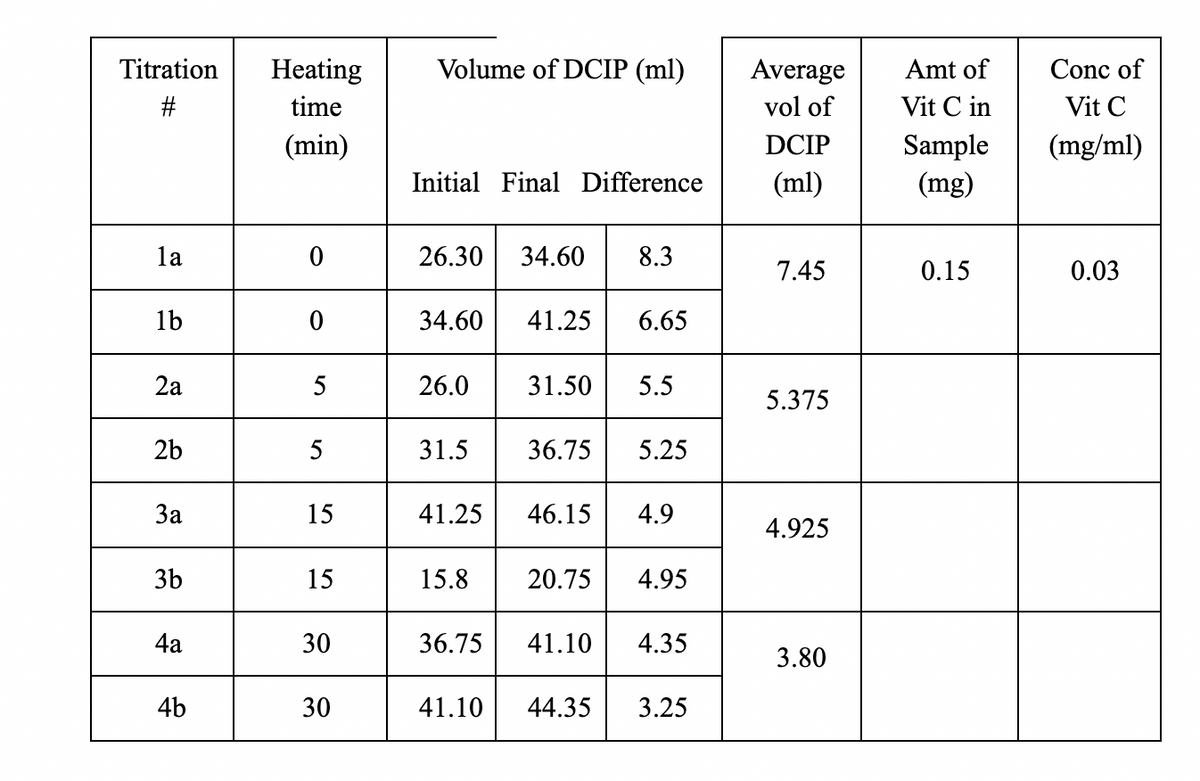
Calculate amount of vitamin c in sample (mg) and concentration of vitamin c (mg/ml). Given: DCIP (0.1mg/ml water), Molar mass of DCIP= 268.1 g/mol Molar mass of ascorbic acid (vitamin c)= 176.12 g/mol
Calculate amount of vitamin c in sample (mg) and concentration of vitamin c (mg/ml). Given: DCIP (0.1mg/ml water), Molar mass of DCIP= 268.1 g/mol Molar mass of ascorbic acid (vitamin c)= 176.12 g/mol
Chapter31: Introduction To Analytical Separations
Section: Chapter Questions
Problem 31.25QAP
Related questions
Question
Calculate amount of vitamin c in sample (mg) and concentration of vitamin c (mg/ml).
Given: DCIP (0.1mg/ml water),
Molar mass of DCIP= 268.1 g/mol
Molar mass of ascorbic acid (vitamin c)= 176.12 g/mol
Transcribed Image Text:Titration
Heating
Volume of DCIP (ml)
Average
Amt of
Conc of
#
time
vol of
Vit C in
Vit C
DCIP
Sample
(mg)
(min)
(mg/ml)
Initial Final Difference
(ml)
la
26.30
34.60
8.3
7.45
0.15
0.03
1b
34.60
41.25
6.65
2a
5
26.0
31.50
5.5
5.375
2b
5
31.5
36.75
5.25
За
15
41.25
46.15
4.9
4.925
3b
15
15.8
20.75
4.95
4a
30
36.75
41.10
4.35
3.80
4b
30
41.10
44.35
3.25
Expert Solution
Step 1
Each cm3 of 1% DCIP solution equvalent to 6.071× 10^-4 g ascorbic Acid.
268.1gm DCIP=1Mol
0.1/1000=0.0001gm=(1/268.1)×0.0001mol =3.73×10-7M
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning



Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning