Q: Both pictures are of one problem
A: The movement of water molecules from a region of lower concentration of solution to a higher…
Q: 3.) 1.) DO", D20 (excess) :ö:0 a) 1.) b.) 1.) c.) 1.) D3C CD3 2) 2) 2) 3.) 3.) 3.) 6 CD3 O
A: Applying concept of reagents and reaction .
Q: a) Explain matrix effect.
A: “Since there are multiple questions and it is not mentioned that which one has to be solved so I am…
Q: 3v point group is given as Co
A: In group theory, representation of higher dimension can be reduced in to representation of lower…
Q: + -> 2le Li why iP instead of +]= 22 Why I P instead of -{e or°B???
A: During a nuclear reaction total atomic number and mass number should be balanced. And if coefficent…
Q: Match the following to their electron configuration. 1. F 2. Se²- 3. Cu* 4. K+ F- Drag and drop…
A: Electronic configuration represents the number of electrons present in an atom or ion. The…
Q: Attached problem
A: As per rules only three subparts can be answered. In thin layer chromatography (TLC) Retention…
Q: determine the Kb for A- at 25C . The Ka for HA is 1.49 x 10^-10
A:
Q: a) Find the Linear density for FCC (111) direction assuming the atomic radius R-0.230mm b) Find the…
A: Solutions Given that A) Find the linear density for FCC B) Find the linear…
Q: Derire the eapluession for in terms 8 actinity. 0-4 AG the term
A: The study of flow of matter is called thermodynamics. Gibbs free energy is defined as the…
Q: A solution is made by combining 1.000 ml of 0.1000 M Cd(NO)2 with.000 ml. of 1.000 M HBr and 3.000…
A: The equilibrium constant can be written as the ratio of concentration of products to the…
Q: Calculate the lowest energy Δo for a solution
A: Orgel diagram is only applicable for weak field ligands and it shows the relative energies of…
Q: <Chapter 7 Part 1 Quantum - Problem 7.27 9 ot 9 llaviaw | Constants |'enodic lable A"Co molecule,…
A: The smallest quantum of energy that can be absorbed by given molecule in rotational excitation is…
Q: What are the types of MSDS forms? Explain each of them.
A: MSDS - Material Safety Data Sheet. It contains information about the various types of hazards…
Q: Elaborate in detail about shrinking core model.
A: The shrinking core model is used to describe situations in which solid particles are being consumed…
Q: what the energy of neutron is suitable for crystal diffraction [the atomic spacing between two…
A: Given The atomic spacing between two bonded atoms is generally around
Q: Tuz köprüsü Zn Ag 0,1 M 0,01 M Zn2 (suda) Ag (suda) 100 mL 200 mL 1. kap 2. kap Zn (suda) + 2e +…
A:
Q: Create a Benesi-Hildebrand Plot. To do this, plot the data of 1/(F-F0) (y-axis) vs. 1/[CD]O…
A:
Q: mcdT dq T AS = = mcln T/T
A: Entropy change is a definite quantity independent of the path followed for the change and depends…
Q: Type of UV transition
A: Type of UV transitions
Q: s Cu(TPP) active in UV-Vis AND why? EXPLAIN YOUR REASON. If you replace Cu2+ with Zn2+ and you have…
A: The UV-Visible Principle The absorption of ultraviolet or visible light by chemical compounds…
Q: MCQ 22: The name given to sub-shells are s-sharp, p-principal and d for A. different B. diffuse C.…
A: The given options are,
Q: The effect on motion when an electric potential is applied and positive ions drag a group of…
A: Retardation effect: Retardation is the delaying the process. Thus by Retardation effect is when…
Q: Find Five candidates of RR Lyrae type with magnitude under +11?
A: RR lyare With periods ranging from a few hours to two days and optical brightnesses that normally…
Q: for subparts a, c, d, e, f
A: Lithium aluminum hydride reduces ester into alcohol. Carboxylic acid and alcohol react in presence…
Q: 15. Lenses are coated with thin film to reduce a. Diffraction d. Refraction C. Reflection foronce…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: 1. Determine the Ecell values for the following: a. Tl (s) | Tl*(aq) || Sn²+(aq) | Sn(s) b. Zn(s)|…
A: Disclaimer: Since you have posted a question with multiple sub-parts, we will solve the first three…
Q: 5) Discuss the sources of error in your experiment. 6) List three practical applications of MgO and…
A: 1,2,3 and 4 is correct. 5. Sources of error a. At the time of measuring weight , fault in digital…
Q: hedict the poduct that will be frimed by the neachon below. Colzlag) + Mn (5)>
A: Here aqueous cobalt iodide CoI2 and solid manganese Mn have been reacted. We have to predict the…
Q: Match the items in the left-hand column to the items in the right-hand column by writing the…
A:
Q: I, Meort, 60c 92%
A: Deprotection of acetal.
