Q: Propose a structure for Compound C (molecular formula C10H120) consistent with the following data,…
A: The NMR spectrum provides the following details about the structure of the molecules: The number of…
Q: Identify each compound below from its molecular formula and its 13C NMR spectrum.
A: The 13C NMR always helps in recognizing carbon atoms that exist in given organic molecule. Each…
Q: 7 4 3 3.99 2.00 3.01 1.99
A: The answer oh the following question is given below:
Q: Based on 13C-NMR data, what compound corresponds to the following spectrum? A A B с с D D E E B
A: In 13 c nmr spectra number of protons are equal to number of chemically non-equivalent carbons
Q: Which structure drawn below for the compound with a formula of CgH10 has four chemically unique…
A: In 1H NMR, chemically unique proton shows different peak while chemically equivalent proton give…
Q: Give the structure that corresponds to the following molecular formula and ¹H NMR spectrum::…
A: Secondary hydrogens are represented by the chemical shift (1.93); tertiary hydrogen carbon connected…
Q: Identify the following compounds from its molecular formula and its IR and 1H NMR spectra:
A: IR spectroscopy- is the vibrational motions of the atoms that are important. This theory utilizes…
Q: Identify each compound from its molecular formula and its 1H NMR spectrum: C9H12
A: To see the presence of double bond we calculate the degree of unsaturation for the given compound…
Q: Identify the compound with molecular formula C6H10O that gives the following DEPT 13C NMR spectrum:
A: The molecular formula of the compound is C6H10O: Calculation of Double Bond Equivalent of the given…
Q: Use the 1H NMR and IR data to determine the structure of each compound.
A: Compound A: The molecular formula of the Compound A is C5H10O. Information from IR spectrum: The IR…
Q: Provide a structure for the given compound. C,H1,O2; IR: 1743 cm-l; 'H NMR spectrum chemical shift,…
A: Splitting pattern in 1H NMR is assigned according to n+1 rule where n represent the number of…
Q: Identify compound below from its molecular formula and its 13C NMR spectrum.
A: Calculation of double bond equivalent:
Q: Deduce the structure given the 1H-NMR and IR spectrum. Formula is C5H1002. Name structure if…
A:
Q: Which compound is responsible for the 'H-NMR spectrum below? 3H 6H 一 2H 2H 1H 8 7 4 PPM 2 1 O a. O…
A: 1H NMR spectrum
Q: Identify compound below from its molecular formula and its 13C NMR spectrum.
A:
Q: 5. How might the two trimethylcyclohexane isomers shown below be most readily distinguished using…
A:
Q: How could you distinguish the 1H NMR spectra of the following compounds?
A: Spectroscopy is a tool to identify the unknown compounds in the given solution. Different…
Q: A compound with a molecular formula C9H12 has the ¹H NMR spectrum below. Which structures given is…
A:
Q: A bottle is labeled only "Compound B (C,H,,)" and contains a colorless liquid. You have been called…
A: Given, Chemical formula = C6H12 Peaks = 888 cm-1, 1646 cm-1, 3080 cm-1. Coupling constant, J = 6.9…
Q: Give the structure that corresponds to the following molecular formula and 'H NMR spectrum: C,H,F,I:…
A: The analysis of the results obtained from proton NMR, 13C-NMR, and mass spectra such as HRMS and…
Q: NH2 n the 13C NMR spectrum of compound 1? 2.
A: In the 13CNMR spectroscopy, you will see the spectrum of chemical different carbon that means…
Q: Which of the following compounds is responsible for the 1H NMR spectrum shown below?
A: Given spectrum shows 2 signals. One at 2.3ppm and another at 7.1ppm. The peak at 7.1ppm is the…
Q: Identify compound from its molecular formula and its 1H NMR spectrum:
A: First, calculate double bond equivalent of C9H12. It is given as: DBE = C + 1 - H2 - X2 + N2= 9 + 1…
Q: The 1H-NMR spectrum of an unknown compound shows two signals, each of which is a singlet. Explaining…
A: NMR or nuclear magnetic resonance spectroscopy is a technique used to determine a compound’s unique…
Q: CH3 H3C `CH2 CH2
A: Concept: The number of NMR signals represents the number of different types of protons in a…
Q: Compound P has molecular formula C5H9ClO2. Deduce the structure of Pfrom its 1H and 13C NMR spectra.
A: Calculation of double bond equivalence: DBE = C +1 – (H + X – N) / 2 Here, C is the number of carbon…
Q: Which structure below best matches the following H NMR spectral data? 61.2 (triplet, 3H), 20…
A:
Q: Give the structure that corresponds to the following molecular formula and 'H NMR spectrum: C,H,F,I:…
A: The structure of the compound having molecular formula C2H2F3I is given below
Q: An organic compound with molecular formula C4H8O2 has the following nmr values. Propose the…
A:
Q: Identify compound from its molecular formula and its 1H NMR spectrum:
A: First, calculate the double bond equivalent as shown as follows: DBE = C + 1 - H2 - X 2 + N2= 9 + 1…
Q: The 1H NMR spectra of two compounds, each with molecular formula C11H16, are shown here. Identify…
A: (a)
Q: Which of the structures below better matches the provided 13C-NMR spectrum? 200 180 160 140 120 20…
A: The NMR spectroscopy is an important tool for the determination of the structure of an organic…
Q: Identify each compound from its molecular formula and its 1H NMR spectrum: C9H12
A: To see the presence of double bond we calculate the degree of unsaturation for the given compound…
Q: CH,O OCH, CH,O осн II IV Oa V Ob." O.!
A: 1-H NMR spectroscopy, also known as proton nuclear magnetic spectroscopy, is a technique used to…
Q: erh/ H Spectrum Expanded peak cluster at 232 ppm Expanded peak cluster at 4.13 ppm Expanded peak…
A:
Q: Predict the 13C NMR spectra of the 2-chloro-2-methylpropan-1-ol. 1 CH3 2 13 HO-CH2 CI 4CH3
A: The analysis of 13C NMR spectra of the above compound is given below
Q: Which structure of molecular formula, C7H140, fits the proton NMR spectrum shown below? doublet…
A: Given, The molecular formula of the compound C7H14O, the structure of the compound based on spectra…
Q: How many negative signals would be observed in the DEPT135 13C NMR spectrum for the molecule…
A: DEPT135 gives negative signals for CH2 carbons
Q: Identify compound below from its molecular formula and its 13C NMR spectrum.
A: 13C NMR spectroscopy studies the carbon-13 nucleus in the compound, and it provides the information…
Q: The 1H-NMR spectrum of Compound C shows five signals – δ 2.38 (1H, dt), 2.72 (1H, dt), 5.34 (1H, t),…
A: To identify the structure of the compound using NMR and mass spectra data.
Q: Identify the following compounds from its molecular formula and its IR and 1H NMR spectra:
A: On the basis of all interpretation structure of compound is
Q: For the following compound, predict the number of signals in a 13C NMR spectrum: ∞ OA5 OB. 6 O C7 O…
A:
Q: CgHgN solvent proton decoupled 200 160 120 80 40 O 8 (ppm) 'H NMR Spectrum (400 MHz. CDCI, soluton)…
A: From the given molecular formula, the degree of unsaturation is; DoU=5-82+1=2 From the DoU…
Q: How many carbon signals are expected in the proton-decoupled 13C NMR spectrum of the compound shown…
A:
Q: Provide the correct structure for the compound with the following 1H NMR spectrum and molecular…
A: 1H NMR spectroscopy is used to determine the structure of compound with respect to hydrogen nuclei…
Q: How would integration distinguish the 1H NMR spectra of the following compounds?
A: 1H NMR- It is application of NMR spectroscopy called as proton Nuclear Magnetic resonance…
Q: Br NaOH Molecule A H20 130 140 130 120 110 100 90 80 70 60 50 40 30 Option 1 Option 2 OH Br HO O…
A: Every nuclei will have spin and if an external magnetic field is applied, there will be an energy…
Q: Which of the hydrogens gives the 1H NMR signal with the highest frequency? F-CH2CH2CH2CH2CH2-Br 1 2…
A:
Q: 6 Name and draw the compound using the C-NMR spectra MS: M+=152 13C-NMR 160 139…
A:
Q: Identify each compound from its molecular formula and its 1H NMR spectrum: C9H12
A: The double bond equivalent for the compound is equal to 1. This suggests the presence of carbonyl…
Identify compound below from its molecular formula and its 13C NMR spectrum.
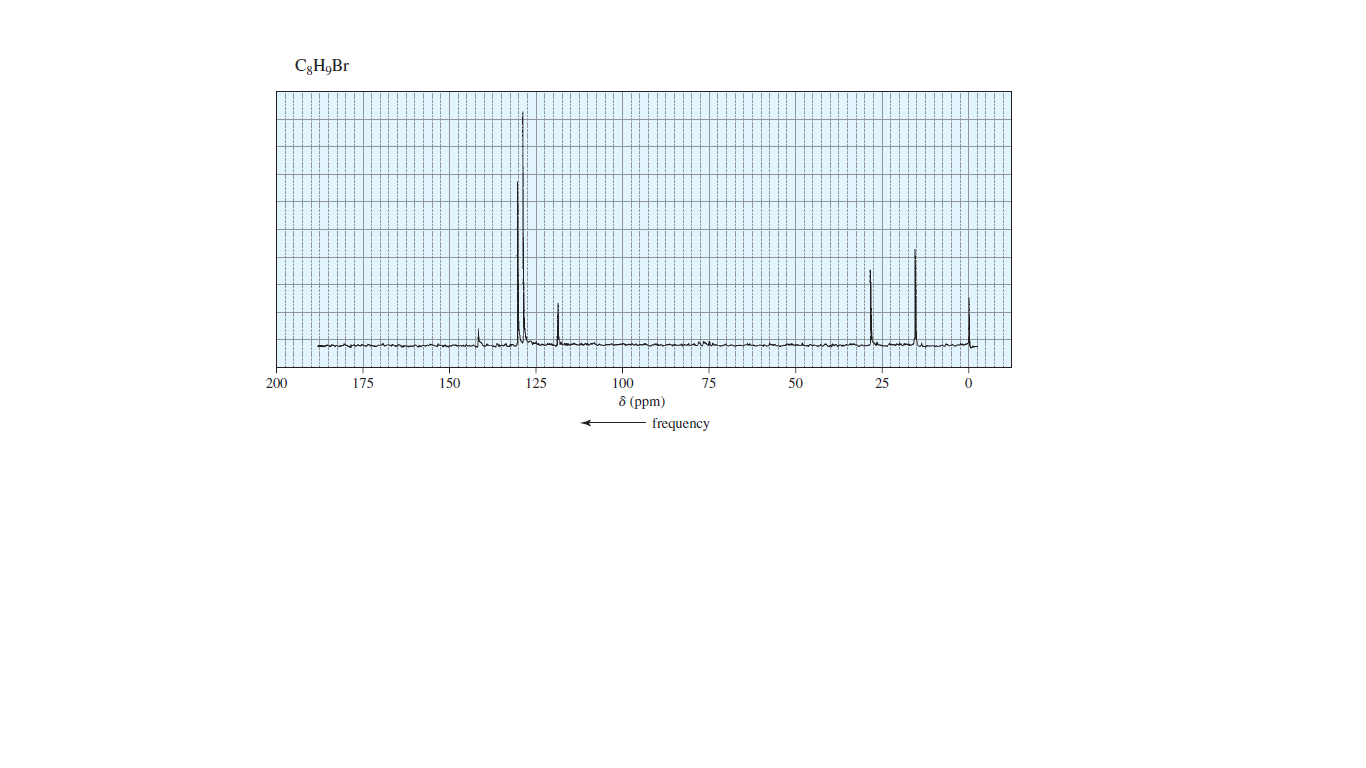
.jpg)
Step by step
Solved in 2 steps with 2 images

- Please answer subparts vii, viii, and ixPlease create a caption for this table. Solution NaCl Conc. (%) Osmolality (mOsm) % transmittance Absorbance % hemolysis % crenation C distilled 0 0 0.001029 4.987584625 100 0.03354 1 0.177179111 54.61 0.001551 4.809388202 96.42720001 0.05837 2 0.297126222 91.58 0.01012 3.994819487 80.09527231 0.08444 3 0.442542222 136.4 3.849 1.414652089 28.3634704 0.134 4 0.590164444 181.9 64.8 0.188424994 3.777880643 0.2125 5 0.74752 230.4 95.64 0.019360433 0.388172513 0.3368 6 0.89644 276.3 99.56 0.001915112 0.038397585 0.5336 7 1.095648889 337.7 99.98 8.68676E-05 0.001741676 0.9834 8 1.336711111 412 100 0 0 2.1 9 1.755568889 541.1 100 0 0 7.9 10 2.674395556 824.3 100 0 0 57.83 11 4.490211111 1384 100 0 0 99.72Please round to 3 sigfigs
- Why does fluorescence tend to be the mirror image of absorption?Calculate the following exercise with correct sig figs: ((2.9365mL - 3.00mL)/3.00mL)x100%Choose all correct answers for the following.Percent Transmittance is defined as: Question 6 options: T = Po/P %T = (P/Po) x 100% T = P/Po %T = (Po/P) x 100%
- Show that: Cp-Cv= -T(dv/dT)^2 (dp/dv)Given the following frequency table of data, what is the potential outlier? Value Frequency 8 1 9 0 10 0 11 0 12 0 13 0 14 0 15 0 16 1 17 4 18 10 19 4 20 6 21 2 22 3 Select the correct answer below: 8 18 16 20 22How would a 95% Z-interval compare to a 95% T-interval?

