CH4(g) gas reacts with F2(g) to produce CH3F(g) and HF(g). (a) A particulate representation of F2(g) is shown above its formula in the equation below. Using the key provided, draw particulate representations of CH4(g) and the two product molecules of the reaction above their formulas in the equation. CH4 F2 CH3F - HF Key = C H. II ||
CH4(g) gas reacts with F2(g) to produce CH3F(g) and HF(g). (a) A particulate representation of F2(g) is shown above its formula in the equation below. Using the key provided, draw particulate representations of CH4(g) and the two product molecules of the reaction above their formulas in the equation. CH4 F2 CH3F - HF Key = C H. II ||
Chemistry: An Atoms First Approach
2nd Edition
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Steven S. Zumdahl, Susan A. Zumdahl
Chapter5: Stoichiometry
Section: Chapter Questions
Problem 5RQ: Fig. 5-5 illustrates a schematic diagram of a combustion device used to analyze organic compounds....
Related questions
Question
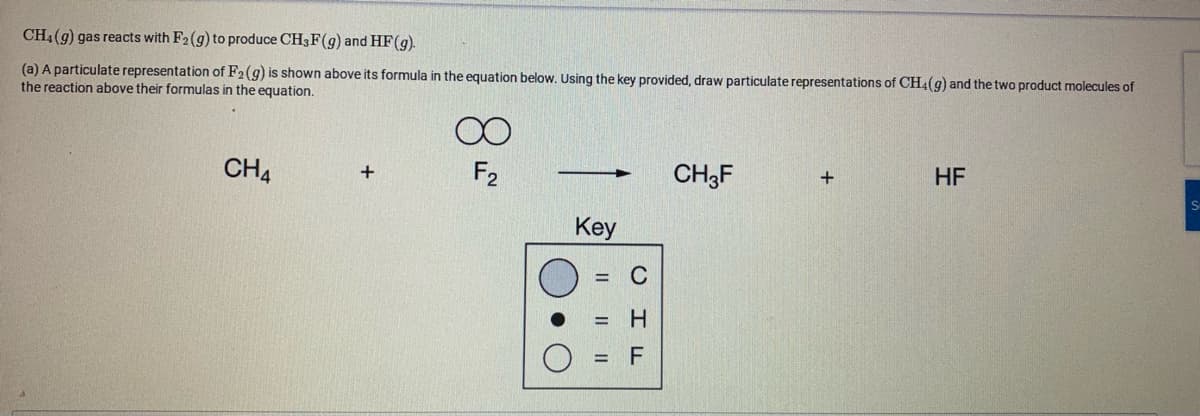
Transcribed Image Text:CH4 (g) gas reacts with F2(g) to produce CH3F(g) and HF(g).
(a) A particulate representation of F2(g) is shown above its formula in the equation below. Using the key provided, draw particulate representations of CH4(g) and the two product molecules of
the reaction above their formulas in the equation.
CH4
F2
CH3F
HF
Key
= C
= H
%3D
Expert Solution
Step 1
The reaction taking place is given as,

Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry for Engineering Students
Chemistry
ISBN:
9781285199023
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning