9:5xtrac) f Three nass of sap red) f fr sample of samplsy DURE lued 30 nmyer rer fi IM Na Id the St sapor gas 3. fraction of species Below is the speciation diagram for 2-naphthol. Figure 2. Speciation diagram for 2-naphthol 0.8 0.6 0.4 0.2 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 Hd The pH of a 1 M NaHCO3 solution is 105. What is dominant at this pH, 2-naphthol (the conjugate acid) or 2-naphthoxide (its conjugate base)? 7. The pH of a 3 M NAOH solution is greater than 14. What is dominant at this pH, 2-naphthol (the conjugate acid) or 2-naphthoxide (its conjugate base)? 8. Use your answers to questions 3 to 7 to explain how adding 1 M NaHCO3 to benzoic acid and 2- naphthol allows you to separate these compounds. of Sam mussi ared) of S DURE lued por 2. fraction of species *. Kecan that deprotonation is required to form a water-soluble salt and thus achieve an extraction. The raction of the acid and its conjugate base at different pH values can be calculated using the Ka. This information can be summarized on graph called a speciation diagram. This is a plot of the fraction of different species (compounds or ions) vs. pH. A fraction of 0 indicates the substance is not present, and a fraction of 1 indicates that is the only substance present. The diagram for benzoic acid is below. Figure 1. Speciation diagram for benzoic acid 0.8 0.6 0.4 0.2 y-ö: 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 Hd The pH of a 1 M NAHCO3 solution is 10.5. What is dominant at this pH, benzoic acid (the conjugate acid) or benzoate (its conjugate base)? 5. The pH of a 3 M NaOH solution is greater than 14. What is dominant at this pH, benzoic acid (the conjugate acid) or benzoate (its conjugate base)?
9:5xtrac) f Three nass of sap red) f fr sample of samplsy DURE lued 30 nmyer rer fi IM Na Id the St sapor gas 3. fraction of species Below is the speciation diagram for 2-naphthol. Figure 2. Speciation diagram for 2-naphthol 0.8 0.6 0.4 0.2 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 Hd The pH of a 1 M NaHCO3 solution is 105. What is dominant at this pH, 2-naphthol (the conjugate acid) or 2-naphthoxide (its conjugate base)? 7. The pH of a 3 M NAOH solution is greater than 14. What is dominant at this pH, 2-naphthol (the conjugate acid) or 2-naphthoxide (its conjugate base)? 8. Use your answers to questions 3 to 7 to explain how adding 1 M NaHCO3 to benzoic acid and 2- naphthol allows you to separate these compounds. of Sam mussi ared) of S DURE lued por 2. fraction of species *. Kecan that deprotonation is required to form a water-soluble salt and thus achieve an extraction. The raction of the acid and its conjugate base at different pH values can be calculated using the Ka. This information can be summarized on graph called a speciation diagram. This is a plot of the fraction of different species (compounds or ions) vs. pH. A fraction of 0 indicates the substance is not present, and a fraction of 1 indicates that is the only substance present. The diagram for benzoic acid is below. Figure 1. Speciation diagram for benzoic acid 0.8 0.6 0.4 0.2 y-ö: 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 Hd The pH of a 1 M NAHCO3 solution is 10.5. What is dominant at this pH, benzoic acid (the conjugate acid) or benzoate (its conjugate base)? 5. The pH of a 3 M NaOH solution is greater than 14. What is dominant at this pH, benzoic acid (the conjugate acid) or benzoate (its conjugate base)?
Chemical Principles in the Laboratory
11th Edition
ISBN:9781305264434
Author:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Chapter37: Qualitative Analysis Of Group Ii Cations
Section: Chapter Questions
Problem 3ASA
Related questions
Question
Please help!
Transcribed Image Text:9:5xtrac)
f Three
nass of sap
red) f fr
sample
of samplsy
DURE
lued
30
nmyer
rer fi
IM
Na
Id the St
sapor gas
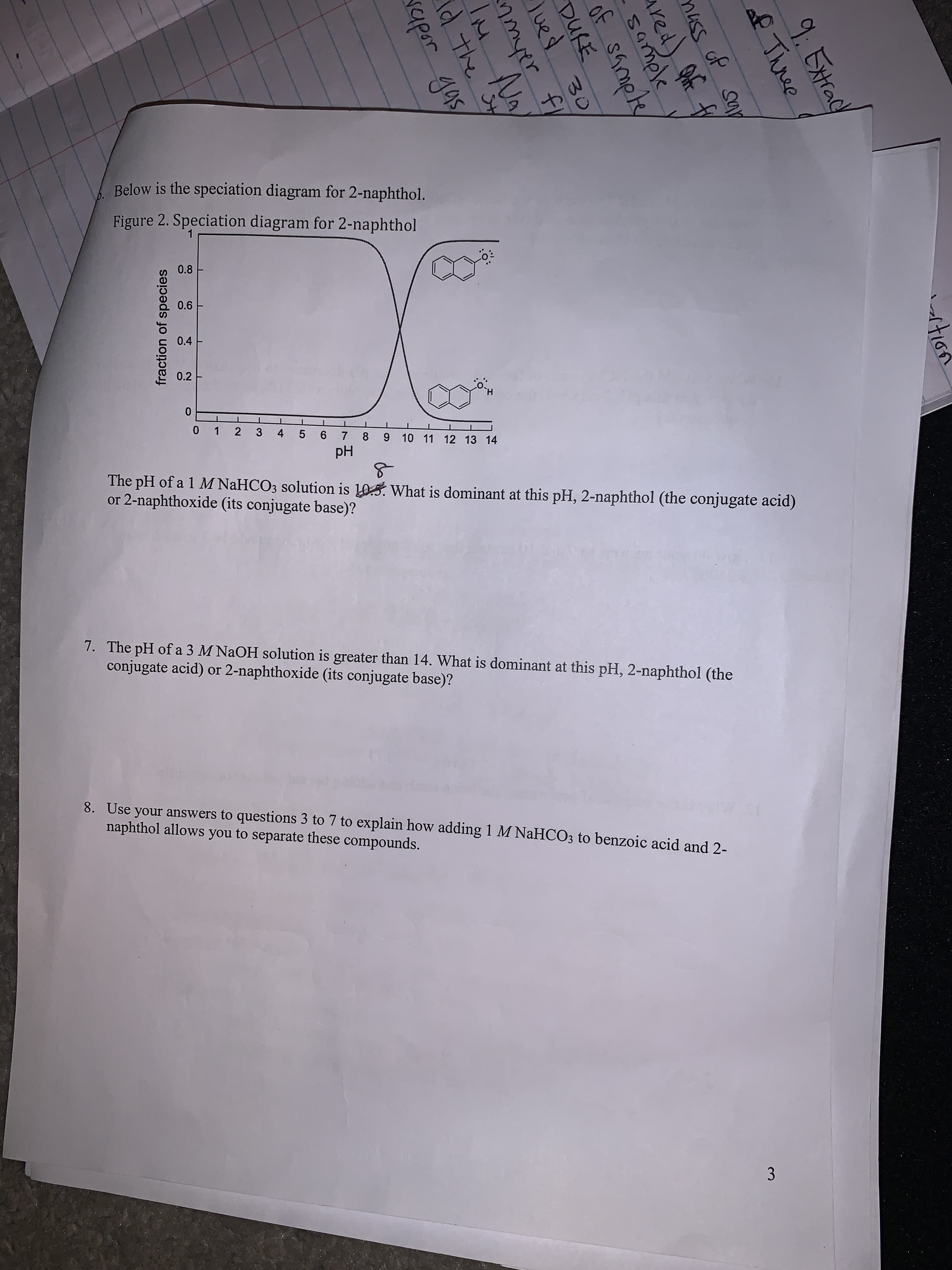
3.
fraction of species
Below is the speciation diagram for 2-naphthol.
Figure 2. Speciation diagram for 2-naphthol
0.8
0.6
0.4
0.2
0 1 2 3 4 5 6 7 8 9 10 11 12 13 14
Hd
The pH of a 1 M NaHCO3 solution is 105. What is dominant at this pH, 2-naphthol (the conjugate acid)
or 2-naphthoxide (its conjugate base)?
7. The pH of a 3 M NAOH solution is greater than 14. What is dominant at this pH, 2-naphthol (the
conjugate acid) or 2-naphthoxide (its conjugate base)?
8. Use your answers to questions 3 to 7 to explain how adding 1 M NaHCO3 to benzoic acid and 2-
naphthol allows you to separate these compounds.
Transcribed Image Text:of Sam
mussi
ared)
of S
DURE
lued
por
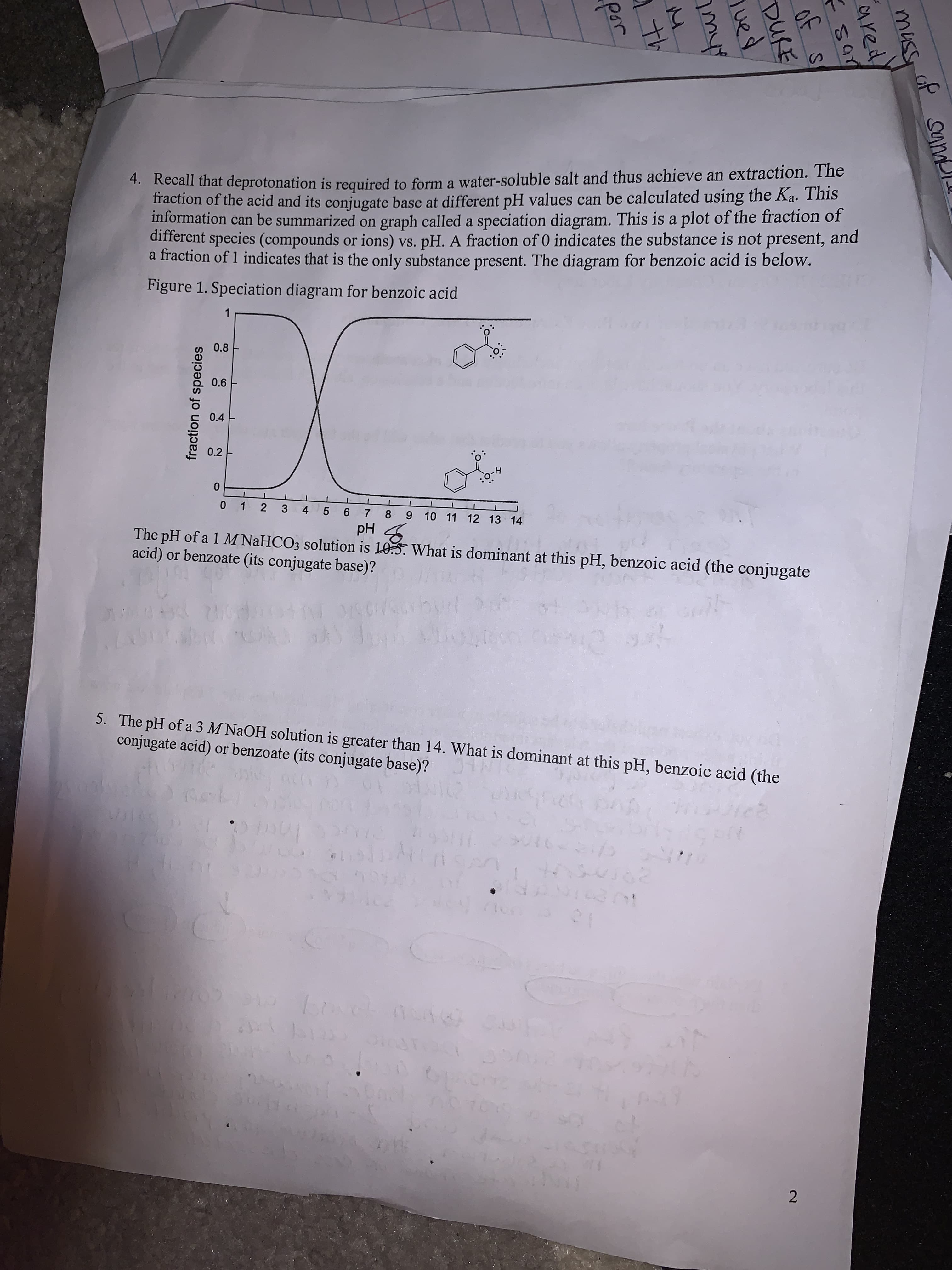
2.
fraction of species
*. Kecan that deprotonation is required to form a water-soluble salt and thus achieve an extraction. The
raction of the acid and its conjugate base at different pH values can be calculated using the Ka. This
information can be summarized on graph called a speciation diagram. This is a plot of the fraction of
different species (compounds or ions) vs. pH. A fraction of 0 indicates the substance is not present, and
a fraction of 1 indicates that is the only substance present. The diagram for benzoic acid is below.
Figure 1. Speciation diagram for benzoic acid
0.8
0.6
0.4
0.2
y-ö:
0 1 2
3 4 5 6 7 8 9 10 11 12 13 14
Hd
The pH of a 1 M NAHCO3 solution is 10.5. What is dominant at this pH, benzoic acid (the conjugate
acid) or benzoate (its conjugate base)?
5. The pH of a 3 M NaOH solution is greater than 14. What is dominant at this pH, benzoic acid (the
conjugate acid) or benzoate (its conjugate base)?
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole



Chemical Principles in the Laboratory
Chemistry
ISBN:
9781305264434
Author:
Emil Slowinski, Wayne C. Wolsey, Robert Rossi
Publisher:
Brooks Cole