Q: What is a Buchner funnel and what is it used for?
A: A Büchner funnel is a glass or plastic piece of laboratory equipment which is in shape of a funnel.…
Q: What is inductive effect with example and its applications
A: We need to explain inductive effect.
Q: 20. In current-sampled polarography the polarographic wave of which metal ion will appear first? Pb…
A: The modification of a classical DC polarography experiment was developed for the reduction of effect…
Q: TOF ion trap orbitrap
A: Mass spectrometry is an analytical technique.
Q: Interpret IR data
A: A question based on IR spectra that is to be accomplished.
Q: 5, 14 12 11 16 15 -y mond shown above give the point indices for atom 15. (grs) format. So if…
A:
Q: used for?
A: This question is related to x ray techniques.
Q: 3, "The DeNo) valurin [ Ru (up), (ni) x]" are x - MecN, z:3, VNo = (970 cm!! X=cl, Z=2, VCNO1927 cm?…
A: Electrophilic behaviour is dependent upon IR Streching Frequency which is explained below:
Q: Slits separated by 0.02 cm are used to form a first order line on a screen 100 cm away. The lines…
A: It is a double slit experiment question. The solution is provided in step 2.
Q: Find the first-order Stark effect for a hydrogen atom in state n=3 Note: Please show complete…
A: Given Find the first-order Stark effect for a hydrogen atom in state n=3
Q: Direction: Cite 5 elements that can be magnetized and 5 that can nullify magnetism. MAGNETIZERS…
A: Magnetisers can be defined as the substances that can impart magnetism to other substances.
Q: (0.30M) + 24gCl PbSO4 + 248s + (0.20M)
A:
Q: 25 substarce D1toF(KS/mel) G(KJ(mcl) s°(SImel.Kl) P2 PC13 g POCsg Clay 144.3 -2881 -542.2 218.1…
A: Given: So(P2(g)) =218.1 J/mol.K So(Cl2(g)) =222.96 J/mol.K So(PCl3(g)) =311.7 J/mol.K
Q: NEITEHR OF THE ANSWERS SHOULD BE PSI -60 PLEASE IF YOU PUT THAT I WILL DISLIKE AND If you copy ur…
A:
Q: if peter wants to remove the effect of the matrix because of the interference it causes, he should…
A: A question based on tools in analytical chemistry that is to be accomplished.
Q: Greb e the mmoleule thead pola Bd explein nohy? or
A:
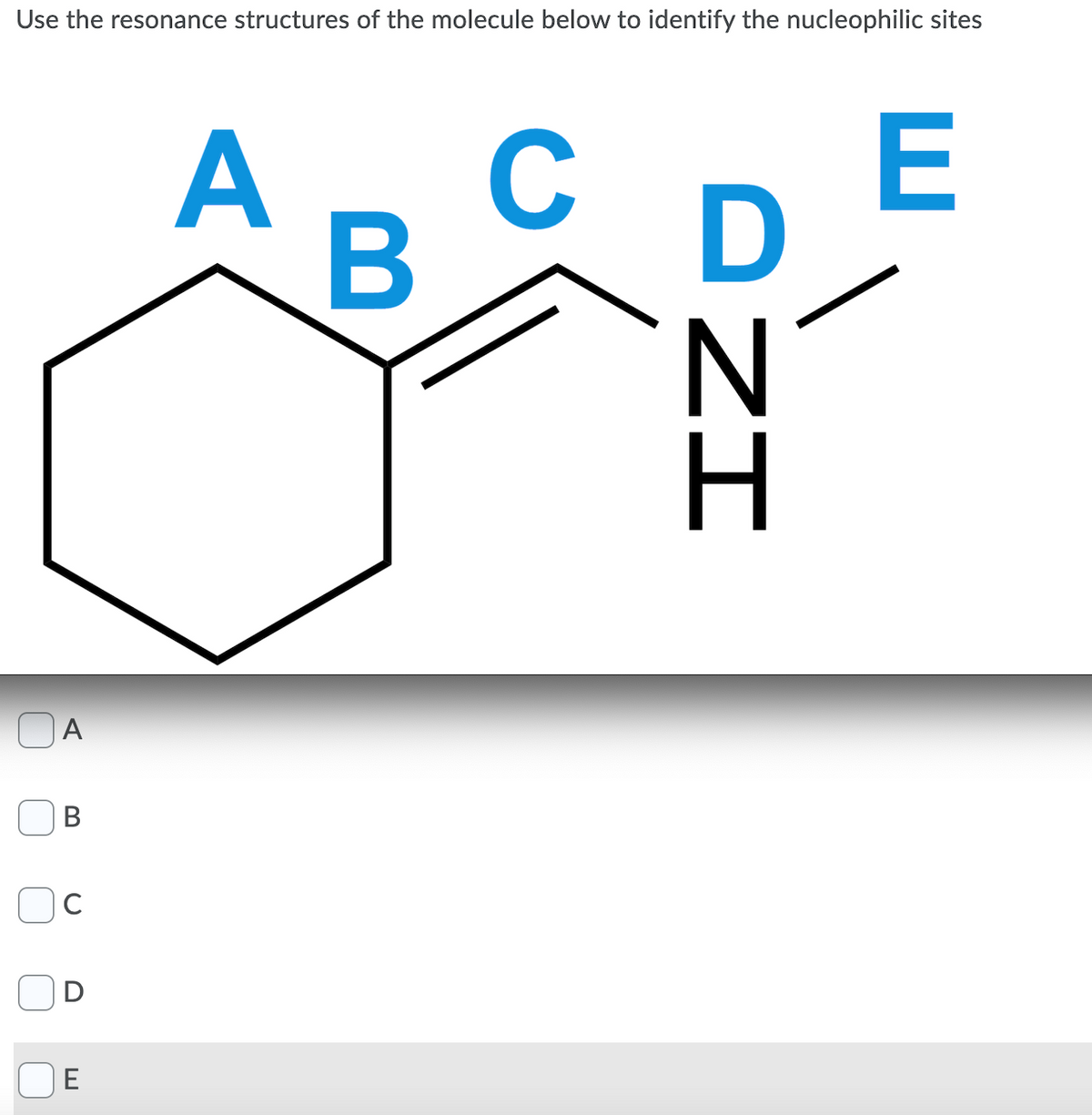
for the first pic u can choose more than one site
