Substance X is known to exist at 1 atm in the solid, liquid, or vapor phase, depending on the temperature. Additionally, the values of these other properties of X have been determined: melting point 25. °C boiling point 55. °C enthalpy of fusion 9.00 kJ/mol enthalpy of 49.00 kJ/mol vaporization 2.00 g/cm (solid) density 37. J.Kmol 1 1 (solid) olo 1.70 g/mL (liquid) heat capacity 51. J·Kmol (liquid) -1 Ar 43. J-к "mol (vapor) You may also assume X behaves as an ideal gas in the vapor phase. Suppose a small sample of X at 0 °C is put into an evacuated flask and heated at a constant rate until 15.0 kJ/mol of heat has been added to the sample. Graph the temperature of the sample that would be observed during this experiment. 100 90- ? 80- 70 60 50 40 30 20 10 10 15 heat added (kJ/mol) temperature (*C)
Substance X is known to exist at 1 atm in the solid, liquid, or vapor phase, depending on the temperature. Additionally, the values of these other properties of X have been determined: melting point 25. °C boiling point 55. °C enthalpy of fusion 9.00 kJ/mol enthalpy of 49.00 kJ/mol vaporization 2.00 g/cm (solid) density 37. J.Kmol 1 1 (solid) olo 1.70 g/mL (liquid) heat capacity 51. J·Kmol (liquid) -1 Ar 43. J-к "mol (vapor) You may also assume X behaves as an ideal gas in the vapor phase. Suppose a small sample of X at 0 °C is put into an evacuated flask and heated at a constant rate until 15.0 kJ/mol of heat has been added to the sample. Graph the temperature of the sample that would be observed during this experiment. 100 90- ? 80- 70 60 50 40 30 20 10 10 15 heat added (kJ/mol) temperature (*C)
Chemistry: Principles and Reactions
8th Edition
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:William L. Masterton, Cecile N. Hurley
Chapter9: Liquids And Solids
Section: Chapter Questions
Problem 20QAP
Related questions
Question
Please help with graph!
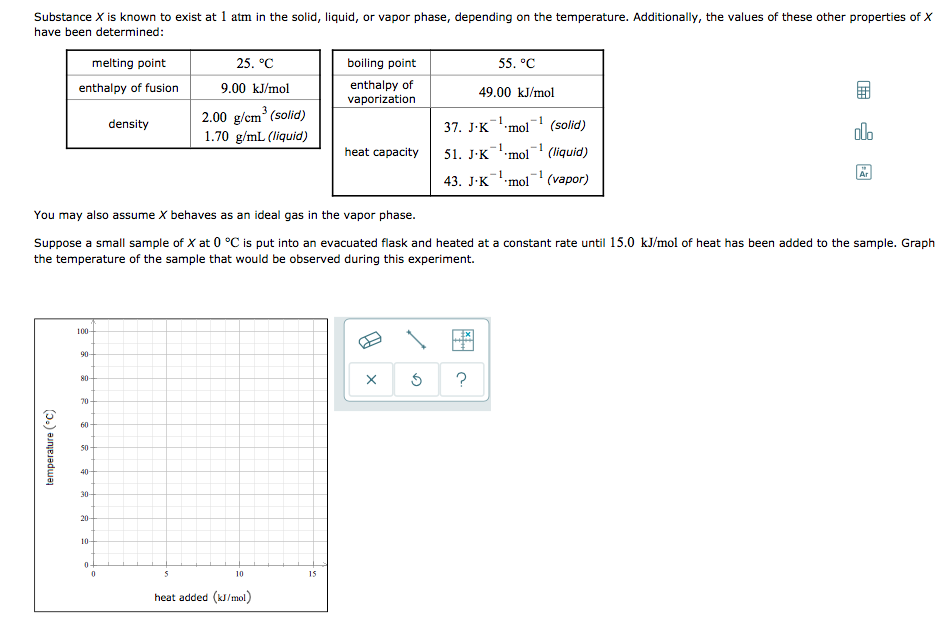
Transcribed Image Text:Substance X is known to exist at 1 atm in the solid, liquid, or vapor phase, depending on the temperature. Additionally, the values of these other properties of X
have been determined:
melting point
25. °C
boiling point
55. °C
enthalpy of fusion
9.00 kJ/mol
enthalpy of
49.00 kJ/mol
vaporization
2.00 g/cm (solid)
density
37. J.Kmol
1
1
(solid)
olo
1.70 g/mL (liquid)
heat capacity
51. J·Kmol (liquid)
-1
Ar
43. J-к "mol (vapor)
You may also assume X behaves as an ideal gas in the vapor phase.
Suppose a small sample of X at 0 °C is put into an evacuated flask and heated at a constant rate until 15.0 kJ/mol of heat has been added to the sample. Graph
the temperature of the sample that would be observed during this experiment.
100
90-
?
80-
70
60
50
40
30
20
10
10
15
heat added (kJ/mol)
temperature (*C)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 5 steps with 7 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning