Q: Molecule valence electrons NBRO NFO херод XeTea Formal Charge N: N: Xe: Xe: Electron-Group Geometry…
A:
Q: Identify the molecule or molecules containing polar covalent bonds.
A: Bonds which are formed by sharing of electrons are called covalent bonds .
Q: 2. Draw all products HC- (8 Substitution, 9 elimination) H N CH3 CH3 CH₂OH Δ
A: Substitution will have through the leaving of iodine atom, as iodine (I-) is a good leaving group.…
Q: 15 p) All of the carbon atoms of the products must come from the starting material for this one! ?
A: In this question, we will Synthesized the final product with the help of reagents which are…
Q: 1. If 5.00 g of copper (II) metal reacts with a solution containing 20.0 g of silver nitrate, which…
A: As per bartleyby guidelines i answered only first question so please don't mind and post other…
Q: A patient is prescribed 375 mg of the antibiotic erythromycin. The label on the medicine bottle…
A: The patient is prescribed 375 mg of erythromycin. The antibiotic solution contains 275 mg of…
Q: Predict the correct product of the following reaction. 1) PhMg Br (excess) OCH, 2) H₂0*
A:
Q: Calculate the volume of a 4.56 L sample of gas in a cylinder, if the cylinder is warmed from 50K to…
A: We know accordingly to Charles's law, pressure of a gas is inversely proportional to the temperature…
Q: Draw the dipeptide Gly-Ala. Which new functional groups is formed?
A:
Q: A student is using 10.0 moles of an ideal gas. He measures the temperature as 323K and the pressure…
A:
Q: Labetalol, a β‑blocker antagonist of epinephrine and norepinephrine, is commonly used in treating…
A: Organic chemistry is branch of chemistry in which we deal with compound having carbon and hydrogen…
Q: Calculate the freezing point and boiling point of 10.5 g FeCl3 in 1.50 x10^2 g water, assume…
A:
Q: :O: 하 H H-O Draw the all the products. Add non-bonding electrons where applicable. Select Draw с…
A:
Q: The reducing agent accepts electrons. O True O False
A: Oxidation reduction reaction (Redox reaction) It can be defined as the chemical reaction in which…
Q: Convert 480 Lb into ng
A: This question is based on the conversion of the units. The given units are Lb and ng. Lb means pound…
Q: 5.00 g of lead was heated from 21.0°C to 48.0°C. How much heat was applied to the ead?
A: mass of lead (m) = 5.00 g change in temperature (∆T) = 48.0 oC - 21.0 oC = 27.0 oC Heat applied (q)…
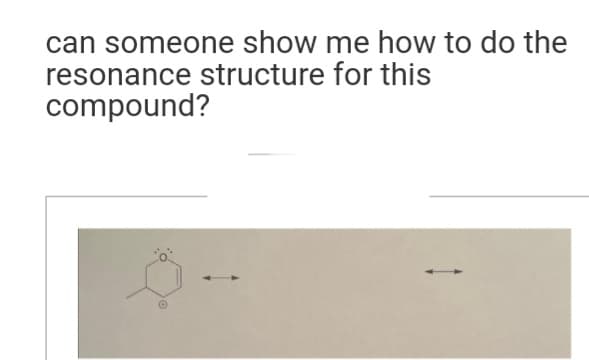
Q: Draw an equivalent resonance structure that minimizes charge. Include all lone pairs in your…
A:
Q: The target diol is synthesized in one step from 1-methylcyclopentene, but your lab partner exhausted…
A:
Q: Q8. Calculate the second ionization energy of He. A) 8.71 x 10-21 J B) 8.71 x 10-15 J C) 8.71 x…
A: 8) Second ionization energy : It is the energy required to remove the outermost electron from a uni…
Q: Part 1: ونكم Base Keq Acid 10-19 view structure Conjugate acid view structure Part 2: Calculate the…
A: This acid base reaction is a combination of two individual reactions. The equilibrium constant of…
Q: Perform calculations to determine the amount of 5.00x10-5 M stock solution needed to prepare 20.00…
A: The dilution of solutions is an important process in chemistry that involves reducing the…
Q: Draw the major product formed when 1,3-cyclohexadiene reacts with this acid. Do not include any…
A: Given reaction is addition reaction . HI added across carbon carbon double bond. This reaction is…
Q: 어 13 H Br D ОН 스
A: The first reaction proceeds via SN2 reaction mechanism with inversion of configuration. The second…
Q: Calculate the freezing point for a 2.50 M CaCl2 aqueous solution assuming ideal behavior. (Kf for…
A: Answer:- Firstly molarity of the solution is converted into molality using the density of the…
Q: If the temperature of a 25.5 mL sample of N₂ gas is increased from 25.0 °C to 75.0 °C as its…
A:
Q: How much heat (in kilojoules) is evolved in converting 1.00 molmol of steam at 155.0 ∘C∘C to ice at…
A: According to the question, The mass of 1.00 mole of steam = 18.00 g of H2O The latent heat of the…
Q: E- PCC D B C SOCI₂ conc HCI conc H₂SO4 A A A HBr 1. 2. OH Na CH₂Cl OH conc H₂SO4 H₂CrO4 PBr3…
A: HBr converts secondary alcohol into the corresponding bromide by an SN2 reaction. Alcohol by…
Q: Draw out the following peptide sequence (Ala- Asn- His- Lys- Glu) from N-terminus to C-terminus.
