CH3. H3C, H3C" HH CH3 Ring-flip H3C H3C H- -CI A B (A) A=1.0 kJ/mol; B=11.4 kJ/mol; difference = 10.4 kJ/mol (B) A=2.0 kJ/mol; B=22.8 kJ/mol; difference = 20.8 kJ/mol (C) A=3.0 kJ/mol; B=22.8 kJ/mol; difference = 19.8 kJ/mol (D) A=1.0 kJ/mol; B=22.8 kJ/mol; difference = 21.8 kJ/mol %3D (E) A=2.0 kJ/mol; B=11.4 kJ/mol; difference = 9.4 kJ/mol
CH3. H3C, H3C" HH CH3 Ring-flip H3C H3C H- -CI A B (A) A=1.0 kJ/mol; B=11.4 kJ/mol; difference = 10.4 kJ/mol (B) A=2.0 kJ/mol; B=22.8 kJ/mol; difference = 20.8 kJ/mol (C) A=3.0 kJ/mol; B=22.8 kJ/mol; difference = 19.8 kJ/mol (D) A=1.0 kJ/mol; B=22.8 kJ/mol; difference = 21.8 kJ/mol %3D (E) A=2.0 kJ/mol; B=11.4 kJ/mol; difference = 9.4 kJ/mol
Chemistry for Engineering Students
4th Edition
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Lawrence S. Brown, Tom Holme
Chapter10: Entropy And The Second Law Of Thermodynamics
Section: Chapter Questions
Problem 10.59PAE: 10.59 The normal melting point of benzene, C6H6, is 5.5°C. For the process of melting solid benzene,...
Related questions
Question
Please answer this NEATLY, COMPLETELY, and CORRECTLY for an UPVOTE.
Long and detailed explanations are not needed. Keep it short, brief, and direct because I only need the answers as soon as possible.
Calculate the total strain energy in each of the two chair conformations of cis-1-tert-butyl-4-chlorocyclohexane. By how much energy is the more stable conformation favored (this is the difference in energy between the two conformations)?
NOTE: The strain energy for one H,Cl interaction is 1.0 kJ/mol while for H,-C(CH3)3 (t-butyl) is 11.4 kJ/mol.
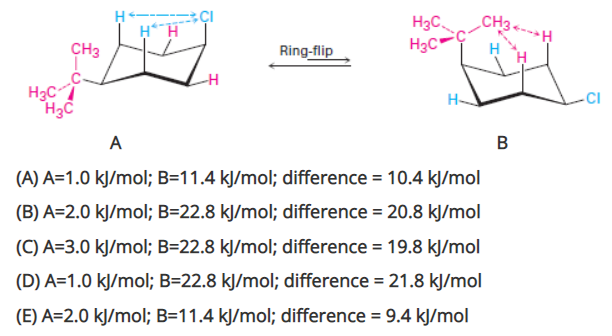
Transcribed Image Text:CH3.
H3C,
H3C"
HH
CH3
Ring-flip
H3C
H3C
H-
-CI
A
B
(A) A=1.0 kJ/mol; B=11.4 kJ/mol; difference = 10.4 kJ/mol
(B) A=2.0 kJ/mol; B=22.8 kJ/mol; difference = 20.8 kJ/mol
(C) A=3.0 kJ/mol; B=22.8 kJ/mol; difference = 19.8 kJ/mol
(D) A=1.0 kJ/mol; B=22.8 kJ/mol; difference = 21.8 kJ/mol
%3D
(E) A=2.0 kJ/mol; B=11.4 kJ/mol; difference = 9.4 kJ/mol
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning