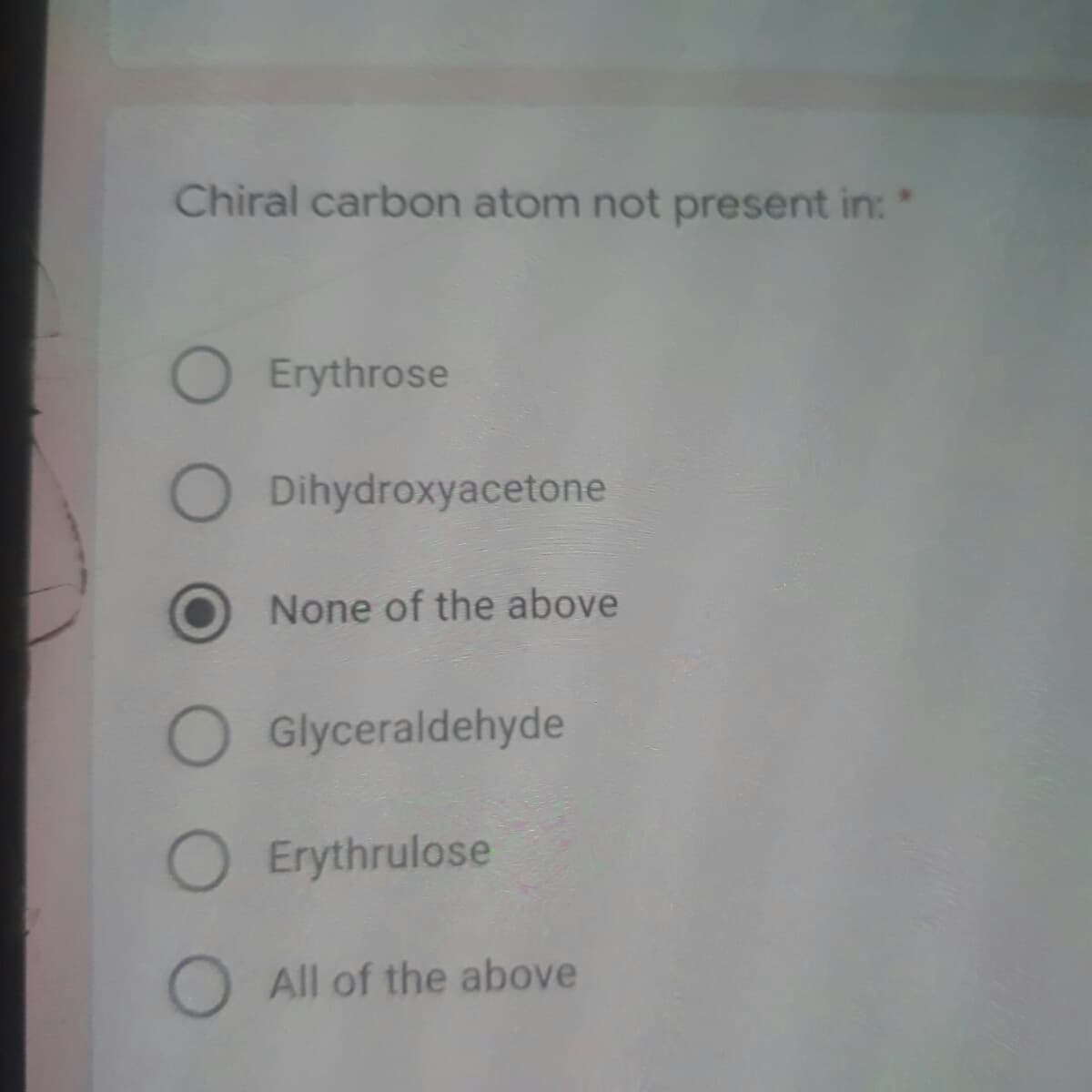
Chiral carbon atom not present in: * Erythrose Dihydroxyacetone
Q: Thiophene ring contains ------- as heteroatom.
A: Thiophene is a compound with ring structure, in which there will be atoms of two or more different…
Q: 3. Polyunsaturated fatty acid with 14 carbon atoms.
A: Polyunsaturated fatty acid: a. These are essential fatty acids that cannot be synthesized in the…
Q: Difference between saturated and unsaturated fatty acids.
A: Fatty acids contain carboxylic group thus they are carboxylic acids. They have side chains…
Q: functional groups in palmitic acid, a saturated fatty acid and oleic acid, an unsaturated fatty acid
A: Biological lipids are a chemically diverse group of organic compounds which are insoluble or…
Q: Functional Group Name Structural Diagram (draw all bonds) Found where in the body??? Hydroxyl H -N H…
A: Functional groups can be described as the groups of molecules that get linked to organic molecules…
Q: Comparision between neutral amino acid (tyrosine) and cyclic alcohol
A: Amino acids are monomers of protein they are linked with each other by forming peptide…
Q: Lipid to protein ratio that transport insoluble materials in the blood
A: The basic structure of the membrane is provided by the lipid bilayer. However, membrane proteins…
Q: Cycloalkanes considered. because there are no double or triple *.bonds between carbon atoms…
A: Cycloalkanes are alkanes where the carbon atoms are arranged in a closed ring. They are also called…
Q: Type of interaction that determine the conformation of polypeptide chain
A: The chemistry of amino acids is crucial to protein structure because its side chains can bond with…
Q: The physical properties of cholesterol are best described as amphipathic, charged amphipathic,…
A: Biomolecules that are composed of both hydrophobic and hydrophilic segments are termed amphipathic.…
Q: names of the following polyatomic ions:
A: Many molecules are present as simply binary materials s which are made up of metal cation as well as…
Q: Nomenclature of fatty acids
A: Fatty acids:These are organic acids that have alkyl group chains having a minimum of a 4 and a…
Q: element responsible for the distinct odor of thiols and thioether
A: Thiols give off strong, unpleasant odors resembling that of garlic or rotten eggs hence are used as…
Q: If the second amine would the protein s
A: Oxytocin may be a amide secretion and neuropeptide. it's usually made within the hypothalamus and…
Q: why the notation Ala-Gly-Val-Ala and Ala-Val-Gly-Ala represent two different molecules rather than…
A: The primary structure of protein is a sequence of amino acids. The secondary structure includes…
Q: Glucose is a polyhydroxyaldehyde. Determine the structureof the product of the reaction of glycine…
A: Glucose was a simple sugar that was defined as polyhydroxy aldehydes associated with reducing…
Q: The most unstable conformer in cyclohexane? Boat half chair Chair Twist boat
A: Cyclohexane is a non-polar molecule and it does not have any ring strain, which is the abnormality…
Q: Sequence of Carminic acid
A: Carminic acid is a type of red dye that is extracted from various insect taxa like- Kermes vermilio,…
Q: What special purpose methionine serves
A: Methionine is referred to as an amino acid comprises of the sulphur group responsible for forming…
Q: The functional group involved in glycosidic linkages is: ester carboxylic acid O amide ether
A: Glycosidic linkage is the bond that connects the monosacharide sugar units to form disaccharides or…
Q: Definition from phospholipid inositol
A: Phospholipids are molecules with hydrophilic phosphate heads and hydrophobic lipid tails. They…
Q: pentose phosphate
A: 5. The pentose phosphate pathway is a kind of metabolic pathway that is alternate to the glycolysis…
Q: eL isomers of this sugar.
A:
Q: Sphingolipid Function Name and Structure of Attached Molecule
A: Lipids can be defined as organic substance that are moderately insoluble in water but soluble in…
Q: Chemical properties of fatty acids. Determination of the titer of higher fatty acids in solution.…
A: Fatty acids play an important role of fat in our bodies and food. Fats are broken down into fatty…
Q: structure of phosphatidylinositol that contains oleic acid and arachidonic acid
A: Fatty acids are also lipids with carboxylic acid group attached to long hydrocarbon chain. If there…
Q: pindolol
A: In normal medicine tests in man and animals, beta-adregenic blocking agents attenuates increases in…
Q: Difference between weak and strong acid
A: An acid is a chemical substance or agents that release hydrogen ions when dissolved in water. The…
Q: correctly identifies a phosphate ion? Chemical symbol Cation Anion PO 3+ A 4- В PO, PO РОЗ- PO 3- 4.
A: Biomolecules are those that are produced or found in living organisms. They are of different types,…
Q: The central compound found in the structure of sphingolipids: OA. glycerol OB. sphingosine O c.…
A: A biological membrane having polar lipids as an essential component includes phospholipids…
Q: Common chatacteritics of cholesterol, ergosterol, and bile acids and state its effect in overall…
A: Cholesterol is a waxy substance present in the plasma and in tissues of all animals. Chemically,…
Q: Capable of forming acetylides *
A: Alkyne is an unsaturated compound with a triple covalent bond between C atoms. Ethyne or Acetylene…
Q: D-Erythrulose in ring structure:
A: Introduction: Carbohydrates are also known as saccharides consist of carbon, hydrogen, and oxygen.…
Q: Complete Structural Formula Molecular formula (a) C-C=c (b) C-C-C-č-OH c-c- -c-c-NH,
A: Organic compounds are those that consists mostly of carbon, hydrogen and oxygen. Other atoms…
Q: 2. Circle & Name functional groups -C-N, H. CH2 но CH H,C CH, HICICI
A: Amino acids: It is considered the building block of life as a polymer chain of amino acids forms a…
Q: Monomer of milk sugar. * none galactose lactose maltose fructose
A: Milk sugar
Q: Structural formula of a fat molecule (triglyceride) made of myristic acid, oleic acid, palmitic…
A: Triglycerides are triesters of glycerol and long-chain carboxylic acids called fatty acids. In…
Q: formation of o bonds in (a) BF, (b) PF,. 3
A:
Q: .solubility of vitamins
A:
Q: Sugar-phosphate backbone Hydrogen bond
A: Hydrogen bond -- noncovalent bond, these bonds are found inbetween complementary base pairs of DNA…
Q: Biological significance of protein
A: Proteins are biopolymers made of monomeric units called amino acids. The amino acids are of…
Q: Methionine has a sulfur atom in its formula. Explain why methionine does not form disulfide bridges.
A: The monomers, amino acids are linked by peptide bonds to form a polypeptide chain. One or more…
Q: What type of compound is this? Alkane Alkene Alkyne Arene| Cycloalkane
A: In biochemical reactions, many chemical substrates take part and these chemical compounds are formed…
Q: Glycophospholipid that is associated with lung surfactant
A: Lung surfactant is a mixture of lipids and proteins which is secreted into the alveolar space by…
Q: cis, cis-9,12-OCTAdecadiENoic acid Enumerate the number of carbons and unsaturations (double bonds)…
A: Linoleic acid is also known as Ocatadecadienoic acid is a polyunsaturated essential fatty acid that…
Q: The prosthetic group of hemoglobin and myoglobin is The organic ring component of heme is Under…
A: Hi! As you have posted multiple questions, I will be answering the first three questions for you. If…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- in which of the following alternatives is the greatest amount of energy released from the hydrogenation of:a) 1 mole propeneb) 1 mole of butadiene 1.3c) 1 mole pentyn-2d) 3 moles of etheneIsobutylene, CH2=C(CH3)2, is used to prepare cold-flow rubber. Draw a structure for the addition polymer formed from this alkene.Carvone exists as a pair of enantiomers. (R)-(-)-carvone smells like spearment, whereas (S)-(+)-carvone smells like caraway. Why do these enantiomers have different smells (i.e., different biological activity) ?
- In the monosaccharide derivatives known as sugar alcohols, the carbonyl oxygen is reduced to a hydroxyl group. For example, D-glyceraldehyde can be reduced to glycerol. However, this sugar alcohol is no longer designated D or L. Why?Glucose is a polyhydroxyaldehyde. Determine the structureof the product of the reaction of glycine with glucose.cis, cis-9,12-OCTAdecadiENoic acid Enumerate the number of carbons and unsaturations (double bonds) in this molecule
- Solid iron(II) fluoride (FeF2, Ksp= 2.36 x 10-6) is dissolved in water. If 8.1 x 10-3 mol L-1 of iron(II) ion is found to be in solution. Is the solution saturated, unsaturated, desaturated or supersaturated.True or False: a. D-enantiomer is a mirror image of L-enantiomer b. In a D-carbohydrate, the hydroxyl group on the chiral carbon farther from the carbonyl group points to the right. c. L-carbohydrates are preferred by the humans when processing them after ingestion. d. Sugars that have the cyclic hemiacetal; groups on the anomeric carbon atom are reducing sugars e. In the beta-D –galactose the hydroxyl group on the anomeric carbon atom points up or above the plane of the paper. f. Maltose is a non-reducing sugar and does not have a hemi acetal group. g. Sucrose contain a hemiacetal group and hence a reducing sugar. h. Linen is prepared from celluloseOxidation of a thiol produces a disulfide. True or False
- The free energy of hydrolysis for acetic anhydride is221.8 kJ/ mol. The conversion of ATP to ADP also involvesthe cleavage of an anhydride bond. Its free energy of hydrolysis is 230 kJ/ mol. Explain the difference in these values.In observing a Haworth or cyclohexane-chair representation of alpha-d-glucopyranose, the anomeric carbon can de best identified by: a) the carbon atom which is bonded to the most H atoms b) the carbon atom which is bonded to the least H atoms c) The carbon atom which has its OH group pointing down d) The carbon atom which is bonded to two oxygen atoms Cellulose differs from amylose in that: a) cellulose has 1-6 branches while amylose does not b) amylose has 1-6 branches while cellulose does not c) amylose has alpha glycosidic bonds while cellulose has beta glycosidic bonds d) cellulose contains sulfate while amylose does not. The glycosaminoglycan (mucopolysaccharide) which is not normally associated with joint tissue is: a) heparin b) keratan sulfate c) chondroitin sulfate d) hyaluronic acid In glycoproteins, which amino acid does not bond sugar molecules? a) Asn b) Ser c) Thr d) Gly Which feature do all lipid molecules share in common? a) fatty acid molecules…true or false; 1. aarachidnonic acid is the amjor starting material for eicosanoids 2.both gylycoholic and taurocholic acid contain a side chain amide linkage 3. both cholesterol and cholic acid contain methyl group attachement

