Q: 5) Draw a clear mechanism for the following reaction that accounts for the formation of ALL…
A:
Q: Every transformation can be completed in one 'synthetic ste added sequentially, ex. "1. BH3, THF; 2.…
A: Given : We have to write the reagent for the following.
Q: 10. Draw a reasonable mechanism for the following reaction (cat. = catalytic %3D amount). OH H2SO4…
A: Rearrangement of carbocations.
Q: b) Qax HO
A:
Q: 2. Soclz 3. cexcess) TSOH (cat),
A:
Q: Consider the intramolecular nucleophilic substitution reaction shown here. Does the stereochemistry…
A: The given intramolecular nucleophilic substitution reaction is,
Q: 3) Draw a plausible mechanism for the following transformation: 0. 0. H³0Ⓡ NO₂
A: In this question we have to tell the product of the reaction and also its mechanism.
Q: 6. A synthesis of a molecule being explored as an antidepressant drug (J. Org. Chem 2012) utilized…
A: AlCl3---> It is a lewis acid. it accepts the electron. CH2=CH2 (Ethene): It acts as a nucleophile…
Q: 5. Propose a synthetic pathway for molecule X to molecule Y using the reagents boxed below. Note…
A: We have to propose a synthetic pathway for molecule X to molecule Y using the reagents boxed below…
Q: Draw the major E2 elimination product from the following alkyl halide. CH(CH2 HO CH, a draw…
A:
Q: Question 6. Provide a mechanism for the following transformation. NaBH4 -Me Br. Me
A: In the given reaction reduction is take place. There are two steps in mechanism.
Q: Propose a plausible mechanism for the following transformation. NaH CI HO 13.38e This substitution…
A: Organic reaction mechanisms
Q: H* Consider the following reaction: R- H- H,0 R Draw a complete mechanism for the reaction, showing…
A: a) The reaction given is,
Q: 2.Provide the most probable reagent(s) to accomplish the following reaction: HO
A: According to Bartleby guidelines we can answer only one question, So i have solved the first one and…
Q: H2NNH2 'NH R-N N-R NH cat. H+
A:
Q: 5. Draw a detailed arrow pushing mechanism for the following reaction. Is the rate-determining step…
A: When No carbocation is formed then reaction is SN2.if carbocation is formed then reaction is SN1
Q: 6. Determine whether each reaction occurs by an Sn2, Sn1, E1, and/or E2 reaction. Provide a detailed…
A: Hello. There are multiple subparts to this question. The first three subparts have been solved. To…
Q: HO Provide a mechanism for the transformation below. HO H,O/ CH;OH :0:
A: A reaction mechanism is the order of basic steps by which a chemical reaction occurs. A reaction…
Q: 1. Draw the mechanism and major product of the synthesis reaction bel Br E2
A:
Q: 5. Propose a synthetic pathway for molecule X to molecule Y using the reagents boxed below. Note…
A:
Q: 4. Propose a mechanism for the SUBSTITUTION reaction below: HINT AICI, is an ELECTROPHILE. H3C ÇH3…
A: The given reaction is a Friedel - Crafts alkylation reaction. In this reaction alkylation of…
Q: Enio bns 200 HO HO ONLY
A:
Q: 8A. Draw a reasonable mechanism for the following reaction, with clear indication of…
A:
Q: Please provide a in-detail mechanism for reaction below. Note: Must show all the steps and correct…
A:
Q: HCI H20 +
A:
Q: ) ezs Memyes 2) Hb O excess
A:
Q: This reaction is an example of conjugate addition of a nucleophile to an a.B-unsaturated carbonyl.…
A: conjugate 1,4 addition is called Michael addition
Q: (a) PHOOC-CH,-C-CH,-COOP. (1) KCN, A (2) H,0® (o) СООН Onl
A:
Q: 6. Determine whether each reaction occurs by an SN2, SN1, E1, and/or E2 reaction. Provide a detailed…
A: → In above base is weak so it show SN2 reaction.
Q: 6. A synthesis of a molecule being explored as an antidepressant drug (J. Org. Chem 2012) utilized…
A:
Q: но H2SO4,Heat
A:
Q: Draw all possible E1 mechanisms and products involving this alkyl halide and water. Br H2O ?
A: The given alkyl halide is,
Q: 10. Provide a mechanism for the following transformation: soc, OH
A:
Q: 38) Complete the following reactions with the proper major products. SO3/H₂SO4 Br2 FeBr3 MeCOCI…
A: Step 1 : Fridel craft acylation. Step 2 : Ortho isomer ( Sulphonation takes place at para position…
Q: 3. Draw the mechanism for each reaction below; HO H, H CH,CH CH,CH,NH, HyC H. H (pH = 5) H. 1. Ph,P…
A: Note: As per company policy we are supposed to attempt first three subparts only. Please repost…
Q: 3. Explain, by means of a detail mechanism for each, why the two reactions give the same product.…
A: ->In presence of base elimination reaction occur and alkene formed.it removed antiperiplanar…
Q: 4) Provide a mechanism that explains the formation of B and C when compound A is treated with NaOMe.…
A:
Q: Rank the substrates below in order of increasing reactivity (from slowest to fastest) in a…
A: Considering SN2 mechanism and leaving ability of leaving group.
