Calculate M, M, and the polydispersity index for a hypothetical polymer sample that contains equimo- lar amounts of polymer having molecular weights of 30,000, 60,000, and 90,000.
Calculate M, M, and the polydispersity index for a hypothetical polymer sample that contains equimo- lar amounts of polymer having molecular weights of 30,000, 60,000, and 90,000.
Chapter84: Fractional Distillation, Azeotropes
Section: Chapter Questions
Problem 10P
Related questions
Question
Please help me with the question below. A detailed explanation to aid in understanding is welcome.
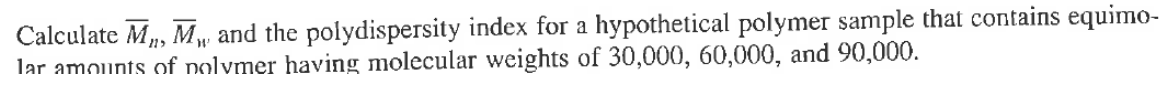
Transcribed Image Text:Calculate M, M, and the polydispersity index for a hypothetical polymer sample that contains equimo-
lar amounts of polymer having molecular weights of 30,000, 60,000, and 90,000.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Recommended textbooks for you

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:
9781305446021
Author:
Lampman
Publisher:
CENGAGE LEARNING - CONSIGNMENT


Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning