Question 31 Metals tend to electrons to form ions of charge. lose; negative lose; positive O gain; positive O gain; negative o gain; neutral Question 32 Which of the following clements is generally not found in the diatomic form in nature? O F Question 33 Which of these groups is incorrectly matched with its group/family name? Group Name 2A Alkaline earth metals ЗА Alkali metals III. 7A IV. 8A Halogens Noble gases V. 2B-3B Transition metals II o II Question 34 What are the Z and A values for an atom of krypton that contains 46 ncutrons? Z-36, A-46 Z-46, A-36 Z-36, A-82 Z-82, A-36 Z-82, A-46
Question 31 Metals tend to electrons to form ions of charge. lose; negative lose; positive O gain; positive O gain; negative o gain; neutral Question 32 Which of the following clements is generally not found in the diatomic form in nature? O F Question 33 Which of these groups is incorrectly matched with its group/family name? Group Name 2A Alkaline earth metals ЗА Alkali metals III. 7A IV. 8A Halogens Noble gases V. 2B-3B Transition metals II o II Question 34 What are the Z and A values for an atom of krypton that contains 46 ncutrons? Z-36, A-46 Z-46, A-36 Z-36, A-82 Z-82, A-36 Z-82, A-46
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter5: Resonance
Section: Chapter Questions
Problem 1E: Occasionally, we will see an ionic compound that has a 1 counterion. (Later we will find that 1...
Related questions
Question
Hi,
I am doing a practice test and need to check my answers. All of this is for Chem 101
Please see attached:
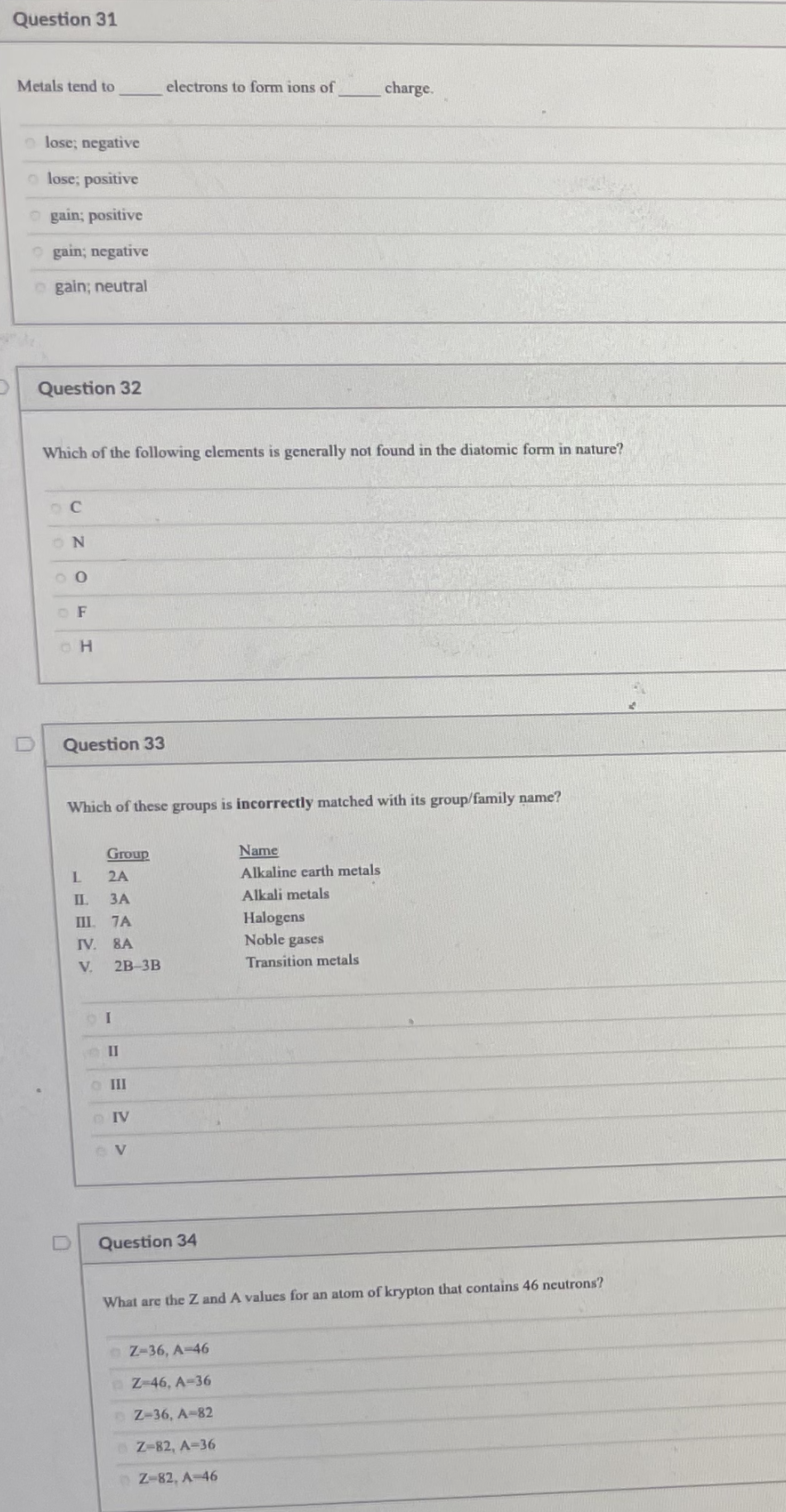
Transcribed Image Text:Question 31
Metals tend to
electrons to form ions of
charge.
lose; negative
lose; positive
O gain; positive
O gain; negative
o gain; neutral
Question 32
Which of the following clements is generally not found in the diatomic form in nature?
O F
Question 33
Which of these groups is incorrectly matched with its group/family name?
Group
Name
2A
Alkaline earth metals
ЗА
Alkali metals
III. 7A
IV. 8A
Halogens
Noble gases
V.
2B-3B
Transition metals
II
o II
Question 34
What are the Z and A values for an atom of krypton that contains 46 ncutrons?
Z-36, A-46
Z-46, A-36
Z-36, A-82
Z-82, A-36
Z-82, A-46
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax