Question 11 A(n) is an explanation of a scientific observation. O speculation o theory O conclusion O prediction o epiphany Question 12 Which two subatomic particles comprise the nucleus of an atom? Protons, neutrons O Protons, electrons O Electrons, neutrons O Alpha particles, protons OAlpha particles, electrons Question 13 A statement that summarizes the data obtained from a series of obseryations is known as a(n) o observation o law o theory o conclusion O experiment Question 14 Determine the density of a 100.0 g sample of mercury having a volume of 7.36 cm. Carry out the calculation to the correct number of significant digits. O 13.58 g/cm 3 O 13.66 g/cm o 183 g/cm 0.0732 g/cm O 0.073 g/cm
Question 11 A(n) is an explanation of a scientific observation. O speculation o theory O conclusion O prediction o epiphany Question 12 Which two subatomic particles comprise the nucleus of an atom? Protons, neutrons O Protons, electrons O Electrons, neutrons O Alpha particles, protons OAlpha particles, electrons Question 13 A statement that summarizes the data obtained from a series of obseryations is known as a(n) o observation o law o theory o conclusion O experiment Question 14 Determine the density of a 100.0 g sample of mercury having a volume of 7.36 cm. Carry out the calculation to the correct number of significant digits. O 13.58 g/cm 3 O 13.66 g/cm o 183 g/cm 0.0732 g/cm O 0.073 g/cm
Introductory Chemistry: An Active Learning Approach
6th Edition
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Mark S. Cracolice, Ed Peters
Chapter5: Atomic Theory : The Nuclear Model Of The Atom
Section: Chapter Questions
Problem 51E
Related questions
Question
Hi,
I am doing a practice test and need to check my answers. All of this is for Chem 101
Please see attached:
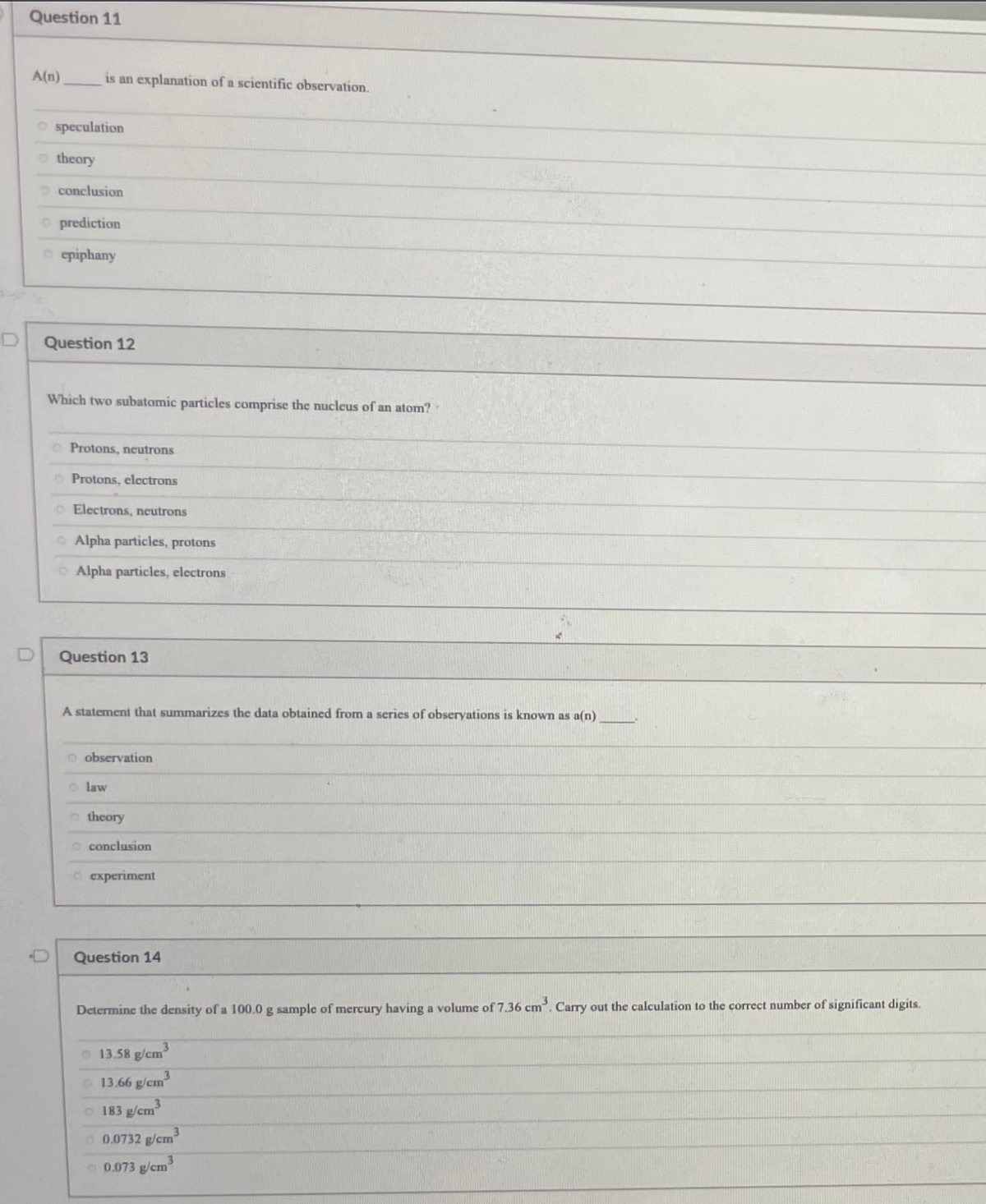
Transcribed Image Text:Question 11
A(n)
is an explanation of a scientific observation.
O speculation
o theory
O conclusion
O prediction
o epiphany
Question 12
Which two subatomic particles comprise the nucleus of an atom?
Protons, neutrons
O Protons, electrons
O Electrons, neutrons
O Alpha particles, protons
OAlpha particles, electrons
Question 13
A statement that summarizes the data obtained from a series of obseryations is known as a(n)
o observation
o law
o theory
o conclusion
O experiment
Question 14
Determine the density of a 100.0 g sample of mercury having a volume of 7.36 cm. Carry out the calculation to the correct number of significant digits.
O 13.58 g/cm
3
O 13.66 g/cm
o 183 g/cm
0.0732 g/cm
O 0.073 g/cm
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER


Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:
9781559539418
Author:
Angelica Stacy
Publisher:
MAC HIGHER


Chemistry: Matter and Change
Chemistry
ISBN:
9780078746376
Author:
Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:
Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning