Calculating the composition of a buffer of a given pH A chemistry graduate student Is given 100. mL of a 0.7OM dlethylamine ((C,H) NH) solution. Dlethylamine is a weak base with K,=1.3× 10. What -3 9. mass of (C,H.) NH,Br should the student dissolve In the (C,H,) NH solutlon to turn it Into a buffer with pH =10.627 2 You may assume that the volume of the solutlon doesn't change when the (C,H,) NH,Br Is dissolved In It. Be sure your answer has a unit symbol, and round it to 2 significant digits. do Ar x10 Explanation Check 2021 McGraw-Hill Education. All Rights Reserved. Terms of Use Privacy Accessibility 10:54 hp esc backsc & #3 $ 8. 6. 3 4 1 i t y r
Calculating the composition of a buffer of a given pH A chemistry graduate student Is given 100. mL of a 0.7OM dlethylamine ((C,H) NH) solution. Dlethylamine is a weak base with K,=1.3× 10. What -3 9. mass of (C,H.) NH,Br should the student dissolve In the (C,H,) NH solutlon to turn it Into a buffer with pH =10.627 2 You may assume that the volume of the solutlon doesn't change when the (C,H,) NH,Br Is dissolved In It. Be sure your answer has a unit symbol, and round it to 2 significant digits. do Ar x10 Explanation Check 2021 McGraw-Hill Education. All Rights Reserved. Terms of Use Privacy Accessibility 10:54 hp esc backsc & #3 $ 8. 6. 3 4 1 i t y r
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter15: Additional Aqueous Equilibria
Section: Chapter Questions
Problem 34QRT
Related questions
Question
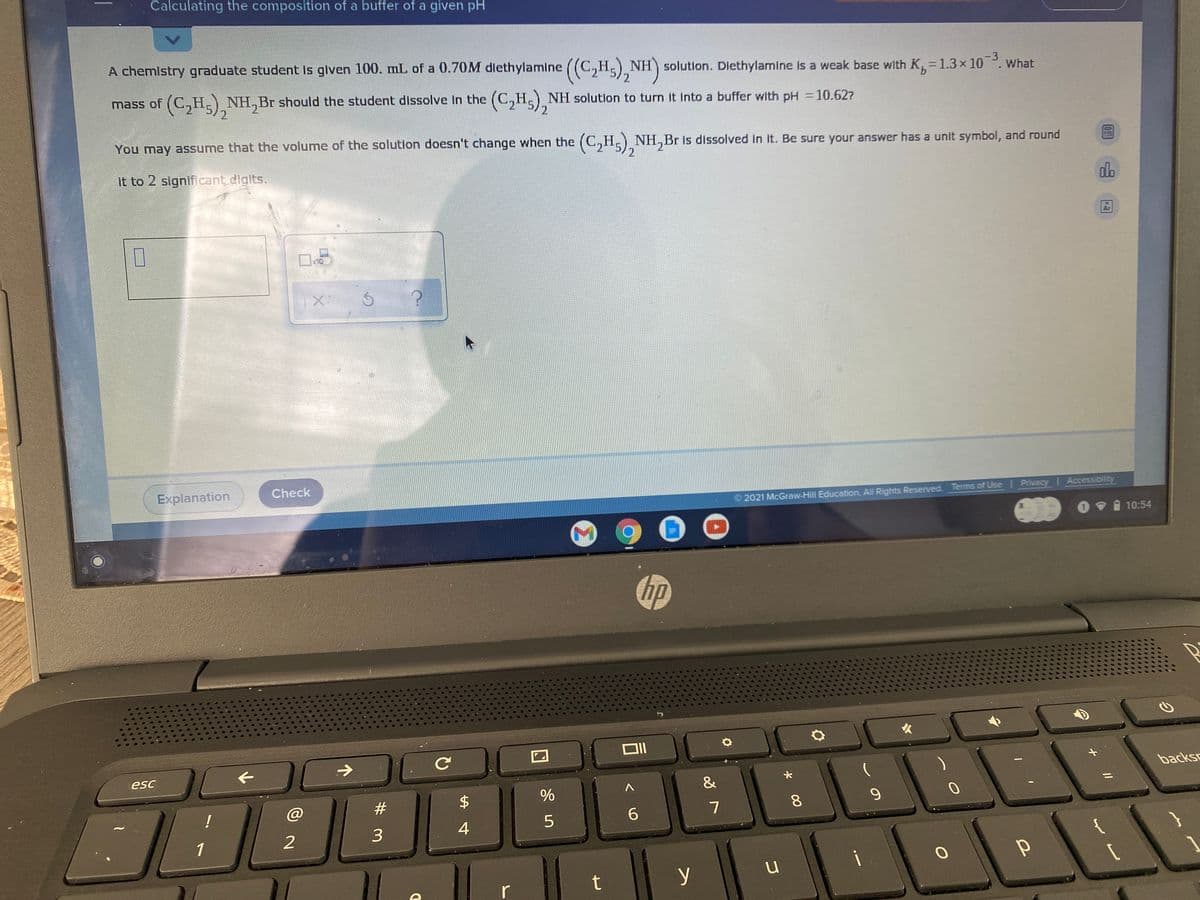
Transcribed Image Text:Calculating the composition of a buffer of a given pH
A chemistry graduate student Is given 100. mL of a 0.7OM dlethylamine
((C,H) NH) solution. Dlethylamine is a weak base with K,=1.3× 10. What
-3
9.
mass of (C,H.) NH,Br should the student dissolve In the (C,H,) NH solutlon to turn it Into a buffer with pH =10.627
2
You may assume that the volume of the solutlon doesn't change when the (C,H,) NH,Br Is dissolved In It. Be sure your answer has a unit symbol, and round
it to 2 significant digits.
do
Ar
x10
Explanation
Check
2021 McGraw-Hill Education. All Rights Reserved. Terms of Use Privacy Accessibility
10:54
hp
esc
backsc
&
#3
$
8.
6.
3
4
1
i
t
y
r
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning
