9:57 Done 6 of 6 ****.. Insert Table Chart Text Shape Media Comment Collaborate beaker? Statistical Analysis of Laboratory Result: accuracy and precision O Shantel was exploring the different types of glassware in the chemistry lab, and wanted to compare how well a graduated cylinder delivered 5.00 mL of water. The below data was collected by weighing an empty beaker, zeroing the balance, and weighing the water delivered. The temperature of the water is 20.4°C. The density of water at this temperature is 0.998120 g/ mL. Determine the average, % error, standard deviation and CV for the graduated cylinder. You may do the calculations in excel if you wish. Report your answers to the appropriate number of significant figures. Trial 1: 4.9821g Trial 2: 4.9745 g Trial 3: 5.0112 g Trial 4: 5.0041 g MacBook Air 80 F3 DII DD F4 F5 F6 F7 F8 F9 F10 F1 %24 &
9:57 Done 6 of 6 ****.. Insert Table Chart Text Shape Media Comment Collaborate beaker? Statistical Analysis of Laboratory Result: accuracy and precision O Shantel was exploring the different types of glassware in the chemistry lab, and wanted to compare how well a graduated cylinder delivered 5.00 mL of water. The below data was collected by weighing an empty beaker, zeroing the balance, and weighing the water delivered. The temperature of the water is 20.4°C. The density of water at this temperature is 0.998120 g/ mL. Determine the average, % error, standard deviation and CV for the graduated cylinder. You may do the calculations in excel if you wish. Report your answers to the appropriate number of significant figures. Trial 1: 4.9821g Trial 2: 4.9745 g Trial 3: 5.0112 g Trial 4: 5.0041 g MacBook Air 80 F3 DII DD F4 F5 F6 F7 F8 F9 F10 F1 %24 &
Chapter2: Basic Statistical Analysis With Excel
Section: Chapter Questions
Problem 12P
Related questions
Question
Transcribed Image Text:9:57
Done
6 of 6
****..
Insert
Table
Chart
Text
Shape
Media
Comment
Collaborate
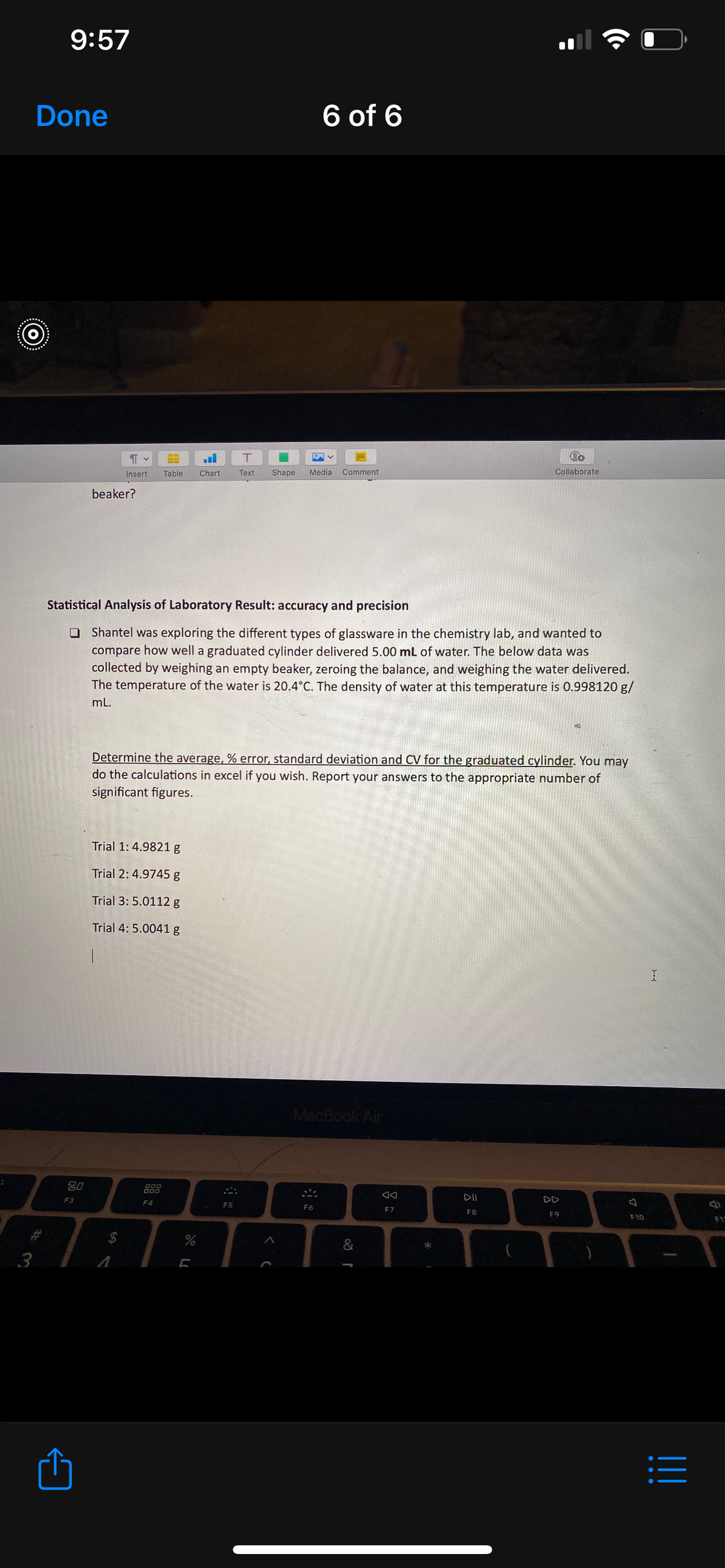
beaker?
Statistical Analysis of Laboratory Result: accuracy and precision
O Shantel was exploring the different types of glassware in the chemistry lab, and wanted to
compare how well a graduated cylinder delivered 5.00 mL of water. The below data was
collected by weighing an empty beaker, zeroing the balance, and weighing the water delivered.
The temperature of the water is 20.4°C. The density of water at this temperature is 0.998120 g/
mL.
Determine the average, % error, standard deviation and CV for the graduated cylinder. You may
do the calculations in excel if you wish. Report your answers to the appropriate number of
significant figures.
Trial 1: 4.9821g
Trial 2: 4.9745 g
Trial 3: 5.0112 g
Trial 4: 5.0041 g
MacBook Air
80
F3
DII
DD
F4
F5
F6
F7
F8
F9
F10
F1
%24
&
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps with 1 images

Recommended textbooks for you


Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,



Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,



Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning