1. Given the following reactions and data, use addition (similar to Hess's Law) to calculate AG° for H3PO4 (s). (Hint: The overall reaction is for the formation of 1 mol of H3PO4 (s) from elements in their natural state (elemental phosphorus is P4 (s)). PA () + 5 02@ → P,O10 (5) AG; - 2698 kJ P,O10 (9) + 6 H0 m → 4 H3P04 () H2 ( + ½ 02 (2) → H;O @ AG° = – 396 kJ - 228.6 kJ AG; : AG vap H,O 0 → H2O @ 8.6 kJ Required work: * rewrite the reactions above in the correct direction, using multipliers to change coefficients and modify AG° values if necessary * show that after cancelling, the sum of your reactions gives the desired overall reaction Write the goal equation first?
1. Given the following reactions and data, use addition (similar to Hess's Law) to calculate AG° for H3PO4 (s). (Hint: The overall reaction is for the formation of 1 mol of H3PO4 (s) from elements in their natural state (elemental phosphorus is P4 (s)). PA () + 5 02@ → P,O10 (5) AG; - 2698 kJ P,O10 (9) + 6 H0 m → 4 H3P04 () H2 ( + ½ 02 (2) → H;O @ AG° = – 396 kJ - 228.6 kJ AG; : AG vap H,O 0 → H2O @ 8.6 kJ Required work: * rewrite the reactions above in the correct direction, using multipliers to change coefficients and modify AG° values if necessary * show that after cancelling, the sum of your reactions gives the desired overall reaction Write the goal equation first?
General Chemistry - Standalone book (MindTap Course List)
11th Edition
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Chapter6: Thermochemisty
Section: Chapter Questions
Problem 6.132QP
Related questions
Question
100%
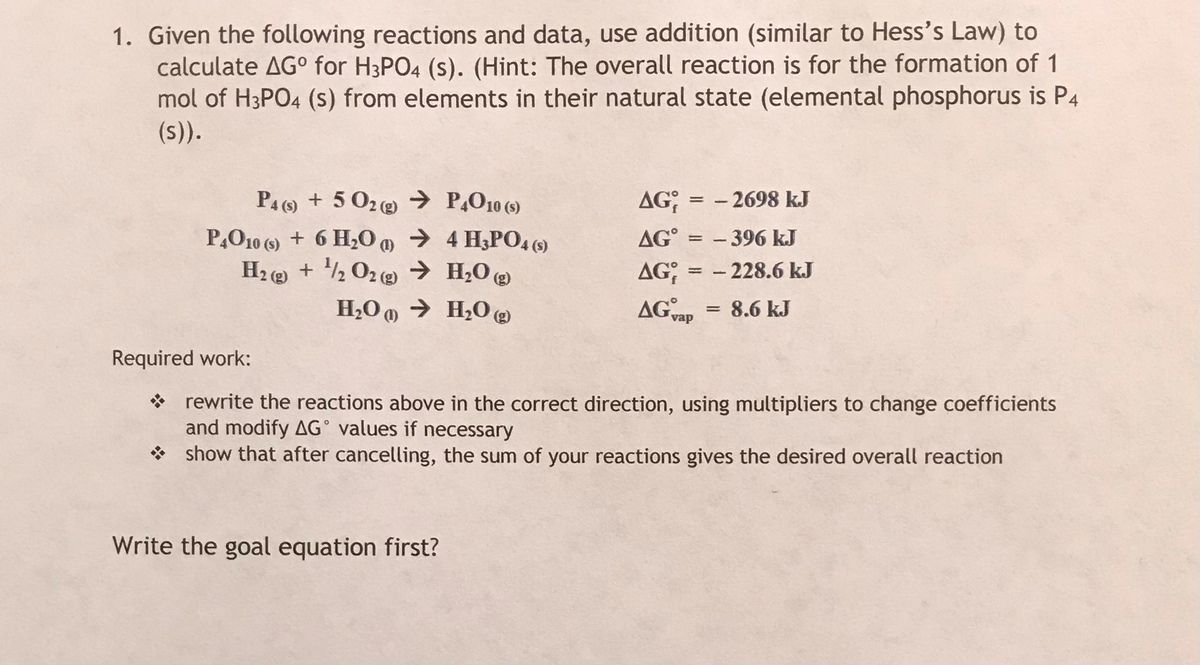
Transcribed Image Text:1. Given the following reactions and data, use addition (similar to Hess's Law) to
calculate AG° for H3PO4 (s). (Hint: The overall reaction is for the formation of 1
mol of H3PO4 (s) from elements in their natural state (elemental phosphorus is P4
(s)).
PA () + 5 02@ → P,O10 (5)
AG;
- 2698 kJ
P,O10 (9) + 6 H0 m → 4 H3P04 ()
H2 ( + ½ 02 (2) → H;O @
AG° = – 396 kJ
- 228.6 kJ
AG; :
AG vap
H,O 0 → H2O @
8.6 kJ
Required work:
* rewrite the reactions above in the correct direction, using multipliers to change coefficients
and modify AG° values if necessary
* show that after cancelling, the sum of your reactions gives the desired overall reaction
Write the goal equation first?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

General Chemistry - Standalone book (MindTap Cour…
Chemistry
ISBN:
9781305580343
Author:
Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning