Chapter26: Molecular Absorption Spectrometry
Section: Chapter Questions
Problem 26.17QAP
Related questions
Question
Calculate the IHD and explain (elucidate) the structure using the 1H and 13C NMR data.
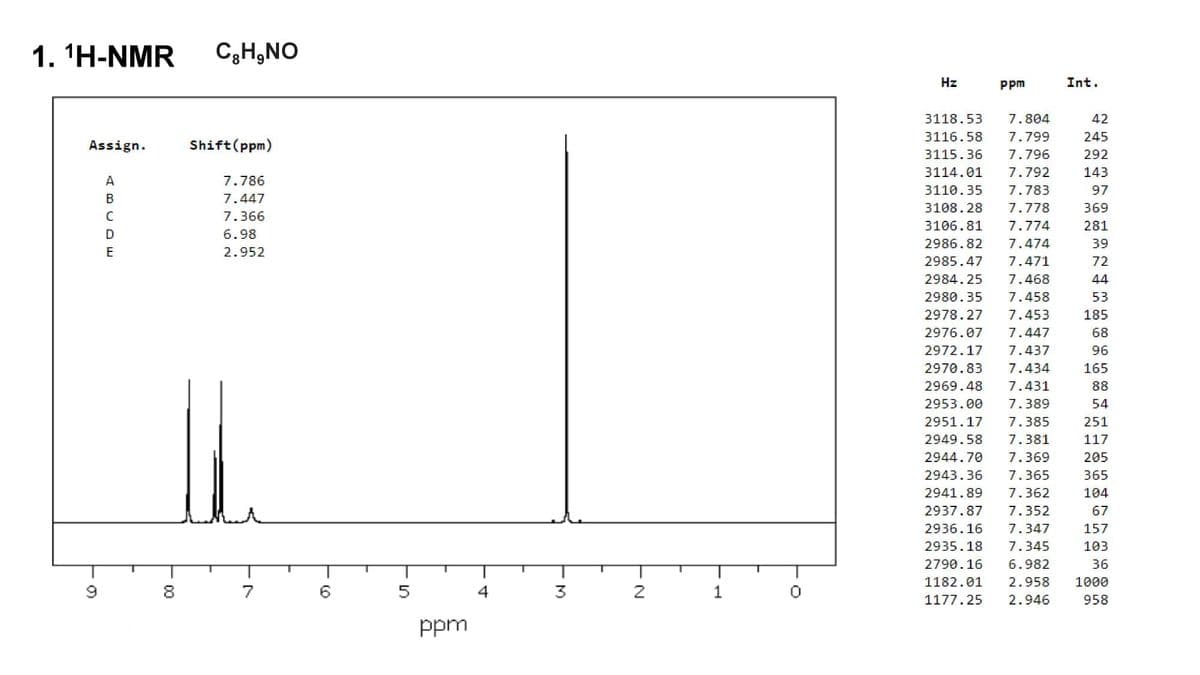
Transcribed Image Text:1. ¹H-NMR C₂H₂NO
Assign.
9
ABCDE
-00
8
Shift (ppm)
7.786
7.447
7.366
6.98
2.952
7
()
5
ppm
- M
3
-N
2
1
Hz
ppm
3118.53 7.804
3116.58 7.799
3115.36 7.796
3114.01 7.792
3110.35 7.783
3108.28 7.778
3106.81 7.774
2986.82 7.474
2985.47 7.471
2984.25 7.468
2980.35 7.458
2978.27 7.453
2976.07 7.447
2972.17 7.437
2970.83 7.434
2969.48 7.431
2953.00 7.389
2951.17 7.385
2949.58 7.381
2944.70 7.369
2943.36 7.365
2941.89 7.362
2937.87 7.352
2936.16 7.347
2935.18 7.345
2790.16 6.982
1182.01 2.958
1177.25 2.946
Int.
42
245
292
143
97
369
281
39
72
44
53
185
68
96
165
88
54
251
117
205
365
104
67
157
103
36
1000
958
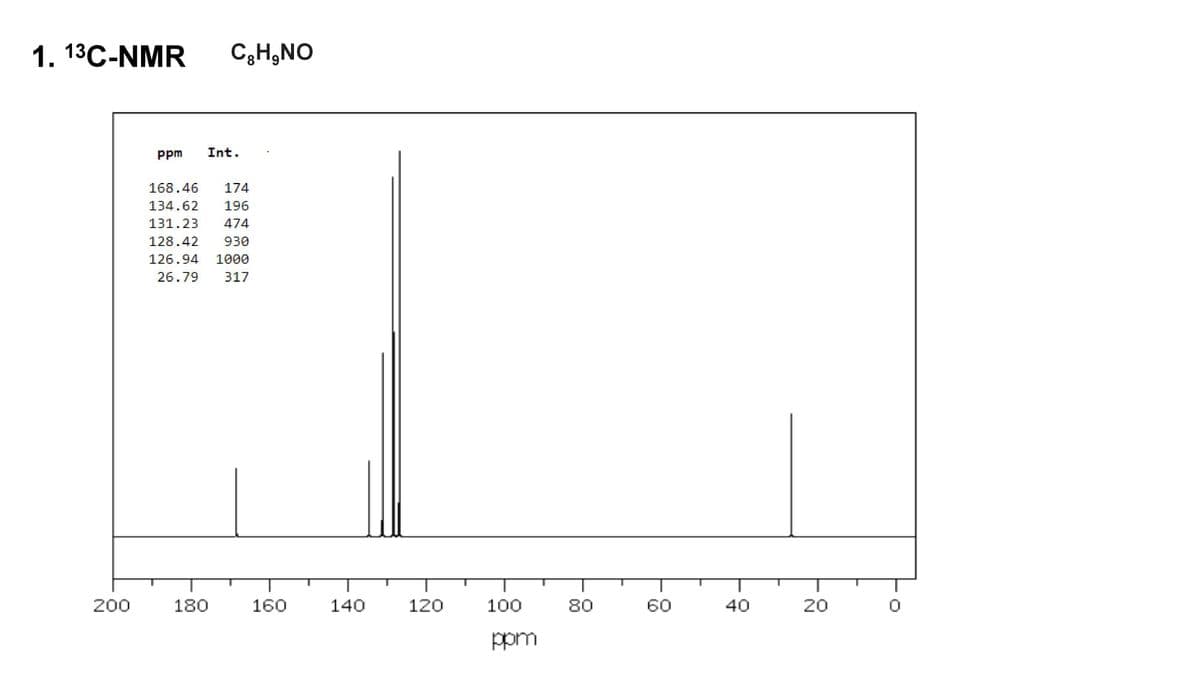
Transcribed Image Text:1. 13C-NMR C₂H₂NO
200
ppm Int.
168.46 174
134.62 196
131.23 474
128.42 930
126.94 1000
26.79 317
180
160
140
120
100
ppm
80
60
40
20
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

