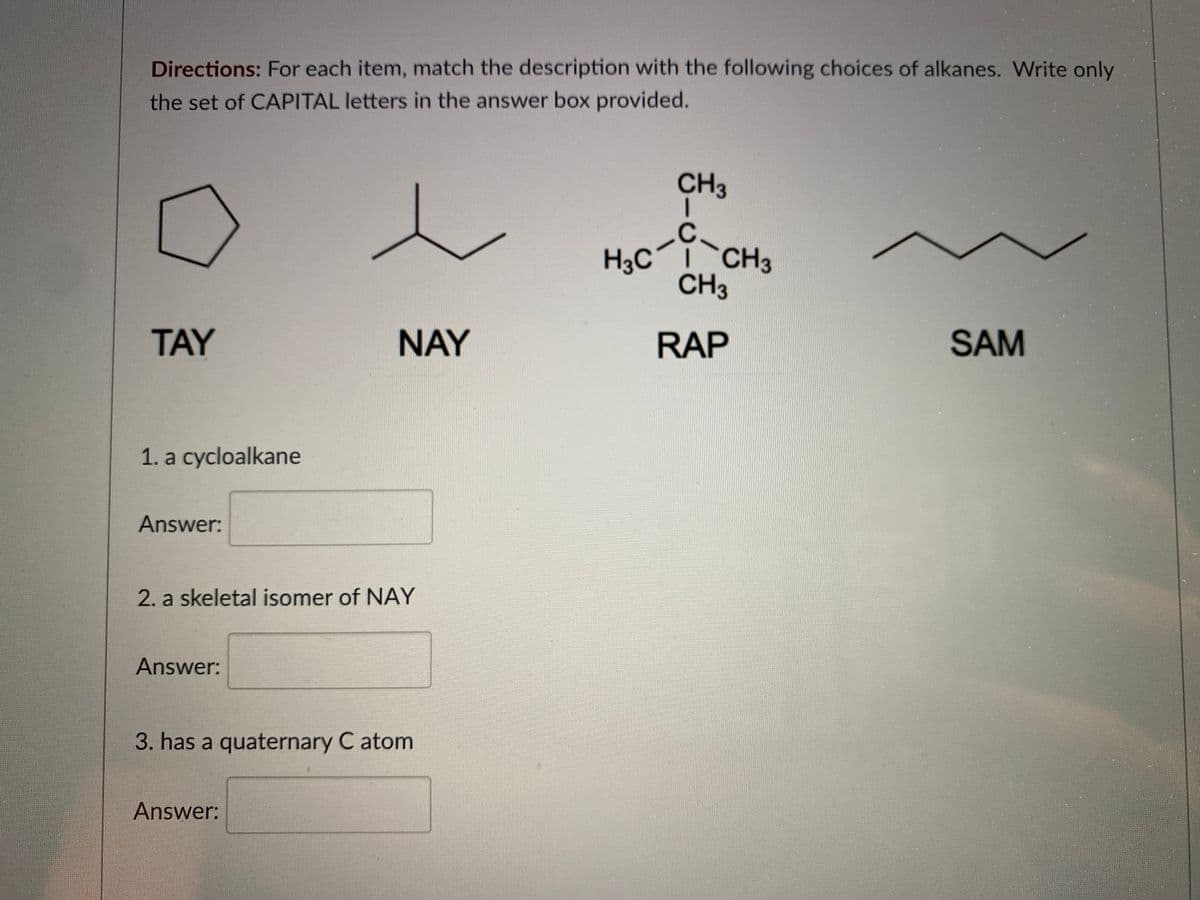
Q: ?What is the IUPAC name of the following compound (CH3CH2)½C=CHCH,CH2CH2CH(CH3)2…
A: IUPAC name of the compound is: 3-Ethyl-8-methyl-3-nonene
Q: 2. Name each of the following functional Groups. H R-C-OH R-C-OH H H R-C-0-CH3 H C-CIRC=CH₂ RC=CH i…
A: Since, Functional group is the collection of atom which is application to identify what type of…
Q: iccording to the IUPAC system: CH2 H.C 2. ( Name this according to the IUPAC system, including…
A: Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: The most stable conformation of the compound to the right has CH2CH3 A) an axial methyl group and an…
A: This is a cyclohexane with three substituents attached to it. The cyclohexane is found in chair…
Q: Give the IUPAC name for the following compound. CH3 H H CH,CH3 CH3-C c-C-C-CH3 H H ČHĄCH,CH,CH3
A: IUPAC is an international federation of national adhering organizations. It is the authority for…
Q: 2 C,H MgBr H,0 H3C CH3 CrO3 ----> 1-pentanol OH
A:
Q: 4) Identity the longest Curbon chain (wrife carbons) a) CHs GC- C- c=C CHs - -८-८- -C- c) d) -C- C -…
A: The primary carbon chain in the IUPAC nomenclature is the chain containing the largest number of…
Q: C Give The IUPAC Name for the following Compounds CH3 2 HS 3,Ct3 CH= CH-CHz-CH3 4, CCH),-CH- C =CH…
A:
Q: CH2 H2C CH3 CH-C-CH3 H2C. CH ČH3 CH3-CH-CH3
A: The IUPAC name of the molecule is explained as follows:
Q: The condensed formula of 2,3.4-trimethy lpentane is which of the following? CH3 24. c. a. CH3 CH3…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 1. Complete the following diagram so that it represents (R)-2-bromobutane. 2. Complete the following…
A:
Q: ОН H .N. .CH3 CH3 H,N
A: Here we have to predict the types of streoisomerism shown by the following compound. The streoisomer…
Q: ل طبيب 25 ولت اكول شلون د 2, CH3 CH₂ CH₂ CH 7-tert-butyl-8-isobutyl-4-isopropyldodecane CH…
A:
Q: Draw cyclohex-1-enecarbaldehyde
A: Draw structure of cyclohex-1-enecarbaldehyde
Q: Give the IUPAC name for each compound. Br CH3 a. CH3-C-CH2CH,F ČH3 c. (CH),CCH,Br g.…
A:
Q: C-C-C-C-C Structiral formula for CuHi2 AIkene DAh douke bond at Carbon 2 that Shows as-somensm.…
A: IUPAC Name: Hex-2-ene The double bond is on C2. It is a cis alkene. Both the least priority groups…
Q: These two compounds have “butane” in their name. Are theyisomers?
A: The 2 compounds given are
Q: Below is a H но H но -N(CH2)6N--Ċ-(CH2)«C-N(CH2)6-N-C(CH2)4--C~~ n O sugar O nylon O fat O polyester
A: A sterogenic center or a chiral center is defined as an atom that is bonded with different groups of…
Q: Now, The condensed form is. HC -CH₂ сна CHI CH CH₂ CH3 3,4-diethyloctane HC- IUPAC name is i- 52…
A: Condensed structure of any organic compound is the representation of that compound in a way where no…
Q: Classify each compound as identical to A or its enantiomer.
A: If any molecule is non-superimposable mirror image of A , then the other compound is called…
Q: Which structure is trans-3-hexene? OCH₂ H CH3. H OCH₂CH₂ H c=c OCH3CH₂ H FC. FC FC. H CH₂CH₂CH3…
A: Which structure is trans-3-hexene ?
Q: 3. Give IUPAC names of the following structures. Clearly show the numbering, H. CH, CH CH CH HyC CH…
A: Some rules to follow for writing names organic compounds: Identify the longest carbon chain. This…
Q: Provide the proper IUPAC name for the alkene shown below (indicate R or S). CH3 "CH,CH3
A:
Q: Below are representations of isopentane except H-C- -C–H CH3-C-H ČH3 HICI エーO-0-エ
A:
Q: is CH;CH2 -C-H An isomer of CH;CH, CH3-C-H CH3-C-CH3 O CH3-O-CH2CH3 O CH3CH2CH2-OH
A:
Q: CH =C - CH – Coci i CH;
A: In the above given molecule two functional groups are present i. e. a triple bond, a acid chloride…
Q: name of the following alkene is CH(CH3)=CH-CH(CH3)(CH2CH3)
A: Alkenes are the type of unsaturated hydrocarbons that consist of one or more double bonds between…
Q: CH3 CH3-CH2-C-CH3 CH2 CH2 CH3 methyl-2-propylbutane ethyl-2-methylpentane
A: It is polysaccharide that our body makes and stores in liver to use as fuel storage
Q: NAMIE Ihe Compound HE- Cts CH CH- CH - T. CH3 CH3 CHS
A: Given, here a compound and we are asked to write the name of compound.
Q: CH3-CH2-OH reacts with H+/140C = ? + H2O
A:
Q: NH2 C' HC C-c=C- H2C-HC CI H2 HC
A: On basis of saturation the organic compound can be classified as saturated and unsaturated…
Q: CHO H. OH (HỌ -H- OH -ОН CH2OH 2. The above structure is glucose
A: Please find your solution below : Pyranose structure is a 6-membered ring with oxygen as hetero…
Q: Tell whether the following pairs of compounds are identical constitutional isomers, or…
A: Identical compounds are those compounds which have all atoms in same spatial orientation.…
Q: 2. Constitutional isomers or stereoisomers? a) OH OH b) CH3 CH3 CH3 OH CH3 c) Он ОН d) OH CI ÕH
A: Hi, since you have posted multiple subparts, we will answer the first three subparts for you. Kindly…
Q: CH3 H2SO4 H2C=C + H3C-OH CH3
A: The given reaction is of 2-metylprop-1-ene with methanol in presence of H2SO4 . THE Presence of…
Q: 2. Name the following Alkanes using the IUPAC nomenclature rules: HHHH Н-С-С-С-С-Н HHHH a. H- HH- H…
A: In the given questions we are provided by three structures and asked to give nomenclature.
Q: 5. Give IUPAC names for the following compounds: (a) CH3 (b) H3C CH3 (c) CH3 *CH3 H
A: Since you have posted multiple questions, we will answer the first one for you (first 3 sub-parts of…
Q: (a) Name the compounds whose structures are shown below: CH-CH-CH3 či CH (ї) Н (ii) CH,CH H2C=C-CH…
A: The IUPAC name of the compound can be written on the basis of the number of carbon atoms in the main…
Q: The name of CH3CHBRCH=CHCH3 is - O 4-bromo-2-pentene O 3-bromo-2-pentene O 2-bromo-3- pentene
A:
Q: Give The IUPAC Name for the followiny Compounds CH3 C2 HS 3, Ct3 CH= CH-CHz -CH3 4, CH),-CH-C =CH…
A:
Q: 0:2.which of the Compound show geametrical Isomerism ? Bs Bo C-CH= CHL CH3 -C=C - CH z CH3, CH CH CH…
A: Geometrical Isomers are the structures of the same compound which have the same molecular formula…
Q: H3O+ /A a. H3C-OH H3O+ /A b. CH;CH-OH POC3 / pyridine C. HO. OH H2SO4 /A d.
A: a. Methanol undergo protonation to give protonated methanol which is attacked by CH3OH to give…
Q: CH3-C C-CH3 CI ČH2-CH3 2-chloro-3-ethyl-2-buteno -
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: + HCI CH:- CH-CI + CH3
A: substitution reaction are reaction in which one functional group which is good leaving group is…
Q: HI H3C-0-CH2 CH3
A:
Q: 2) For the following steroid skeleton: H. Ме 11H H7 Ét a) If a substituent is placed at C-11 in the…
A: The above compound has a fused ring structure with trans trans ring junction which means that the…
Q: ns of Alkenes (Ch. configuration (Ch. mations (Ch.3) ns of alcohols (Ch. atomy of the Humai
A: Chemistry is Broadly classified in three parts Organic Chemistry inorganic Chemistry physical…
Q: Classify each pair (a and b) of compounds as constitutional isomers, stereoisomers or identical…
A: Constitutional isomers are the isomers which have the same molecular formulas, but they have…
Q: H2 CH2= (= CH- CH3 catylyst
A:
Step by step
Solved in 2 steps

- How many moles of Bra are required to completely halogenate the alkene?A. One moleB. Two molesC. Three molesD. Four moles What is the expected arrangement of the bromine atoms relative to each other amongthe carbon involved in pi bonding?A. anti-conformationB. syn-conformationC. trans-configurationD. cis-configuration What happens to bromine when it is adjacent to an alkene during a chemical reaction?A. Bromine becomes stable. (? kasi before brown siya/acidic tas naging colorless? Jk ewan)B. Bromine becomes polarized.C. Bromine becomes hybridized.D. Bromine becomes acidic. The relative arrangement of bromine atoms in the product is primarily due to:A. ElectronegativityB. RepulsionC. Hydrogen bondingD. Atomic weightWhat is your observation after the reaction?A. A yellow flame is produced.B. Bromine water decolorizes.C. The alkene becomes denser.D. A brown precipitate forms.Provide a molecule that contains the indicated functional groups and molecules. Each functional group must be single on your examples. Give an IUPAC accepted name for your molecules. a) Bicyclo compound: Alcohol (alcohol functional group must be on the 3° carbon atom), alkene, one cyclohexane and one cyclopentane. Plz do Asap ...! And send ur ans in clear manner ...!Give IUPAC name of the following molecule and indicate Z or E. Answer all four of them
- Draw the most stable conformation of the disubstituted cyclohexane below.Consider the molecule 1-bromo-2-methylbutane. C3 and C4 should be drawn as Et as in theexample. This group is called an ethyl group and can be considered a sphere about twice the sizeof a methyl group. Draw the following Newman projections sighting down the C1C2 bond... a. The lowest potential energy conformation. b. The highest potential energy staggered conformation.Draw the structures based on iupac name digitally (highly suggest chem-space .com for easy drawing) 1,1-Dibromo-4-isopropylcyclohexane 4-sec-Butyl-2-chlorononane 1.1-Dibromo-4-Pert-Butylcyclohexane
- Name each of the following alkenes. This is all one question please help with parts A,B,C,D.Please synthesize a Butene -that includes 4 different reactions -includes functional groups -please include 2 C-C bonds and a detailed arrow pushing mechanism.According to the following structure, draw the Newmann projections in which you must indicate: a) the most stable conformation and explain why it is more stable, b) the least stable conformation and explain why; c) a conformation where there is a gauche interaction.
- How to solve this newman projection cyclohexane question?Hello, I want a simple explanation on this topic (Stability of cycloalkanes) Can you explain to me when it is (axial and equatorial) and how it defines the most stable form? I need to explain the concept and mention the cases on this topic. Thank youRank the alkanes below in order of increasing stability:



