Q: Redox Titrations p:chromade= 24EEM Titration of Tin (1I) with Potassium Dichromate Trial #1 Trial #2...
A: The equation of this titration is , 3Sn2+ +Cr2O72-+14H+ ---->3Sn2+ +2Cr3+ +7H2O Oxidising agent--...
Q: How many grams of AgNO 3 (molar mass = 169.87 g/mol) are needed to prepare 0.125M solution in 250 mL...
A: Given - Molarity = 0.125 M Molar mass = 169.87 gm.mole-1 Volume = 250 ml = 0.250 L (1L = 1000ml)
Q: A student attempts to determine the buffer capacity of a solution by titrating 40.0 mL of solution w...
A: Given: Volume of buffer solution = 40.0 mL = 0.040 L (since 1 L =...
Q: Illustrate the electron spin quantum number of Boron
A: Quantum mumbers are used to describe position of electrons in orbitals of an atom. There are four di...
Q: Determine the concentration (M) of H3O+ in a 0.577 M solution of a triprotic weak acid, H3A, given t...
A: Here we have to determine the pH of 0.577M triprotic acid H3A . Given Ionization constants are Ka1= ...
Q: What is the use of 95% ethanol in caffeine extraction from tea?
A:
Q: Provide the reaction mechanism.
A:
Q: Calculate the percent yield of the reaction if 1.88 g of aspirin (acetylsalicyclic) were obtained
A:
Q: lustrate the electron spin quantum number of Fluorine
A: Given : We have to write electron spin quantum number of Fluorine.
Q: calculate the pH a solution prepared by adding 20.0 mL of 0.020 M NaOH to 0.10 M Na3AsO4.
A:
Q: When aqueous solutions of NaBr and AGNO3 are mixed, the reaction produces NaNO3(ag) and AgBr(s). Wha...
A: Correct option is (C)
Q: (1) What is the valence electron configuration for the krypton atom? (2) What is the valence electro...
A:
Q: एकचर्चर नेकट ा ४ mssme missmg greagend ठेवतत A)1. Li Al Hy 2 द00 anके Bar (2 09) 2. H301 १. ५३० CH3I...
A: Organic reactions are those in which organic reactant react to form organic products. In this reacti...
Q: How many moles of sodium hydroxide would have to be added to 125 mL of a 0.339 M hypochlorous acid s...
A:
Q: For the central atom in each formula, draw the Lewis Structure with all valence electrons shown. 1. ...
A:
Q: 25) Consider the following set of reactions. Assume that A, B, C and D are atoms. (1) A + A → B (2) ...
A: A question based on kinetics of reactions in solution that is to be accomplished.
Q: Q: What is the difference between addition polymerization and condensation polymerization? In additi...
A: A polymer is a large molecule that is formed by the repetition of small units called a monomer, and ...
Q: a) 100 2-methyl-2-propanol 80- 40 20 0.0tt 10 20 30 40 50 60 70 80 m/z b) 100 n-propyl acetate 80 40...
A: Fragmentation of 2-methyl -2-propanol shows the elimination of small molecule OH , Methyl, H2O and ...
Q: 1. Write oxidation half-reactions for the metals Ag, Cu and Zn. Ag: Cu: Zn:
A:
Q: Substance State KCo,
A: State of K2CrO4: Potassium chromate is a yellow crystalline solid. It is soluble in water.
Q: Why do you think that understanding the principles behind colligative properties of solutions is ben...
A: Colligative property of a solution is defined as the property which depend upon the concentration of...
Q: CARBONYL CHOMISTRY - NUCLEO PHHL IC d- SUBSTITUTION NaocHy octy •octly 3) reflux Clue : Naoctty ects...
A: The reaction is a classic example of Favorskii rearrangement. It is a rearrangement of alpha-halo k...
Q: List the types of intermolecular forces that exist between molecules (or basic units) in each of the...
A:
Q: The properties of the polymers are usually governed by its structure. One such import property of po...
A:
Q: Consider the rate law: Rate = k [A]2[B][C]1/2. How does the rate of the reaction change if the conce...
A:
Q: Activity 1 List 15 isomers of Decane and name them using the IUPAC system of nomenclature - (Use the...
A:
Q: Note: I need a simple explanation for this lesson for my presentation. Thank you!
A: Polymers are composed of many small repeating units (monomer) joined together. Polymers having hig...
Q: For each of the following substances, identify whether the substance is classified as an Arrhenius a...
A:
Q: Follow up questions: 1. What happens to any solid that's added to an already saturated solution?
A: As per bartleby guidelines I answered only first question so please don't mind.Thanks in advance.
Q: Propose a structure for an amine of formula C,H,N, which liberates a gas when treated with NaNO, and...
A: The NMR spectroscopy is an important tool for the determination of the structure of an organic compo...
Q: (c) Estimate the wavelength of electrons that have been accelerated from rest through a potential di...
A:
Q: If 2.7 kg of Na2CO3 concentration of CO32- ? are dissolved to make 4.6 L of solution, what is the An...
A:
Q: Added 45g water to 150ml flask. Initial temp =21.5C Placed flask in a constant temp. bath at 60 degr...
A: Heat energy absorbed or released in any process is given by Q = m c (T2-T1) where, Q = heat energy,...
Q: raw a picture of the following orbitals: A. 1s, 2s, and 3s on the same set of axes B. 2s and 2px o...
A: Given : We have to draw these orbitals keeping the axes commmon.
Q: A 25.0 mL solution of 0.100 M CH3-COOH is titrated with a 0.200 M NaOH solution. What is the pH afte...
A:
Q: Part A Draw the condensed structural formula of the amine salt formed when lidocaine reacts with HCI...
A: GIVEN:- Draw the condensed structural formula of the amine salt formed when lidocaine reacts HCI.
Q: Öne can review the blast furnace from a simple viewpoint as a process in which the principle reactio...
A: Solution - According to the question - Given -
Q: Gaseous SO2Cl2 reacts with liquid water to form hydrogen chloride gas and liquid sulfuric acid. Usin...
A: Given: Gaseous SO2Cl2 reacts with liquid water to form hydrogen chloride gas and liquid sulfuric aci...
Q: Given that the rate constant for a reaction is 5 ×1010 at 25 °C, what is its rate constant at 37 °C?...
A:
Q: The antibiotic Prontosil, developed by Bayer Pharmaceuticals in 1932, was used in saving the life of...
A: The product given is,
Q: Q1: Give IUPAC names for the following compounds: CH3 CH2CH3 CH;CH=CHCHCH2CHCH,CH2CH3 a. b. ČH;CH2CH...
A:
Q: a. This mo Br sites of electrop HO. you've c and we'l b. он С. HO.
A: Here we have to write IUPAC name and the key reactive sides present in the given compounds.
Q: Please answer all questions below with calculations. 1. What is the empirical formula of a hydrocar...
A: Since you have asked multiple questions questions, we will solve the first question for you. If you ...
Q: Identify all of the IMF's present between like molecules in a sample of CH3F. Dipole force O Hydroge...
A: Intermoleular forces can be defined as the forces existing between two or more than two same or diff...
Q: The progress of rigor mortis through the body starts at the ___. hands and feet, moving toward...
A: Please find your solution below : Rigor mortis is the third stage of death. It is the stage in which...
Q: What can affect the formation of different crystalline phases
A: A question based on phase diagram that is to be accomplished.
Q: Using activities, calculate the pH of a solution containing 0.015 M HCI, 0.036 M HCI and 0.050 M NaN...
A:
Q: How many moles of CO2 (g) are present after 25.0 mL of dry ice sublimes? Do not include the unit of ...
A: Number of moles of dry ice is given below
Q: Туpes of Combustion (partial or complete) Compound Class of Saturated or Hydrocarbon Unsaturated n-Н...
A:
Q: Which of the following has the correct notation for the ground state electron configuration? O Ca; 1...
A: Ground state electronic configuration of an element is written using its atomic number and aufbau pr...
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

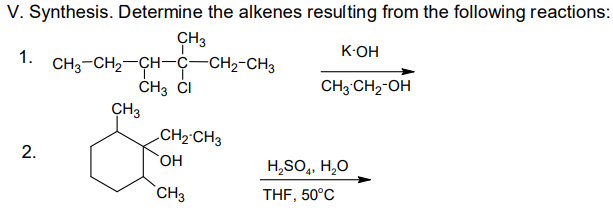
- Most methods of making alkenes yield predominately the more stable isomer, usually the trans. Outline all steps in the conversion of a mixture of 75% trans-2-pentene and 25% cis-2-pentene into essentially pure cis-2-pentene.Please predict the outcome of the addition of HBr to (a) trans-2-pentene, (b) 2-methyl-2-butene, and (c) 4-methylcyclohexene. Also please list how many isomers can be formed in each case? Thank youWhich of the following is most soluble in basic medium? a. cyclopropane b. 1,3-cyclobutadiene c. 1,3-cyclopentadiene d. benzene
- Table 6.4. Oxidation: Reaction with Baeyer’s reagent (cold, dilute, neutral, aqueous KMnO4) Write (+) if the reaction is positive, (-) if negative. SAMPLE OBSERVATIONS +/- cyclohexane cyclohexene benzene toluene acetylene Enumerate the classes of hydrocarbons that are oxidizable by Baeyer’s reagent. ________________________________________________________________________ ________________________________________________________________________ What is the structural feature of these classes of hydrocarbons? ________________________________________________________________________ ________________________________________________________________________Draw the one most stable alkene that is formed when the structure shown below is heated with H₃PO₄. .How to draw the more stable conformer of trans-1-ethyl-2-methylcyclohexane? Thank you.
- A chemist allows some pure (2S,3R)-3-bromo-2,3-diphenylpentane to react with a solution of sodium ethoxide(NaOCH2CH3) in ethanol. The products are two alkenes: A (cis-trans mixture) and B, a single pure isomer. Under thesame conditions, the reaction of (2S,3S)-3-bromo-2,3-diphenylpentane gives two alkenes, A (cis-trans mixture) and C.Upon catalytic hydrogenation, all three of these alkenes (A, B, and C) give 2,3-diphenylpentane. Determine the structuresof A, B, and C; give equations for their formation; and explain the stereospecificity of these reactions1. Using Br2 in C2H4Br2 will result in HBr and ______. a. C2H3Cl3 b. C2H4Cl3 c. C2H2Cl3 d. none of the above 2. How many halogenation are posible in propane? a. 3 b. 8 c. 6 d. 10 3.Sulfonation of pentane will result in ________ and water. a. C5H11SO3H b. C5H12SO3H c. C5H14SO3H d. none of the above 4.Nitration of hexane will result in ________ and water. a. C6H13SO3H b. C6H15NO2 c. C6H13NO2 d. C6H14NO2 5.How many moles of O2 in heating a C12H26 (dodecane) a. 27 b. 37 c. 24 d. none of the abovePlease help with both parts Labeling the trans-1-ethyl-2–isopropylcyclohexane and determining which version is more stable

