Q: 3. Polyunsaturated fatty acid with 14 carbon atoms.
A: Polyunsaturated fatty acid: a. These are essential fatty acids that cannot be synthesized in the…
Q: What two chemical groups are involved in the formation of a peptide bond?
A: The bond that holds together the two amino acids is a peptide bond.
Q: Amino, acids Amino acids Hydrogen- bond Beta pleated sheet Alpha helix Transthyretin polypeptide…
A: Note: Since you have posted multiple independent questions in the same request, we will solve the…
Q: Cycloalkanes considered. because there are no double or triple *.bonds between carbon atoms…
A: Cycloalkanes are alkanes where the carbon atoms are arranged in a closed ring. They are also called…
Q: Macromolecule Carbohydrates CH₂OH H OH H H OH H HO Lipids QOH HHHH HH My HH Monomer(s)/ Subunit(s)…
A: Biomolecules are carbon-based organic compounds produced by living organisms. They are made up of…
Q: The bond between 2 amino acids ● A. glycosidic bond OB. N-glycosyl linkage ● c. peptide bond OD.…
A: Introduction :- Proteins are made up of substances called amino acids. Proteins and amino acids are…
Q: Identify the chiral carbons in -D-fructose. Identify the appropriate atom by selecting an atom and…
A: A chiral center is defined as an atom in a molecule that is bonded to four unique atoms or groups…
Q: The bond Indicated with a green arrow below Is referred to as a... bond. Check the proper term that…
A: Monosaccharides- Simple sugar molecule- having One oxygen atom and two Hydrogen atoms for a single…
Q: Chemical formula of Nucleotides
A: The nucleic acids are formed from monomers called nucleotides. There are four nucleotides in DNA,…
Q: H C-H O: H- OH HO H- C OH H C-H OH Which of the following best describes the saccharide shown? a…
A: Sacharides are defined as sugar molecules which are nothing but carbohydrates. The sacharides are…
Q: Hydrophobic bonds are too weak to influence the three-dimensional structure of macromolecules.
A: Biological macromolecules play a major role in different cellular functions and aids in the growth…
Q: NUCLEIC ACID "BASE PAIRING" 1. 5'-A-T-A-A-G-G-G-G-C-T-C-3' 2. 5'-G-G-G-G-C-T-A-C-T-C-3'
A: The nucleic acid base pairing follows simple rule A pairs with T while G pairs with C i.e purine…
Q: Structures: Purine Pyrimidine
A: Nucleic acids are the biomolecules that are responsible for conserving the genetic information of an…
Q: dna nucleotides cannot h-bond to rna nucleotides. true or false
A: DNA is a double helix and forms hydrogen bond between the base pairs ( Adenine , uracil , guanine…
Q: Describe the structure of an amino acid. Be sure to describe how the R-group of an amino acid will…
A: Structure of Amino acid: Generally, amino acids are considered as the building blocks for proteins.…
Q: Amyloid fibers, which are insoluble and are the source of mad cow disease, alzheimer disease, and…
A: A protein is considered to be a biological macromolecule made up of one or more polypeptide…
Q: Bond types Primary Secondary Tertiary (Quaternary) Protein levels Covalent H-bond lonic Van der…
A: All proteins exist in primary structure and most of them found in secondary, tertiary structure, and…
Q: Molecule Amino Acid (Glycine R=H) Glycerol a-glucose Molecular Formula Variable C2H5NO2 Structural…
A: Biomolecules are carbon-based organic compounds produced by living organisms. They are primarily…
Q: What sugar units make up the structure shown? ÇH,OSO,- Но Lo- H. H. ÇOO- NHCCH, OH H H он
A: Carbohydrate consists carbon, hydrogen and oxygen. It is of different types like monosaccharides,…
Q: H H |H I peptide chains H Type of bond Choose ペーUーエ アー O=U 1.
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Macromolecule Proteins Amino +H H H Nucleic Acids phe Carboxyl 0 Monomer(s)/ Subunit(s) and Name of…
A: There are 4 types of Biomacromolecules. They are; Proteins Nucleic Acids Carbohydrates and Lipids…
Q: CH OH НО fatty acid chain
A:
Q: pentose phosphate
A: 5. The pentose phosphate pathway is a kind of metabolic pathway that is alternate to the glycolysis…
Q: Haworth Projection Formulas Practice Exercise Student Name:. Has 2 carbon Pentose? Ketose? Has pyran…
A: Carbohydrates contain aldehyde or ketone compounds with multiple hydroxyl groups present in them.…
Q: Aldehyde and ketone functional group
A: Functional groups are substituents that are attached to a molecule and are responsible for the…
Q: NIT CH OH CH: H. CH: H CH CH: NH -COO ーC-ド HU Using the polypeptide above; How amino acids in the…
A: The polypeptide chain is composed of amino acids linked together via peptide bonds. The polypeptides…
Q: Which bases can exist in either keto or enol form
A: Tautomers are molecular isomers found in fluid or in cells. Chemical bonds are changed numerous…
Q: Check all that applies to the structure of fatty acid. * С —с — с —С — С — с —С С- с —с — н нн H H H…
A: Fatty acids are the most common lipids. They are basically of two types, these are: Saturated…
Q: A chiral center is a carbon with four different substituents. The number of chiral centers in…
A: Carbohydrates are polyhydroxy aldehyde or polyhydroxy ketone or compounds which produce them on…
Q: Encircle the glycosidic bond in the molecule. Determine how many glycosidic bonds are in the…
A: A glycosidic bond is the bond that is formed between the hemiacetal or hemiketal group of a…
Q: Contribution of the features of the alpha-helix to the stability of the protein.
A: Protein play wide variety of essential function in our body. They provide strength and structural…
Q: 7. general structure of a TAG 8. structure of glycerol 9. structure of a sterol
A: You have asked question with multiple subparts. I will answer 1st three subparts. Triacylglycerol -…
Q: Which linear monosaccharide below corresponds to this cyclic one? A) A B) B C) C D) D E) None…
A: Monosaccharides are the simplest and smallest unit that can not be degraded any further.…
Q: Reduction of the aldehyde group of a monosaccharide yields a _____________.
A: Carbohydrates are simple sugars which are polyhydroxy aldehydes or ketones with reducing property.…
Q: Each protein monomer is made up of four parts bound to a central carbon. List the 4 parts by name
A: Biomolecules are organic compounds found in living organisms. All living organisms will have these…
Q: Hydrophobic interaction. As a polypeptide folds into its functional shape, amino acids with…
A: There are several levels of organization that occurs when a polypeptide chain folds into its…
Q: Alpha-helix of proteins, its characteristics. Draw a diagram of the arrangement of amino acids in…
A: Alpha helix Secondary structure of proteins Right-handed helix Backbone NH group hydrogen bonds…
Q: - TRUE about the peptide bond:
A: With the loss of a water molecule, a covalent link is formed between the - carboxyl group of one…
Q: Polysaccharides are biopolymers composed of hundreds to thousands of simple sugar units…
A: Biomolecules are the biological molecules that are present inside the living organisms. These…
Q: Difference between polypeptide and protein
A: A peptide is two or more amino acids joined together by peptide bonds; a polypeptide is a chain of…
Q: Formation of peptide bonds, di- and tripeptides and their nomenclature. Examples of synthetic…
A: Proteins are the polymers of amino acids. Amino acids are joined together by peptide bond.