Q: ö: Br: H,0 heat ОН DH
A:
Q: 4. Propose a mechanism for the reaction below. ( HS SH cat. H
A: Reaction- In analogy for the formation of acetals from alcohols and acid, treatment of…
Q: 3. Provide full mechanism (curved arrows and intermediates) for the following reaction. OCH3 1.…
A: Answer 3 ) The mechanism for the given reaction = ? Note : As per our company guidelines, we are…
Q: Me „CHO CO2E Me + Ph3Ps .CO2E† Me CH2CI2 Me 0: Me Me 40 °C, 60 min, 89 % (3 steps) Me See the Wittig…
A: Wittig reaction is an organic chemical reaction wherein an aldehyde or a ketone is reacted with a…
Q: Which reaction proceed according to an E2 mechanism? toe. 3D II ethanol heat Br heat Br DMSO towe.…
A: E2 elimination generally occurs in the presence of heat, complete in one step and form alkene. As…
Q: OTS NaOH ? A Ph. J dilute NaOH D OTS ? B NaNH, ? E NaOH ? NaOH
A: The elimination reaction will depend on the stability of the ion formed during the reaction. Higher…
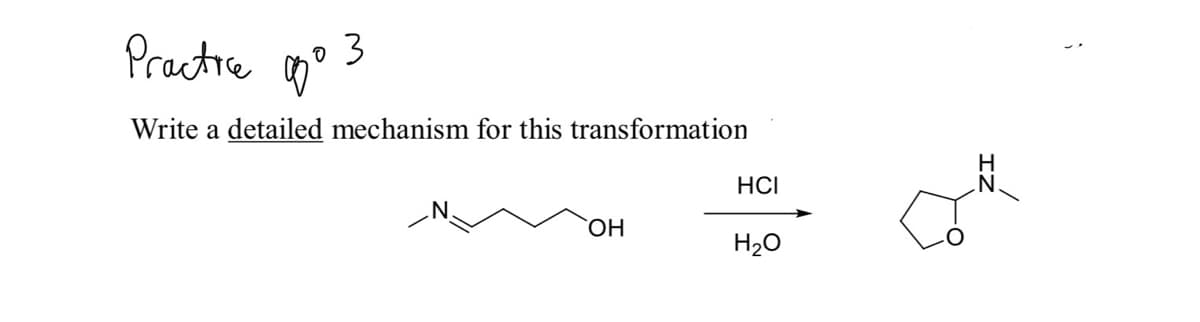
Q: nothe Write a detailed mechanism for this transformation но. HCI HO H20 H
A: The given reaction is an hydrolysis of ether
Q: Please explain the mechanism of the following reaction and why these products are formed in equal…
A:
Q: 4. Provide an arrow pushing mechanism for the following transformation and clearly circle the…
A: Given that : We have to provide an arrow pushing mechanism for the given below transformation and…
Q: What is the major product of the reaction below? Draw a detailed plausible mechanism and all…
A: Here we have to write the major product in the following Diel's alder reaction with correct…
Q: 3) Write a detailed mechanism of: CH. CH HNOT HYSO! "NO.
A: Given is the reaction between Toluene in presence of nitric acid and sulfuric acid.
Q: Provide a complete mechanism for the reaction, including all lone pairs, formal charges, and curved…
A:
Step by step
Solved in 2 steps with 1 images

- Plastic photochromic sunglasses are based on the following reversible rearrangement of a dye inside the lenses that occurs when the lenses are exposed to sunlight. The original dye absorbs UV light but not visible light and is thus colorless, while the rearrangement product absorbs visible light and is thus darkened. (a) Show the mechanism of the rearrangement. (b) Why does the rearrangement product absorb at a longer wavelength (visible light) than the original dye (UV)?Draw the mechanism ffrom benzaldehyde to this using: i)NaBH4 ii)TsCl, py iii)NaCN iiii)H+, H2OProvide the mechanism for the conversion shown below. CH3Li is effectively H3C-Li+. What type of mechanism is occuring?
- What type of mechanism is exhibited in the reaction? SN1, SN2, E2, E1?If bromobenzene is treated with HNO3/H2SO4 the major product should be 4-nitrobromobenzene. Please show the complete mechanism for the reaction and include any intermediates. Thanks!Don't provied handwriting solution. 0Can you please explain the reasonin behind why we split the molecule the way we do to obtain the desired product in addition to showing the mechanism? Thank you!



