Q: On the basis of the electronegativity values of the elements, what would each of the following bonds...
A: Given1. P-P2. N-F3 Mg-F Element Electronegativity Values P 2.19N ...
Q: Consider the following gas phase reaction. 4HCl(g) + O2 (g) --> 2Cl2 (g) + 2H2 O(g) What mass of ...
A:
Q: A cylinder containing ethanol is submitted to a certain pressure. At a temperature of 273 K, when th...
A: Colligative properties are dependent on the number of moles of solute in the solution. There are fou...
Q: Which of the following skeletal structures is/are WHONG as shown (no parial creditys A D A and Dare ...
A: A , C, and D are wrong
Q: Two methods convert an alkyl halide into a carboxylic acid having one more carbon atom. (1] R-X + "C...
A: The given compounds are,
Q: Directions: Show all your work and include units in your answer. Box your final answer. R= 0.0821 L ...
A: Given mass of Carbon mono sulfide = 12 gram Temperature ( T ) = 300 K Pressure( P ) ...
Q: 8) During ice ages the oxygen 18 to oxygen 16 ratio in the ocean will _____. A) decrease because g...
A: 8) The two isotopes of oxygen given are oxygen-18 (heavy) and oxygen-16 (light). The oceans have wat...
Q: A titration was carried out to determine the volume of base needed to neutralize an acid. Five runs ...
A: The neutralization reaction or the acid-base reaction is often employed as the basis for determining...
Q: Calculate the percentage of Fe in pure iron(II) ammonium hexahydrate: (NH4)2[Fe(SO4)2] x 6H2O
A: The solution is as follows :
Q: What is the the PH and POH of the following solutions: 1. 0.05 M Ba(NO3)2 2. 0.01 M Na2SO4
A: The given solutions are 0.05 M Ba(NO3)2 and 0.01 Na2SO4.
Q: At the end of two half-lives, what fraction of the atoms had not decayed?
A: Answer is given below.
Q: What is the wavelength (in nm) of light that has a change in energy of 1.79 × 10 18 J? O 111 nm O 87...
A: Given- Energy=1.79×10^-18J
Q: 16b.
A: Given that the reaction is exothermic. We will predict the given physical quantities as follows.
Q: Draw the Lewis dot structures of the following compounds. Cl2O H2CCH2 HCCCN SiO2
A: Since we know that a lewis structure is the structure which represent the bonding between the atoms ...
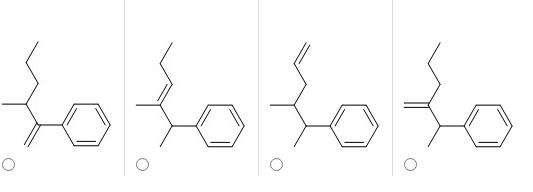
Q: Which of the following compounds would produce the set of mass spectra peaks shown below? All 3 mole...
A: The following Mass spectrum can be analyzed as follows:
Q: Use the changes of state interactive to answer the question. Identify the states of matter present a...
A: Phase change diagram of water is given As the heat is given to the system by increasing the temperat...
Q: Irite the balanced net ionic equation for H,SO4 reacting with Mg(OH)2. Remember to include phase lab...
A: Applying concept of ionic equation determination.
Q: Draw the structure of the following molecule, 4,4-dimethyl-1-decyne. Systematically name the follo...
A:
Q: Answer the following questions about benzphetamine, a habit-forming diet pill sold under the trade n...
A: (a)
Q: A voltaic cell is constructed with two hydrogen electrodes. Electrode 1 has PH2=1.00 atm and an unk...
A: Answer: As the current flows from electrode 1 to electrode 2, we can say that electrode 1 is the ano...
Q: A solution containing a linear molecule is dripped onto a dish of water. The molecule has one polar ...
A: Answer:- The solubility of liquid in liquid follows the like dissolves like rule i.e.,polar will dis...
Q: How many oxygen atoms are in 2.0 g of sucrose (C12H22O11)? O 3.9 × 10²² Oxygen atoms O 3.5 × 10²' Ox...
A:
Q: A sample of gas has a pressure of 1.2 atm and a volume of 2.3 L. What is the new pressure if the vol...
A: To solve this problem we will use Boyle's law According to this law P1V1 = P2V2 Where , P1 = ini...
Q: A fossil is found to have a 14C level of 73.0% compared to living organisms. How old is the fossil? ...
A: Given Remaining Amount of 14C = 73 % Let Initial Amount of 14C = 100% Age O...
Q: 14b
A:
Q: Find the concentration of all the ions in a 0.750M (NH4)3PO4
A:
Q: Consider the following reaction at 293 K Feaqy+ SCNaq)→ FeSCN (aq) Initially, 0.924 moles of FeNO3 a...
A: Given: The volume of the solution is 1 L. The number of moles of each Fe3+ and SCN- added to the sol...
Q: A gas sample is compressed at constant tem- perature. This means that the pressure must be 1. decrea...
A: On compressing at constant temperature there will be increase in Pressure because on compression vol...
Q: Will AgCl be more or less soluble in 0.2 M HCl compared to in pure water?
A: Silver chloride is a chemical compound with formula AgCl. The dissociation AgCl as follows --- AgCl ...
Q: ent/takeCovalentActivity.do?locator=assignment-take [Review Topics) [References] Use the References ...
A:
Q: The density of bromine is 3.1g/cm^3. What is the mass of 17.20cm^3 of bromine with the correct numbe...
A:
Q: Which of the following choices is correct about the reaction shown? H,C. CH CHCI, кон Product(s)? CH...
A:
Q: Devise a synthesis of 1-methylcyclohexene from 1-bromocyclohexene and CH3I. -CH3 + CHẠI Br 1-methylc...
A: Given reaction, 1-methylcyclohexene from 1-bromocyclohexene and methyl iodide
Q: Kindly answer this and choose the correct option. Thank you.
A: Step 1 The general chemical equation is given as; Equilibrium expression is given by K, which is al...
Q: Decide whether each molecule or polyatomic ion is polar or nonpolar. If the molecule or polyatomic i...
A: Regular tetrahedral, regular octahedral, symmetrical linear molecules are non-polar
Q: Chemistry Question
A: The strongest acid is lemon juice (PH=2.23) of the following solutions tested above. ...
Q: What starting materials are needed to prepare ar-turmerone using a directed aldol reaction? ar-Turme...
A: The reaction of propan-2-one with 4-p-tolylpentan-2-one in the presence of OH- results in the format...
Q: QUESTION 2 Which of the following reactions will not take place? OA (CH3CH2CH2CO)20 + CI" --> CH3CH2...
A: In all the given reactions, examples of acid derivatives formation take place. There are four acid d...
Q: Pls explain why?
A: The correct answer with mechanism is explained below.
Q: 37. Which of the following compounds will exhibit ionic bonding? (Circle all that apply) Nao Na,SO, ...
A: Compounds formed from elements with large difference in electronegativity are ionic
Q: Kindly answer this. Thank you.
A: Given that: t1/2 = 8.0 min To find: the value of rate constant k?
Q: When a Lewis acid and a Lewis base combine, the product may be referred to as an acid-base
A: Lewis Acid Base theory is based on electrons. According to Lewis, Acids are electron acceptors and b...
Q: What is the pressure in a gas container that is connected to an open-end U-tube manometer, if the pr...
A: The physical force exerted on an object is known as pressure. The force exerted per unit area is per...
Q: Help with redox questions.
A:
Q: Solvent 12cm front 10cm 8cm 6cm 4cm 2cm If the stationary phase is non-polar and the mobile phase is...
A: Solution : In chromatography, the stationary phase is a paper used for the process while the mobile ...
Q: Identify whether each of the following solutions is acidic, basic, or neutral. Drag the appropriate ...
A: pH is defined as the negative logarithm to the base 10 of hydrogen ion concentration expressed in mo...
Q: What is the energy per photon of light with a wavelength of 426nm?
A: Given Wavelength of light of photon = 426 nmEnergy per photon of light = to be determined = E
Q: Rank the following molecules by order of increasing bond energy: F2, F2^+, F2^2+. Justify your answe...
A: Answer:- This question is answered by using the simple concept of bond order. Higher the bond order ...
Q: Briefly explain why it is necessary to recrystallize during a synthesis.
A: Recrystallization is the method during which the matter is separated from its impurities by dissolvi...
Q: Based on their structures and molecular weights, which of the four Claisen condensation products is ...
A: In Claisen condensation beta-keto ester is formed while in aldol condensation, the beta-hydroxy carb...
which is
Step by step
Solved in 2 steps with 1 images

- 1.76 How does Gorilla Glass differ from more commonly found alumina silicate glass?1.83 What type of transition is represented in the following molecular-scale illustration?Which of the following techniques does not allow recovery of all components? a. distillation b. filtration c. evaporation d. chromatography



