Q: Draw the product (including stereochemistry) formed from each pair of reactants in a thermal [4 + 2]…
A: The thermal [4+2] cycloaddition reaction of the given reactants is shown below.
Q: 4. Draw all resonance structures for the carbocation formed by ortho attack of the electrophile…
A: Concept is based on resonance.
Q: Draw the structures of A -D from the following electrocyclic and cycloaddition reactions. CH heat hv…
A:
Q: The accepted mechanism for the following transformation involves a carbocation rearrangement. Rather…
A: Note: step 1 : proton addition to double bond results secondary carbocation. Step 2 : a carbon of…
Q: 5. Predict the structure of the product formed (with correct stereochemistry) for each of following…
A: Since you have asked multiple questions, we will solve the only first question for you. If you want…
Q: In the following reaction, what is the configuration of the major products? Cl2 CH3OH O No…
A: the reaction of alkene in presence of chlorine, the reaction proceeds with the formation of…
Q: Classify each reaction as an electrocyclic reaction, a cycloaddition, or a sigmatropic…
A: In organic reactions there are many types of reactions like elimination, addition and substitution.…
Q: 6. Provide the line structure of product E in the following reaction. Include stereochemistry for…
A:
Q: 12. Draw the products of the following reactions. Indicate stereochemistry when relevant. a CH-C =…
A: The reactions are provided below:
Q: 1. a) Draw the structures of A - D from the following electrocyclic and cycloaddition reactions. hv…
A:
Q: Draw the missing product from the reaction below. OTs K* "OC(CH3)3 + K*¯OTS + HOC(CH3)3 draw…
A:
Q: Draw the structures of A - D from the following electrocyclic and cycloaddition reactions. hv CH3…
A: The key steps involved in these process are ring opening reaction and 4+2 cycloaddition reaction to…
Q: the major product obtained upon addition of Br2 to (R)-4-tert-butylcyclohexene
A:
Q: Identify the following pericyclic reaction; explain the course, product distribution and…
A: To identify the pericyclic reaction, explain the the course, product distribution and…
Q: Draw the reaction product for the following Sy2 reaction: NASH Q5.2 What is the best description of…
A: Interpretation - To draw the product of the given SN2 reaction - To explain which type of product…
Q: Draw the mechanism and product for each of the following Diels–Alder reactions.
A: The given reactions are the example of [4+2] cycloaddition reaction. The reactions take place in the…
Q: d) The Diels Alder reaction between diene 4 and dienophile 5 can give four products, draw the four…
A:
Q: Classify each reaction as an electrocyclic reaction, a cycloaddition, or a sigmatropic…
A:
Q: Draw a representation of the transition state for the following endergonic mechanistic step.…
A: To find: the transition state
Q: 15. Consider the substitution reaction: The product(s) of the reaction is(are): a. R enantiomer C.…
A: Hence option (c) is correct answer..
Q: 27. Draw a representation of the transition state for the following exergonic mechanistic step. ..O…
A: In this equation, we will draw a transition state for given mechanism. How we can draw a transition…
Q: Classify each pericyclic reaction as an electrocyclic reaction, cycloaddition, or sigmatropic…
A: (a) Concept introduction: Electrocyclic reactions are pericyclic reactions which takes place by…
Q: (2R,3S)-2-Bromo-3-phenylbutane undergoes an E2 elimination when treated with sodium methoxide. Draw…
A:
Q: Markovnikov Hydration w/ possible rearrangement – H30* (SYN & ANTI) H H H H. :ö-H Scenario A H H H.…
A: Hello. Since your question has multiple sub-parts, we will solve first three sub-parts for you. If…
Q: Name the kind of sigmatropic rearrangement that occurs in each of the following reactions. a. Using…
A: Sigmatropic rearrangement reactions are named with digits. For example a [1, 3] sigmatropic…
Q: Draw the products of each reaction and indicate the stereochemistry atany stereogenic centers.
A: (a)
Q: Classify each reaction as an electrocyclic reaction, a cycloaddition, or a sigmatropic…
A: Electrocyclic Reaction is a type of pericyclic reaction in which one pie bond is converted into one…
Q: 3. Explain the stereochemistry of the product by drawing the orbitals in the following electrocyclic…
A: Bb
Q: When 1,4-dimethylcyclohepta-1,3-diene is treated with HBr at elevated temperature, the 1,2-adduct…
A: We have find out why 1,2 adduct product is predominant.
Q: 2 Conseeutive 3,3- sigmatropic rearrangements 2.
A:
Q: 2. Consider the E2 elimination of compound A and answer the following questions. D H t-BUOK Products…
A:
Q: 7-55 trans-1-Bromo-2-methylcyclohexane yields the non-Zaitsev elimination product…
A:
Q: e. Sn1 SN2 E1 E2 Initial Stereochemistry = R OH H3C-CH2- CH CH3 + Н3РО4 Product/Stereochemistry(ies)…
A: According to question we have to write products of reactions .
Q: Draw the final product of this series of reactions. |||** OH 1. SOCI₂ 2. NaCN • Use the wedge/hash…
A: In presence of SOCl2, the given compound undergo SNi mechanism , there will be retention of the…
Q: Q4. The major product(s) of the following E2 reaction is/are: CH CH,ONa Br CH3 E2
A: Biomolecular elimination (E2) reaction:E2 reaction involves a one-step mechanism in which…
Q: Which reaction mechanism will create a shift in the stereochemistry of the resulting product? a…
A: In SN1 and SN2 , substitution takes place but in E1 and E2 elimination takes place.
Q: The major product formed by addition of HBr to (CH3)2C=CH– CH=C(CH3)2 is the same at low and high…
A: Chemical reaction in which an atom or a group of atoms are added to each carbon atom of a…
Q: HO products -CH3 + H. Br2 H,C
A:
Q: What stereoisomers are obtained from the following reaction?
A: When alkene react with HBr that to in presence of peroxide then the substitution takes place…
Q: Draw the products of each reaction. Indicate the stereochemistry of Diels–Alder products.
A:
Q: 4. A. Draw the E2 elimination product of the following molecules and identify the resulting alkene…
A: E2 elimination take place in antiperplaner manner.
Q: 8.64 Reduction of butan-2-one with NaBH4 yields butan-2-ol. Explain why the product is chiral but…
A: The given reactant is Butan-2-one and the reagent is NaBH4. The given reagent is a reducing agent…
Q: Draw the product(s) of the following reactions. CH,CH, C=C-CH3 Na/NH3(1) H3C . Consider E/Z…
A: ->Na/NH3(l) is reducing agent which can reduce internal alkyne to trans alkene .It can not reduce…
Q: What is the product of the following [1,3] sigmatropic rearrangement, A or B?
A: In [1,3] sigmatropic rearrangement require antarafacial ring closure and migrating carbon undergoes…
Q: If the compounds shown here are heated, one will form one product from a [1,3] sigmatropic…
A: First compound undergoes only 1,3-methyl group migration. The reason for this is that H cannot…
Q: 4. Consider the following nucleophilic substitution: CH3 1) CH3O Na* CH3 HO, 2) H* COCH3 A a. Is the…
A: We are authorized to answer three subparts of a question at a time. Please post rest of the subparts…
Q: Draw the product of each of the following sigmatropic rearrangements:
A: Sigmatropic rearrangement is a type of intramolecular rearrangement in which the movement of a…
Q: е. Sn1 SN2 E1 E2 Initial Stereochemistry = R OH H,C-CH2 -CH3 + H3PO4 Product/Stereochemistry(ies) of…
A: The reaction is the elimination reaction
Draw the product of the following sigmatropic rearrangements:
![CH3
СНCH—CHCH—CH,
[5,5] sigmatropic
rearrangement
A](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2F5c0a156d-e84b-4904-9019-2076b1ce1d4e%2F9b03b1f8-33bf-4e05-ae2f-1d34d1dd07d3%2Flgcrb1.png&w=3840&q=75)
Given reaction:
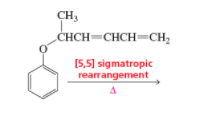
The rearrangement in which a sigma-bonded group or atom, flanked through one or more than one pie-electrons system shift to the new location which leads to the reorganization of pie-bonds is termed as sigmatropic reactions.
Step by step
Solved in 4 steps with 3 images

- 6 is the stereocenter S? MCAT prepHow many asymmetric centers does 2,4,6 tnmethy iheptane have4. For each of the following structures:a. Identify Chiral Centersb. Identify the chiral carbon configuration as R or Sc. Identify internal symmetry centers if they existd. Identify whether the structure is chiral or achirale. Identify mesostructures from existing
- Can you draw the chair conformation of trans 1 methyl 2 chlorocyclohexane1.The [CoCl4]2- anion is intensely blue colored and [CoCl6]4- is a lightly colored pink. explain breifly 2.Why is the method of descending symmetry needed to derive an mo diagram for acetylene (ethyne), but not needed for diazene((NH)2)?Label each molecule with chiral centers (R) and (S). Which ones have more than one?

