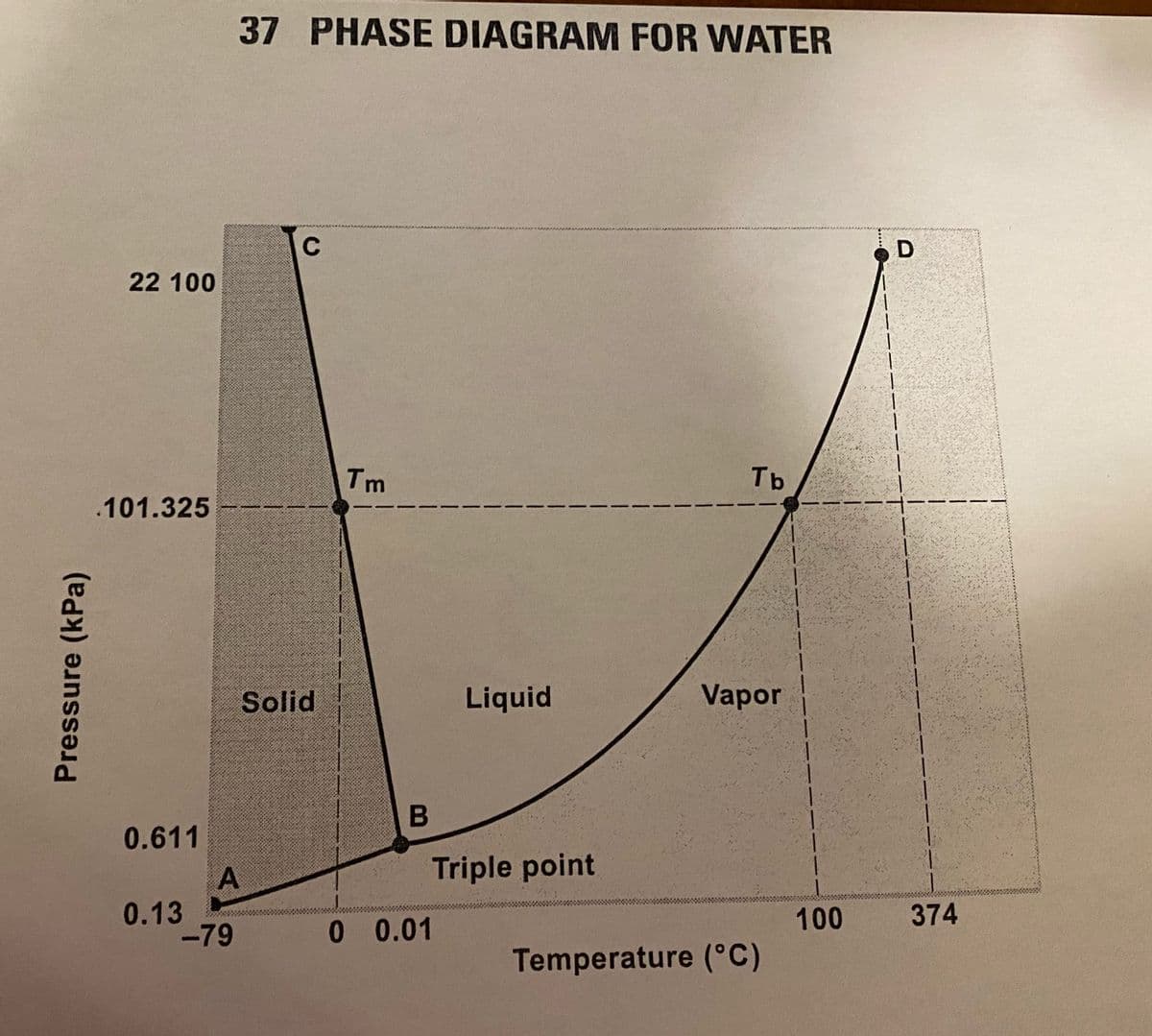
Transcribed Image Text:37 PHASE DIAGRAM FOR WATER
C
22 100
Tm
Tb
.101.325
Solid
Liquid
Vapor
0.611
Triple point
A
0.13
-79
100
374
0 0.01
Temperature (°C)
Pressure (kPa)
....

Transcribed Image Text:Name
Date
Class
Chapter 17
TRANSPARENCY 37
PHASE DIAGRAM FOR WATER
1. In this phase diagram for water, what is being plotted on the x-axis and on the y-axis?
2. What does line AB represent? Explain in terms of equilibrium.
3. What does curve BD represent?
4. What does line BC represent?
5. What is point B called? Explain what is meant by the term.
6. What is point D called? Explain what is meant by the term.
7. What is the temperature at the triple point? What is the pressure at the triple point?
8. What is the value of T, the normal melting point of ice?
9. What is the value of T, the normal boiling point of water?
10. What is the pressure 101.325 kPa equal to?
11. Is the slope of the solid-liquid equilibrium line, BC, positive or negative? What unusual fact about
water does this reflect?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,