The aquation of tris(1,10-phenanthroline)iron(II) in acid solution takes place according to the following chemical equation: Fe(phen) 3 2+ +3 H 30* +3 H 20 → Fe(H 20) 62* + 3 phenH If the activation energy, E a is 126 kJ/mol and the rate constant at 30°C is 9.8 x 10 3 min -1. what is the rate constant at 45°C? 9.6 x 10° min 1 1.1 x 10 min-1 9.3 x 10 min-1 1x10 min1 1.1 x
The aquation of tris(1,10-phenanthroline)iron(II) in acid solution takes place according to the following chemical equation: Fe(phen) 3 2+ +3 H 30* +3 H 20 → Fe(H 20) 62* + 3 phenH If the activation energy, E a is 126 kJ/mol and the rate constant at 30°C is 9.8 x 10 3 min -1. what is the rate constant at 45°C? 9.6 x 10° min 1 1.1 x 10 min-1 9.3 x 10 min-1 1x10 min1 1.1 x
Chemistry by OpenStax (2015-05-04)
1st Edition
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Chapter12: Kinetics
Section: Chapter Questions
Problem 82E: For each of the following reaction diagrams, estimate the activation energy (Ea) of the reaction:
Related questions
Question
Transcribed Image Text:Jave niISWCI
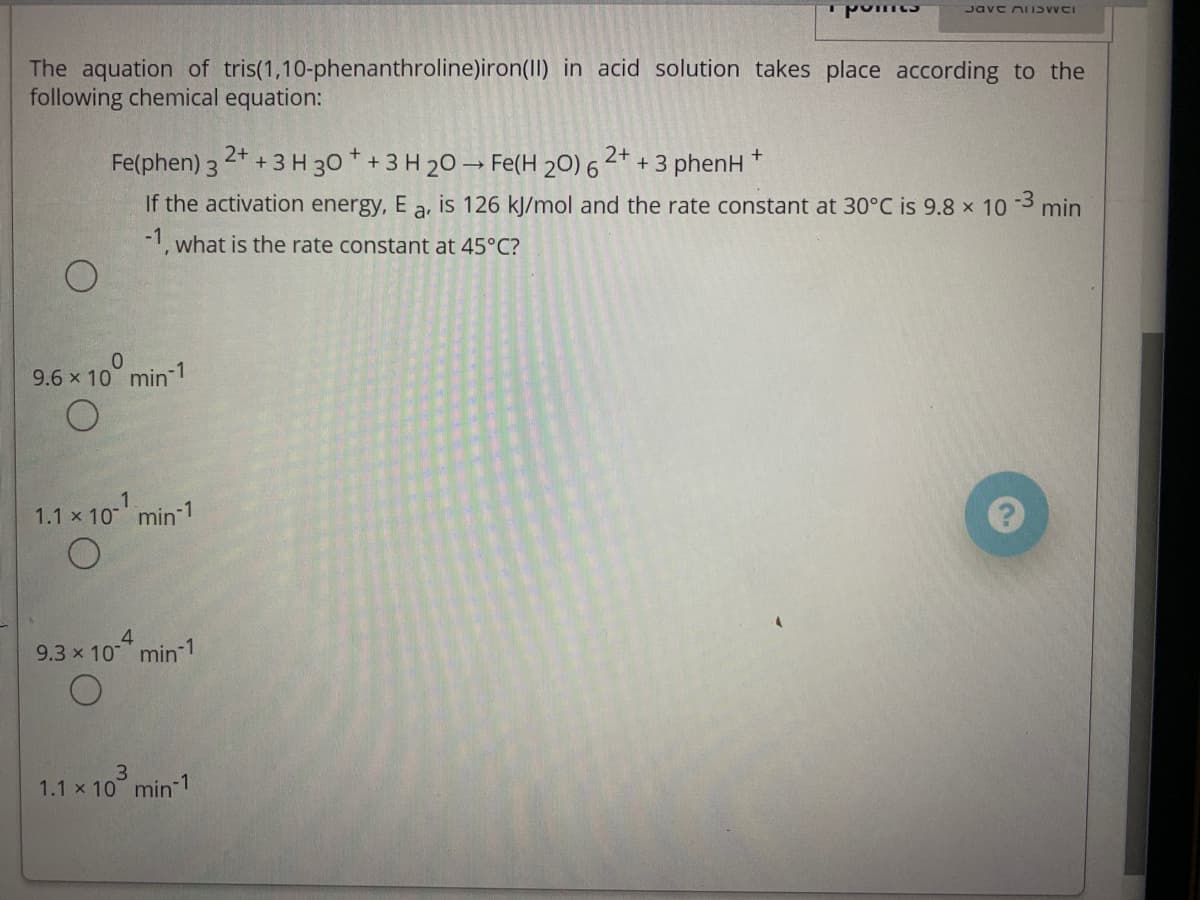
The aquation of tris(1,10-phenanthroline)iron(II) in acid solution takes place according to the
following chemical equation:
2+
Fe(phen) 3 2* +3 H 30 * + 3 H 20 → Fe(H 20) 6
+ 3 phenH
If the activation energy, E a, is 126 kJ/mol and the rate constant at 30°C is 9.8 x 10 3 min
-1, what is the rate constant at 45°C?
9.6 x 10° min-1
1.1 x 10 min 1
9.3 x 10 min 1
1.1 x 10 min 1
Expert Solution
Step 1
Given :-
Activation Energy = 126KJ/mol
Rate constant at 30℃ = 9.8×10-3 min-1
To find :-
Rate constant at 45℃ = ?
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Instrumental Analysis
Chemistry
ISBN:
9781305577213
Author:
Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:
Cengage Learning