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Plastic photochromic sunglasses are based on the following reversible rearrangement of a dye inside the lenses that occurs when the lenses are exposed to sunlight. The original dye absorbs UV light but not visible light and is thus colorless, while the rearrangement product absorbs visible light and is thus darkened. (a) Show the mechanism of the rearrangement. (b) Why does the rearrangement product absorb at a longer wavelength (visible light) than the original dye (UV)?please explain how to get thrree answers to thr following questions thanks. Need all threeDraw the mechanism fot his reaction. B = [IrCl(CO)(PPH3)2]
- Identify the major product of this reaction: (ATTACHED HI) Arrange the ff. in order of DECREASING acid strength: 1ST STRONGEST, 2ND, 3RD WEAKESTPls ignore the highlighted part __________ is reduced in thefollowing reaction: Cr2O72- + 6S2O32-+ 14H+→ 2Cr3+ + 3S4O62- + 7H2O! ( plz ans with proper explanation , aslo give mechanism
- Identify the Nucleophilic and Electrophilic centers in the following molecules.Hi! Are these nucleophiles and electrophiles that I listed int hte image correct? Thanks, Shreya1.Draw the structures of all 3 resonance hybrid forms of the benzenonium intermediatefor both o and p attack for the case of activating groups an indicate which one in eachcase is the most stable form.Draw the structure of all 3 resonance hybrid forms of the benzenonium intermediatefor m attack in the case involving a deactivating group. 2.Halogens are deactivating groups but direct electrophilic attack at the o and ppositions. Why is this the case?
- What carbon radical is formed by homolysis of the C–Ha bond inpropylbenzene? Draw all reasonable resonance structures for thisradical.Draw the neutral organic starting material. The Hint: The reaction conditions support electrophilic addition of Br2 to an alkene's C=C double bond, which would normally yield a dibromo product. However, the product has only one bromine atom, with a C–O bond on the adjacent carbon. This fragment is diagnostic for a halohydrin, where an oxygen nucleophile (water or alcohol) reacts with a bromonium intermediate to generate the O–CH2CH2–Br motif. Work backwards to determine what the starting material must look like. There should be an alcohol and an alkene in the neutral organic starting material.From each pair, select the stronger nucleophile. Q.) CH3COO- or OH-



