Q: Draw the structure corresponding to following IUPAC name. 4-isopropyl-2,4,5-trimethylundecane
A: The IUPAC naming gives the precise and accurate naming of the compound. The IUPAC name has three mai...
Q: Suppose that you have a fixed amount of a gas inside a box with fixed volume. When the temprature in...
A: Using the Gay-lussac law calculate the pressure at the second condition as shown below:
Q: Draw the structure for 2,4-pentanedione :
A: While drawing the IUPAC structure first identify the parent name and the functional group.
Q: How do the concentrations of Pb2+ and S2– change when K2S is added to a saturated solution of PbS?
A: On dissolving the K2S in the saturated solution of PbS, the concentration of S2- increases, and the ...
Q: In neutral solution containing only hydrogen peroxide and iodide ion, a different net chemical react...
A:
Q: 16mL of stock solution that has a concentration of 6.51M is added to water until the new concentrati...
A: Given: The volume of the stock solution, V = 16 mL The concentration of the stock solution, M1= 6.51...
Q: Find attached.
A: Homotopic Protons:As the name suggests the protons which are identical in nature is called Homotopic...
Q: Initial Temperature of Metal 200.0 C Specific Heat of Metal Mass of Water 250.0 grams Initial Temper...
A: The temperature of water increases from20oC to 24.08oC, so the water absorbs heat. That heat comes ...
Q: 6.) The diagram below shows the deposition of iodine vapor at room temperature. Which statement is c...
A: The ordered system is the indication of low entropy. While less orderliness is the indication of an ...
Q: 14) The phase/s in tempered martensite in a steel is/are: a) Body centered cubic Ferrite b) Monoclin...
A: Tempering is the metallurgical process. It is a heat treatment of tempered martensite in steels. Tem...
Q: Avogadro’s number is derived from the number of carbon-12 atoms in 12.00 grams. In a chemical react...
A:
Q: How much heat, in joules and in calories, is required to heat a 28.4-g (1-oz) ice cube from −23.0 °C...
A: Given : Mass of ice cube = 28.4 g Initial temperature of ice cube = -23.0 oC And final temperature o...
Q: Write a balanced chemical reaction equation and determine the reaction type for each of the followin...
A: The reactions in which a compound is breaks into two (OR) more simpler substances is called "Decom...
Q: What is Avogadro’s Law?
A: There are numerous laws of nature which are applicable in chemistry. Various scientists proposed a n...
Q: Chemistry Question
A: An atom is the basic and fundamental unit of matter. An atom consists of electron and the nucleus in...
Q: Explain the regiochemistry of the Friedel-Crafts alkylation of 1,4-dimethoxybenzene (there are three...
A: Friedel-Craft Alkylation is the type of electrophilic substitution reaction in which alkyl group beh...
Q: Predict the product.
A: The reaction involves the conversion of the alkyne group into the corresponding alkene group.
Q: The Periodic Table is named after the periodicity or repeating rise and fall of certain attributes. ...
A: Periodic trends:- These are the specific trends or patterns present in the periodic table, which def...
Q: Write a chart to show the balancing process for each half reaction and the combined redox in an acid...
A: Separate the reaction into two equal halves and balance individually and add after it to get the ful...
Q: What is the IUPAC name of the following amine?
A: For naming of amines carbon attached with NH2 group is taken into longest chain.Naming is done as al...
Q: What is the limiting reactant if 2.2 g of Mg is reacted with 4.5 L of oxygen at STP?
A:
Q: What is the degree of ionization (i) for KMNO4(aq)? A) 1 B) 2 C) 3 D) 4 E) 6
A: Degree of ionization is the number of ions(anions and cations) formed of a particular compound which...
Q: Calculate the percentage of isopropylcyclohexane molecules that have the isopropyl substituent in an...
A: Given: Calculation 1 First, calculate the equilibrium constant for the reaction shown below.
Q: Find the molecular formula or each given compound. Please help with both. Thank you.
A: For 1st compound, Empirical Formula = C2HCl Empirical formula Mass(Emass) = (2 x 12) + 1 +35.5 = 60....
Q: List the sources of error associated with pH measurements made with the glass electrode.
A: The pH electrode is usually made of glass. Coatings on the glass surface can alter the pH readings...
Q: Show how you would use simple chemical tests to distinguish between the following pairs of compounds...