A:
Q: 80°F When 26.62 moles of FeS₂ react with 5.44 moles of O₂ in this reaction below, how many moles of…
A:
Q: For each equation, solve for each variable in terms of the other variables. For example: z=xy:…
A: Since you have posted multiple questions, we will provide the solution only to the first question as…
Q: 100 μL of 0.01 mol dm 3 sodium fluoride and 10 mL of pH buffer were added to a 100 mL volumetric…
A: Dilution law: V1 × S1 (before dilution) = V2 × S2 (after dilution)
Q: Given: ___NH4NO3(s) --> ___N₂O(g) + ___H₂O(l) What volume of dinitrogen monoxide gas is produced at…
A: The balanced chemical equation is written below --> NH4NO3(s) --> N2O(g) + 2H2O(l) From the…
Q: 50 O Convert 203 °K into F
A: The given temperature is 203oK. To convert the degree Kelvin temperature to degree Fahrenheit we…
Q: which pH level has a hydrogen ion concentration ten times lower than pure water? pH scale is showing…
A: We know, pH = –log[H+] For pure water, Kw = [H+][OH–] = 10–14 And, [H+] = [OH–] So, [H+]2 =…
Q: Which of the following is evidence of a chemical reaction producing a gas? An Alka-Seltzer tablet…
A: There are two types of changes occur in chemistry. Physical change and chemical change. In the given…
Q: Calculate the maximum wavelength, Amax, of electromagnetic radiation that could eject electrons from…
A: Work function = 8.76 x 10-19 J Speed = 4.14 X 106 m/s
Q: Draw the major 1,2- and 1,4-addition products formed when this diene reacts with HBr. Do not include…
A: Diene has nucleophilic character hence it can give electrophilic addition reaction.
Q: To use the Beer-Lambert law to quantify the concentration of a compound in solution, the molar…
A: The Beer-Lambert law states that the absorbance of a solution is directly proportional to the…
Q: Titanium metal (d = 4.50 g/cm³) has a body-centered cubic unit cell. Calculate the edge length of…
A:
Q: How does the kinetic molecular theory of gases explain gas pressure?
A:
Q: When 26.62 moles of FeS₂ react with 5.44 moles of O2 in this reaction below, how many moles of FeS2…
A:
Q: 22. If you perform an acid base titration and place 50 mL of 0.25 mol/L acetic acid into an…
A:
Q: Calculate the moles of hydrochloric acid in the desired diluted solution. (To avoid introducing…
A: Given data, Concentration of diluted solution = 0.350 M Volume of diluted solution = 125 mL
Q: Q7. What is the degeneracy of the fifth energy level of a cubic box? A. 1 B. 3 C. 2 D. 4 E. 6 98,…
A:
Q: A calorimeter contains 150g of water at 24.6°C. A 110g block of molybdenum metal is heated to…
A: Mass of water, mw = 150 g Initial temperature of water, Tw = 24.6 °C Specific heat of water, cw =…
Q: Q14. What is the angular momentum of an electron in 2p hydrogen like orbital? A. 0 B. 0.23 h C. 0.40…
A:
Q: Draw the major product formed when 1,3-pentadiene reacts with this acid. Do not include any…
A: -> Diene has nucleophilic character hence it can give electrophilic addition reaction.
Q: Propose an efficient synthesis for the following compounds using the malonic ester or the…
A: Since you have posted a question with multiple sub-parts, we will provide the solution only to the…
Q: Data/Results (use these data) beaker one…
A: Density of water is calculated by using the formula given below. Density of water =mass of…
Q: How much would the temperature (in °C) of 263 g of water at 25°C increase if 33.9 kJ of heat were…
A: Amount of heat,Q required to change the temperature of a substance of mass ,m and specific heat…
Give Detailed Solution with explanation....(*don't give Handwritten answer))
Step by step
Solved in 2 steps with 1 images

- From this determine the identity of the IR spect. include bonds, would love to compare to my own work.Without counting hydrogens, determine which one of the following CANNOT be the unknownmolecule with molecular formula C7H8NOBr , and explain your reasoning.8. How many total electrons reside in MOs of symmetry in the following molecule?
- X, the molecule shown below, is not planar. The terminal ring at one end is actually skewed up relative to that at the other end. Is X chiral? Yes or no?( plz exaplain with diagram in detail)Show work with explanation needed of each options with structures...don't give Handwritten answer..don't use Ai for answering this



