The process of liquid water forming from water vapor is an: Endothermic process, because energy is released from the system due to breaking of interactions between water molecules. O Exothermic process, because energy is released from the system due to formation of interactions between water molecules. O Endothermic process, because energy is absorbed by the system due to breaking of interactions between water molecules. O Exothermic process, because energy is absorbed by the system due to formation of interactions between water molecules. MacBook Air DD DA O00 80 F7 F9 F8 F5 F6 F3 F4 * & 23 3 4 6. 7 8
The process of liquid water forming from water vapor is an: Endothermic process, because energy is released from the system due to breaking of interactions between water molecules. O Exothermic process, because energy is released from the system due to formation of interactions between water molecules. O Endothermic process, because energy is absorbed by the system due to breaking of interactions between water molecules. O Exothermic process, because energy is absorbed by the system due to formation of interactions between water molecules. MacBook Air DD DA O00 80 F7 F9 F8 F5 F6 F3 F4 * & 23 3 4 6. 7 8
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter15: Gases,liquids, And Solids
Section: Chapter Questions
Problem 85E
Related questions
Question
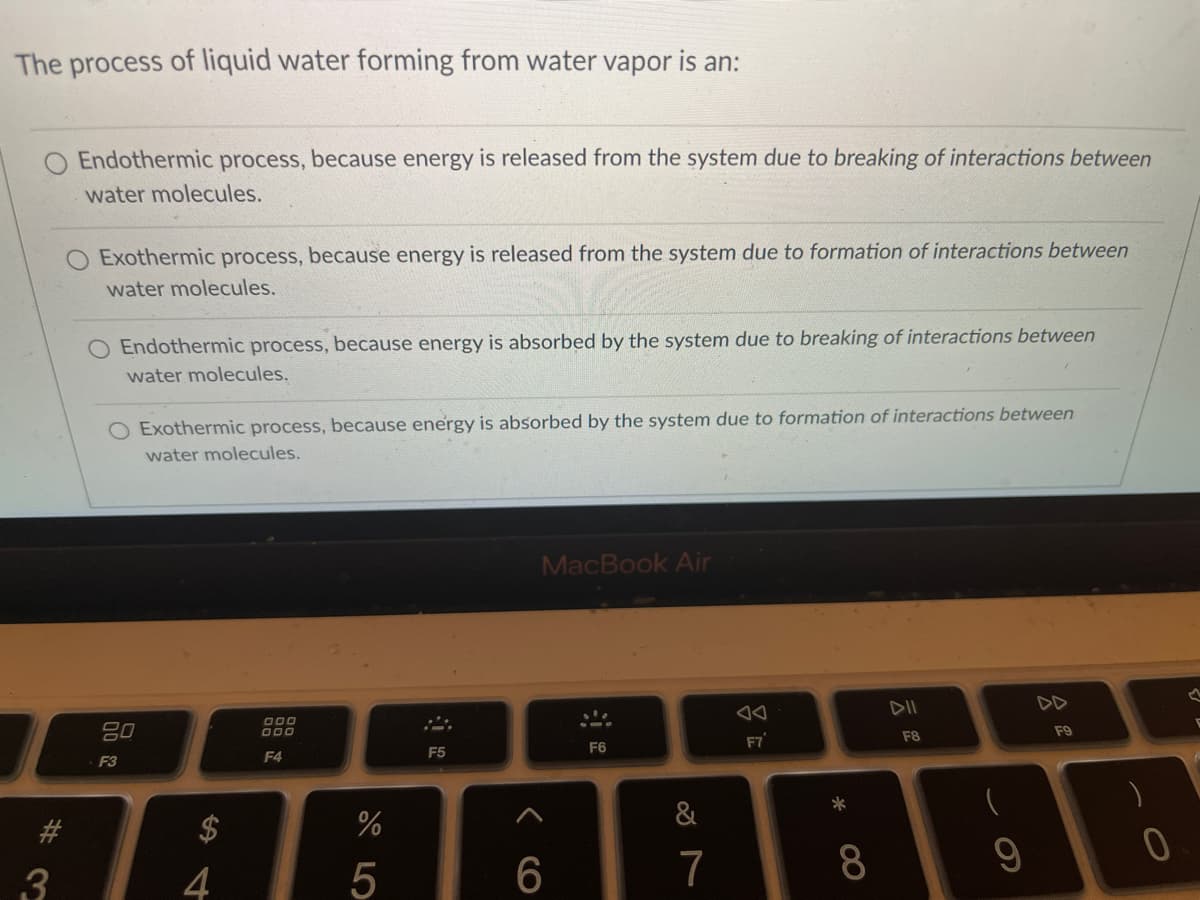
Transcribed Image Text:The process of liquid water forming from water vapor is an:
Endothermic process, because energy is released from the system due to breaking of interactions between
water molecules.
O Exothermic process, because energy is released from the system due to formation of interactions between
water molecules.
O Endothermic process, because energy is absorbed by the system due to breaking of interactions between
water molecules.
O Exothermic process, because energy is absorbed by the system due to formation of interactions between
water molecules.
MacBook Air
DII
DD
O00
80
F8
F9
F7
F5
F6
F3
F4
&
#3
3
4
6.
7
8
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning



Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning



Chemistry for Today: General, Organic, and Bioche…
Chemistry
ISBN:
9781305960060
Author:
Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning