Q: CH; CH;-0-C-H CH, NH,-C=0 CH, 0=C- CH, CH, ОН
A: Functional groups are groups that gives special characteristics to compounds.
Q: a. CH3 CH3 Br H-Br -H H. H - b. SOCI, O,N O2N- + HCI + SO2 OH C. H,O H2C=CH2 + KMNO̟ 0=C=0 + H-OH d.…
A: There are different type Chemical Reactions in organic chemistry. Some of them are addition…
Q: A. Classification of Hydrocarbons. H H 6. H-C=C-Ċ-Ċ–H H H 2. Н-С—С—Н H. Η Η Η Η 3. H-Ċ=ċ=ċ=ċ-H 8. H…
A:
Q: + 2H2 H20 > B Na H3C-C: EC-CH3 + 1H2 A + Cl2 NH3(1) A H20 → B H2SO4 -c=c-CH3 + H2 Lindlar H,O H3C-C…
A: Hydrogenation is process of adding hydrogen across the unsaturated bond
Q: Name the following compounds using R,S designations:
A: The priority to the substituents attached to stereogenic centers is given by following these rules:…
Q: -CH, IUPAC C. -CH2-CH; Common H;C-CH,-CH-CH,-C-H d. CI IUPAC
A: The IUPAC is a system that helps in the naming of organic compounds in a systematic manner.The…
Q: a. H3C-CH2-NH-C-H IUPAC H3C-CH2-NH-ĉ-H Common H3C-CH-CH2-CH2-CH2-C-0-CH3 b. Br IUPAC…
A: (a) IUPAC name : N-Ethylmethanamide Common name : N-Ethyl formamide (b) IUPAC name :…
Q: 1. CH:-CH C-OH OH 2. CH:- CH-CH2-C-C CH: CH3 -C-CH2-CH2-C-OH CH: CI CH3-CH2-C CH2-C-ÖH ČI C-OH
A: IUPAC is a naming system by which we give a particular name to a compound. And this is stand fòr…
Q: Draw curved arrows to show the movement of the electrons that result in formation of the given…
A: Nucleophiles are the electron rich species and it can share at least a pair of electron.…
Q: How are the Fischer projection formulas in each pair related to each other? Are they identical or…
A: Enantiomers are those organic compounds which are non superimposable mirror images of each other.…
Q: H3C CH,OH H,SO, CH3 H3C- H3C- H3C- H3C- OH H3C- OH H;C- OH OH 0-CH3 H3C-O -CH3 H3C-O -CH3 H3C-O A C
A:
Q: What is the major product of each of the following reactions?
A: The complete chemical equation for (a) is shown below.
Q: C. Skeletal and Name: * ** H Н-с-н HHH H H H а. Н-с-с-с-с-с-с-с-с-с-н H HH H HH H H H…
A: 1. Select the principle carbon chain 2. Numbering 3. Naming (prefix + word root + suffix)
Q: -Which of the following structures represent the same substance: a- CH,-CH-CH-CH,-CH, CH, OH CH, b-…
A:
Q: CH3-SH A 6 CH3 -CH=CH -CH2-CH3 OH C C CH3 -CH -CH3 レ C3-CHz -CH E CH3 -CH2-0-CH2では
A:
Q: 0. CH, - NH - C - CH, - CH, - CH, CH, CH, - CH, - C - N-CH, - CH, CH, -C - NH, II CH, CH, - CH, - NH…
A:
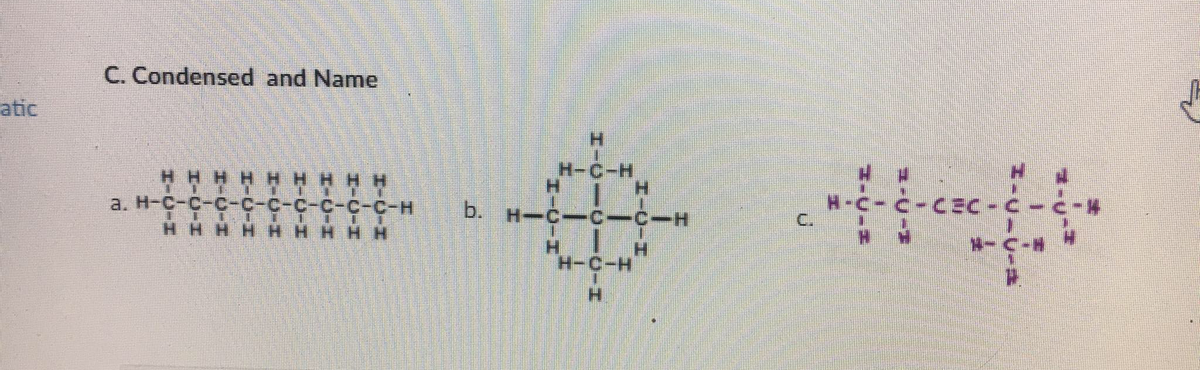
Q: B. Condense and Name: HH H H H b. H-C-- CH,CH,CHCH,CH,CH, CH,CH,CHCH, a. C. ČH, ČH,CH, HH H H H
A:
Q: CH3 OH CI-Br + H20 CH3 OH CI-Br + H20 CH3 Br OH + C-Br + H20 CH, CH-Br + H20 но Br CH3 CI OH CI-Br +…
A: The retrosynthesis is the way of breaking up the molecule in mind in such a way so that it can give…
Q: -CH3 -CH3 H3C- -Br H3C- Br -CI -CI IUPAC.
A: The stereochemistry of a compound can be assign by using Cahn-Ingold Prelog priority rules. the…
Q: Name the following:
A: Since the question has multiple sub-parts, we are entitled to answer only the first 3 parts.
Q: CH3 CH3 CH3 CH3-C CH2 CH2-OH CH3- -C-CH =CH-CH3 CH3-C-CH2-0-CH3 II III I CH3 CH3 CH3
A: The compounds given are,
Q: OH CH,CHCH,CC=CH CH, CH3 Çi C1 CH 3-CH-CH 2-CH-C-CH-CH 2-CH2-OH tH-CH, C1 CH3-CH,-CH;-CH2-NH,
A:
Q: b. CH3 CH2 -N-CH,-C- || -CH,-C-N-CH-C- -N-CH-C-N- H OH OH с. но OH но- OH 0-CH;CH2-0 0-CH;CH-O
A: Following monomers condensed to form the given polymers with removal of water molecule .
Q: k H3C-CH2-CH2-C-CH2-ċ-CH3 IUPAC 1. OH IUPAC OH Common Name OH H3C-CH2-CH-CH2-C-H m. IUPAC OH…
A: Applying concept of IUPAC nomenclature of organic molecules we can determine IUPAC nomenclature of…
Q: C-OH Br CH3-CH2-CH2-0-c-CH2-CH,-CH,-CH d. O =J
A: Note : As per our company guidelines we are supposed to answer only first 3 sub-parts. Kindly repost…
Q: 1) CH3-C-0 O 2) PHÁCH 13--0-C-CH3 3) H₂O 4) -H₂0 5) H O O O a) Ph-CH=CH₂ + CH3-C-OH b) Ph-CH=CH-C-OH…
A:
Q: it-C-H HH # H H-C-C-C-C=C - C -C-H CH3 - CH- CH C-OH H H H-C-H H-C-H CH3 H-C-H O=U HICIH
A: Answer:- This question is answered by using the simple concept of writing the name of the organic…
Q: Name HI CH3 H-C-C-C- H CH3 Name H-C H HIC 2 H1CIN H C H 3 4 C- | | CH3 H HICIN HIC C₂H5 H 51 H -C-H…
A: According to IUPAC nomenclature firstly select the main chain and then branched chain. and the main…
Q: нонн онно С. Н.N-C-с-N-C-с-N—с-с-он CH;CH¿CHCH3 CH3 C,H;CH2 нонн онно T |LI I |LI I ||…
A: Q. C In the peptide chain, the 1st amino acid is isoleucine 2nd amino acid is alanine 3rd one is…
Q: :0: CH3- CH2- CH2- ÖH CH3-CH2-C-H
A: Compound A act as a nucleophile because oxygen have lone pair of electrons that can donates electron…
Q: Ph CH,-C=C-CH–CH, а. H.C CH3 C=C b. H c=c-CH,CH, С. (CH3);C-C=C-CH(CH;)CH,CH;
A: Alkynes is the class of hydrocarbons which contains a carbon-carbon triple bond and it is an…
Q: H,C- -CH- -CH3 1 CH3 2 H;C- CH-CH ČH,-CH; H,C-CH,-CH,-ÇH-CH;-CH;-CH; 3 ČH;-CH3
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Ph CH,-C=C-CH–CH, а. H;C CH3 C=C b. H c=c-CH,CH; С. (CH3);C-C=C-CH(CH;)CH,CH3
A: a) The IUPAC name of the given compound is, 4-phenylpent-2-yne
Q: CH3-o CH2-CH2-CH2.-Ct3 CH8 -CH-CH20-CH2-C H3 CH3-C Hz-0-CHz-CH 2 -Cto CH3-0-CH - CH 2-CH3 (F 0-CH3…
A:
Q: d. C,H-C-CH, H* e. C,Hs-C-CH f. C,Hs-C-CH; NaOH g. CH;-C=C-CH, HgSO., 4 h. C.Hs-C-C.H,
A:
Q: O CH3 CH3 CH,0° CH,OH CH3 HCI (а) (b) CH;OH CH3 H3C H;C 8 CH3 CH;0° CH,OH CH3 HCI (d) CH,OH
A: SOLUTION: Step 1: Hello. Since the question contain multiple sub-parts, the first three are solved.…
Q: CH;–CB12-C=C-CH3 d. CH, CH;-C=C-C-OH е. CH,CH, f. -CH,-C=C–CH3
A: (d) According to IUPAC nomenclature the name of the following compound is: 4,4-dibromopent-2-yne.
Q: CH3 H3C CH3 CH3 CH3 H- -OH H2N- g. h. H3C Br H Br i. NH2
A:
Q: a. CH;(CH2)4I, CH3(CH2)5I, CH3(CH2)çI b. CH3CH¿CH;NH2, (CH3);N, CH3CH¿CH,CH3 c. (CH3);COC(CH)a,…
A: Boiling point of a chemical compound is the temperature at which vapor pressure of a compound…
Q: H H H---ċ-H CH3-C-H ОН ОН CH;- CH- CH,- CH3 CH2 CH, CH3 CH3 C=C H H. CH,-0-CH,CH,
A:
Q: CH3 H2 H3C-C-c-C- он H2 -C-CH3 اجابتك OH H3C он اجابتك
A: The IUPAC name of the given compounds has to be determined. a) b)
Q: H-C-CH2-CH3 H3C- C-O-CH2-CH3 H3C-CH2-OH H3C-NH-CH2-CH3 H3C- -CH2-CH3 H3C-C-H Br H3C-CH2-CH2-C-OH…
A: The functional group is the specific moieties in the molecule that are responsible for all the…
Q: Br CH3 Common Name а. OH NO 2 Br b. Common Name c. H3C-CH-CH-CH2-C-NH-CH2-CH3 IUPAC_
A: Since you have asked a question with multiple sub-parts, we will solve first three sub-parts for…
Q: : ОН :0:0 (а) CH3-CH2-CH2 CH3-CH2-CH2 I or II :0:0 :0:0 (b) CH3 CH2-CH2 CH3 CH=CH or II CH,O : NHO…
A: The bases given are,
Q: CH3 1) NaOCH, ..CH3 2) CH;CH,OH H3C H3C H3C CH3 CH3 H3C-O H3C-O CH3 CH3 H3C H3C H3C H3C OH H3C H3C…
A: A product is formed
Q: H2N-CHO-OH CH2 H2N-CHC-OH CH2 CH2 H2N-CHC-OH H. C=0 NH2 CHa
A: Given: Different- different structure of amino acids. To find: Common name of amino acids. Solution:…
Q: H. OH CH3 1. H3C H3C CH3 and H. OH H3C. CH3 2. Hзс CH3 HO and H. H3C. CH3 CH3 HO HO. H. and 3.
A: As there occurs change in the stereocenter of the optically active structures, despite of the change…
Q: 3 ÇH3 CH3 CH3 CH3 CH,-c -H CH,-C-OH; CH;-C +:OH2 ČH; CH3-C CH;=C ČH3 ČH; CH; 2 CH3 CH;-C-CHCH, Hi…
A: In 2, there is no loss of H+, number of hydrogen in reactant and product are same.
Q: H2N-CHC-OH CH2 H2N-CHC-OH CH2 H2N-CHC-OH CH2 C=0 N. OH NH
A: Amino acids contain carboxylic acid and an amine functional group on the same carbon atom. Amino…
Step by step
Solved in 2 steps with 2 images

- Construct models of the following generalized formula: a. Model 1: CH4 b. Model 2: CH3Cl c. Model 3: CH2ClBr d. Model 4: CHClBrFsample weight: KOH: 1g KOtBu 1g 1-propanol: 1g 1-butanol 0.742g calculate the thoeretical and percent yields of methylbutenesPr0vide the c0rrect IUPAC name 0f the p0lyfuncti0nal c0mp0unds
- For mono-substituted cycloalkanes the “1” is not included. That is, the name1-methylcyclohexane is not correct. The correct name is simply “methylcyclohexane”. a. Draw methylcyclohexane. b. Explain why adding a “1” to methylcyclohexanedoes not add any new information.HOCI HF HCN H₂SO4 HOBr 2.29 x 10-8 O 2.30 K₂= 3.510-8 O 1.17 x 108 O 1.45 x 10-7 K₂ = 7.2 10-4 K₂ = 4.010-10 K₁ = very large K₂ = 1.2 10-2 Ka = 2.5210-⁹ (COOH)2 CH3COOH C6H5NH2 NH3 K₁ = 5.9 x 10-² K₂ = 6.4 x 10-5 K₂ = 1.8 x 10-5 Refer to Equilibrium Constants. What is the [H3O+] of a solution that is 0.0100 M in HOCland 0.0300 M in NaOCI? Kb = 4.2 x 10-10 Kb = 1.8 x 10-5Bottled straight-chain alkanes can be used as fuels, including pentane and hexane . pentane hexane ΔΗ°comb (kJ/mol) -3535.0 -4163.0 Liquid Denisty (g/mL) 0.684 0.655
- Scoring: Your score will be based on the number of correct matches minus the number of incorrect matches. There is no penalty for missing matches. Match the condensed structural formula on the left with the molecular structure on the right. Clear All CH3CONH2 C6H5COOH CH3CH2CH2OH CH3COOCH2CH3 C6H5CH2NH2! ( plz answer question 2 with detail explation , give name of both )Make 2 isomers out of C4H9O, Include a drawing and the names