A: Given, cyclohexanol and cyclohexene
Q: Explain why an alkyne often has a slightly higher boiling point than an alkene of similar molecular ...
A: Introduction- Alkene is an organic compound with 1 or more double bond as unsaturation and it is hav...
Q: 1. In 1992, the first planets outside the Solar System were detected approximately 1300 light- years...
A: 1. As 1 light year =3.725×1017 inches
Q: Equation: Pb(OH), + Cu,0 → PbO, + CUOH Balanced Equation: Word Equation:
A: Given, the chemical reaction as follows,Pb(OH)4 +Cu2O→PbO2+CuOHwe are asked to balanced the above ch...
Q: What is syndiotactic polymers ?
A: Syndiotactic Polymers: the polymers which have group attached in an alternate manner is called syndi...
Q: Which of the following is the least acidic compound? IV Multiple Choice II IV
A: The order of the acidity from lower to higher are: III<IV<I<II
Q: You carefully weigh out 17.00g of CaCO3 powder and add it to 68.85 g of HCl solution. You notice bub...
A: CaCO3 (s) + 2HCl (aq) → H2O (l) + CO2 (g)+ CaCl2 (aq) Given, Mass of CaCO3 =17.00 g Mass of HCl So...
Q: Given the following mass spec data, what is the molecular formula of the compound? (Show pertinent c...
A: We have the mass spectrum data and we need to figure out the molecular formula of the compound. Car...
Q: A blacksmith heats a 1,540 g iron horseshoe to a temperature of 1445°C before dropping it into 4,280...
A: Specific heat (s) is the heat required to raise the temperature by 1°C of the substance. It has the ...
Q: Chrysanthemic acid occurs as a mixture of esters in flowers of the chrysanthemum (pyrethrum) family....
A: A step by step way of representing a chemical reaction is called reaction mechanism . Curved arrows ...
Q: Vhich one of the following can be classified as a weak electrolyte? A) HBr B) CaF2 C) OBR2
A: if the compound is polar hen it may show same electrolyte properties .Non-polar compound do not exhi...
Q: Write the elemental symbol for each of the following atoms and calculate the number of neutrons: (a)...
A: No of Proton= No of electron=No of atomic Number (a):- atomic No is 55, no of proton is 55 (b):- Ato...
Q: a galvanic cell is constructed of a silver electrode immersed in 0.1M silver nitrate solution and a ...
A: For standard hydrogen electrode [ H+ ] = 1.0 M standard reduction potential E°(H+/H2) = 0.0 V Given ...
Q: Chemistry Question
A: Given : Mass of steam to be condensed = 3.5 Kg = 3500 (since 1 Kg = 1000 g...
Q: In the laboratory, hydrogen chloride (HCl(g)) and ammonia (NH3(g)) often escape from bottles of thei...
A: Given that, the partial pressures will be equal and are at their maximum value when at equilibrium. ...
Q: Carbon has two stable isotopes, 12C, abundance 98.892%, and 13C, abundance 1.108%. In the mass spect...
A: The molecular weight peak is called the Molecular Ion peak (M). Since this peak is a radical cation,...
Q: What is the purpose of derivatization in chromatography? Give an example.
A: The technique or process used for the separation of mixture of components present in the mixture is ...
Q: Potassium ion Copper (I) ion Aluminum ion
A: Since you have posted a question with sub-parts , we are entitled to answer the first three sub-par...
Q: Calculate the amount of heat needed to convert 74.6g of water at 25°C to steam at 100°C.
A:
Q: For conjugate acid-base pair, identify the first species as an acid or a base and the second species...
A: The species CH3OH loses a proton to form CH3O- and H+. Hence CH3OH is an acid Lewis structur...
Q: Identify each of the following groups as an activator or deactivator and as an o,p-director or m-dir...
A: Activators are electron releasing groups and are generally o,p- directors whereas deactivators are e...
Q: Equilibrium constant is related to Eθ cell but not to Ecell. Explain.
A: The Nernst equation is given by, Ecell=Ecell0-0.0591nlog k where Ecell0 is the standard cell potenti...
Q: A solution is made using 90.1 g of dimethyl ether (MM = 46.07 g/mol) and 110.0 g of methanol (MM = 3...
A: Molality of the solution is equal to the number of moles of solute present in one kilogram of the so...
Q: i think one of them is wrong idk.
A: Number of moles = mass/molar mass Molar mass of CO = 28 g/mol Molar mass of H2 = 2.0 g/mol Molar ...
Q: The following diagram represents a buffer composed ofequal concentrations of a weak acid, HA, and it...
A: A buffer solution is an aqueous solution that consists of a mixture of a weak acid along with a conj...
Show how the following compound can be prepared from methyl phenyl
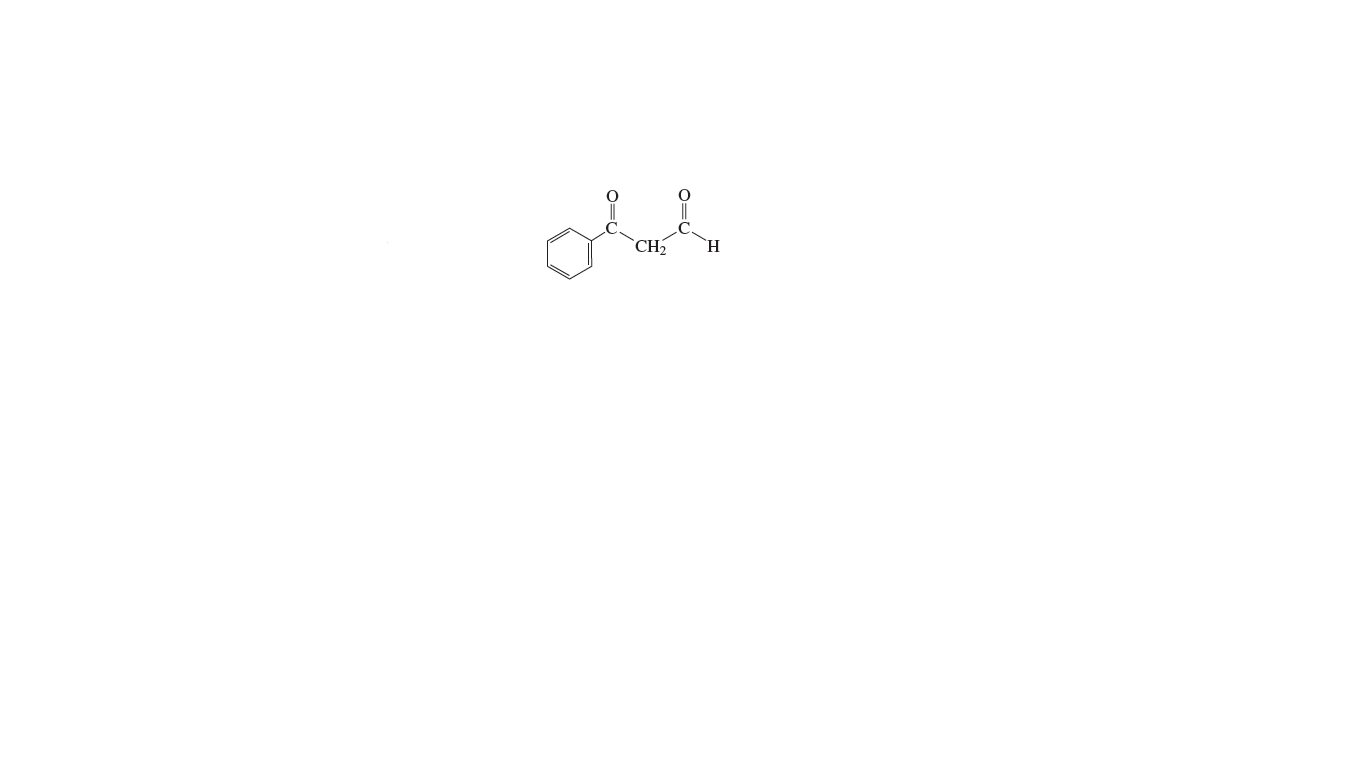
The organic compounds which contains –CO– group as the functional group are termed as ketones. In methyl phenyl ketone, there is –CH3 group on side of –CO– group and –C6H5 group on the other side.
Step by step
Solved in 2 steps with 1 images

- Provide two examples of thermoresponsive polymer and its applications in modern technologiesNumber of components in CH3COONH4a statement about the scale of plastic production and how this has changed over time using simple lanaguge becsuse it is writte for kids. a statement about the scale of plastic production and how this has changed over time using complex lanaguge.