Q: Peptide bond is /has ……………. This structural feature prevents protein to sample all possible…
A: In a protein or a polypeptide chain, two amino acid molecules are joined together by a peptide bond.…
Q: Hydrophobic intercation associated with protein tertiary structure involve
A: Hydrophobic interactions are the type of interactions between water and hydrophobic molecules (low…
Q: Association of two or more polypeptides(ex. tetramer quaternary structure primary structure tertiary…
A: Proteins are unbranched polymers constructed from 22 standard α-amino acids. They have four levels…
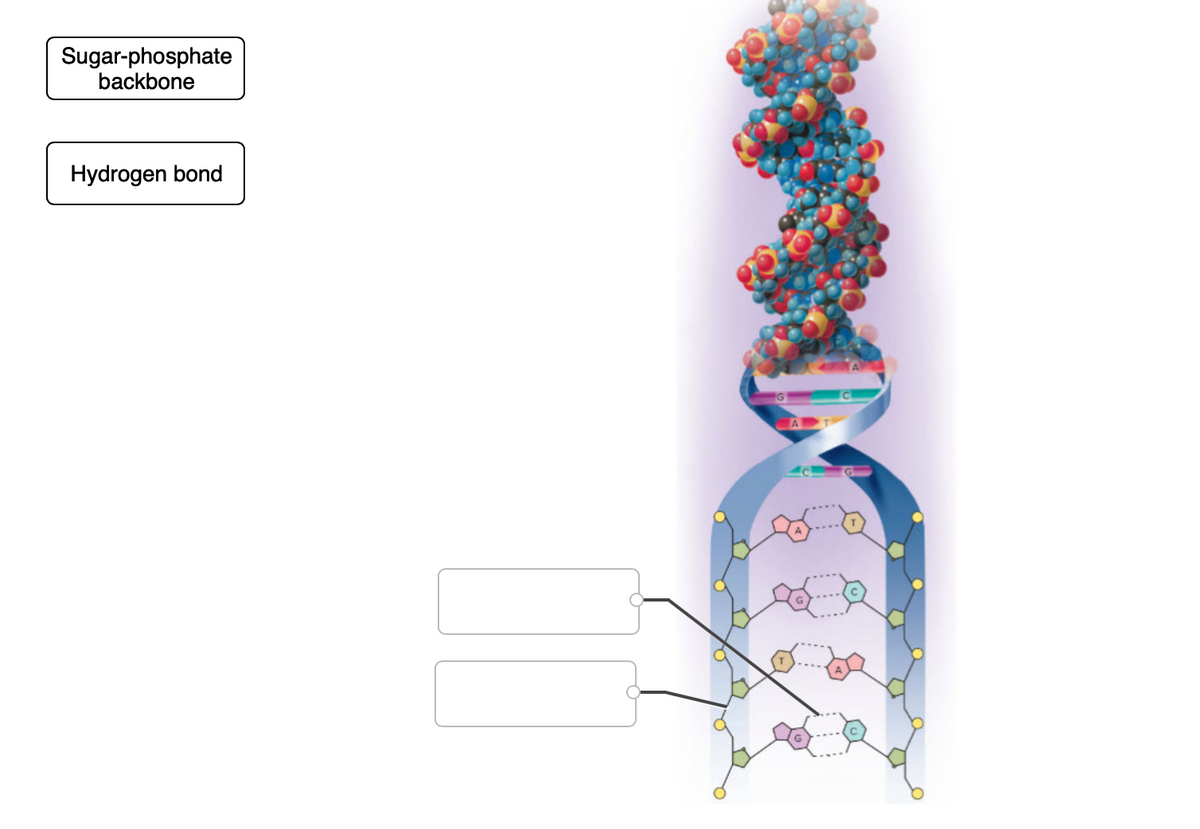
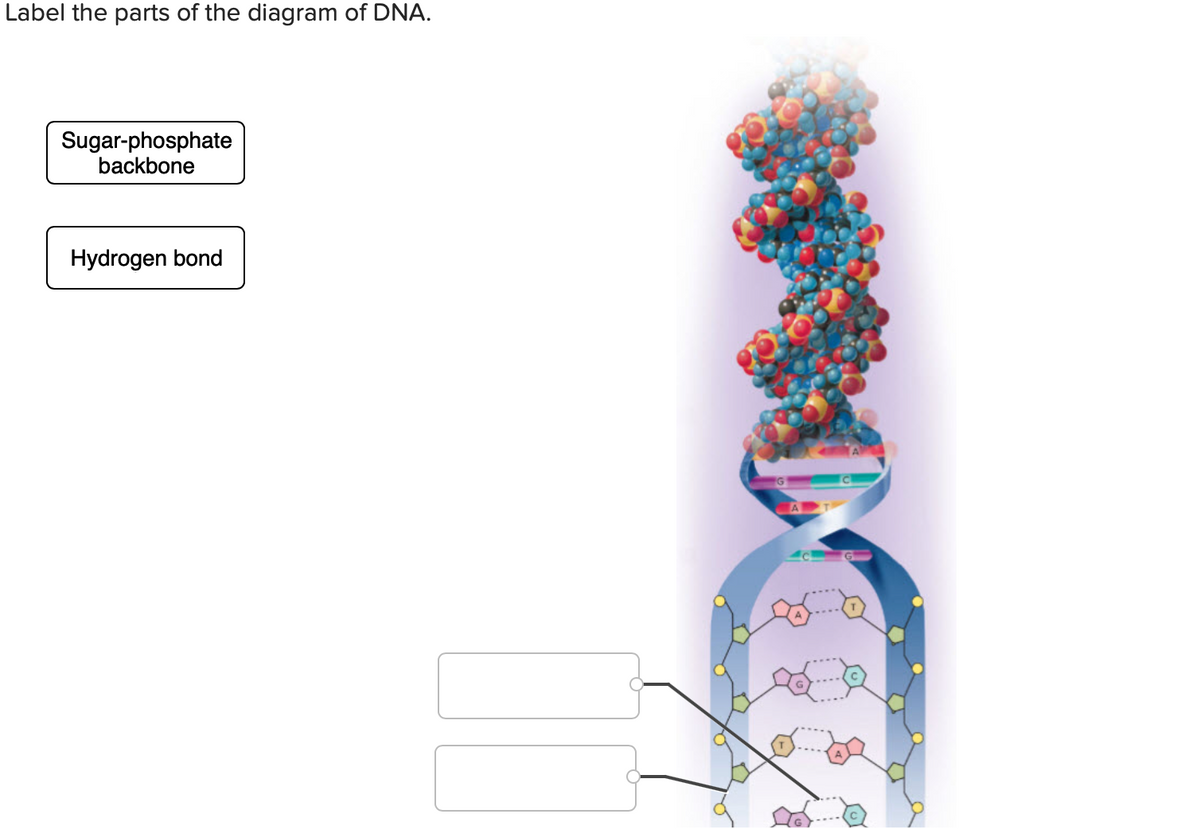
Q: STRUCTURE OF DNA Phosphate Nitrogenous Base Pentose Sugar Referring to the illustration above, the…
A: DNA is the geentic material. It stands for deoxyribonucleic acid. It is present in the nucleus,…
Q: Two sugars which differ from one another only in configuration around a single carbon atom are…
A: Diastereomers, enantiomers, epimers etc are terms used to study stereochemistry of biomolecules.…
Q: Red pepper, green pepper, cassava flour, cornmeal white, onions, tomatoes and shito. State whether…
A: Carbohydrates are the most widely available energy constituents present in the food. These are…
Q: The main chemical structure difference between ribonucleic acid and deoxyribonucleic acid is in the…
A: Nucleic acids are the polymers that are known to be essential to all forms of life. Nucleic acids…
Q: Glycogen is a polysacchsride of which type of macromolecule
A: There are four major types of macromolecules. Lipids Proteins Nucleic acids Crabohydrates…
Q: TRUE OR FALSE Hydrophobic bonding between stacked purine and pyrimidine Hydrogen bonding between…
A: Nucleic acids are macromolecules made up of nitrogenous bases, a pentose sugar, and a phosphate…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

- Choose the combination of answers that most accurately completes the statement. A nucleotide contains which of the following? a. 5-carbon sugar d. b and c only b. nitrogen base e. all of these c. phosphateRNA contains the sugar Group of answer choices ribose. deoxyribose. glucose. phosphate.A......of a DNA consists of a sugar, a phosphate, and a nitrogen containing base. A....... is a change in the base sequence of a DNA molecule Took x-ray images of DNA molecule Name 3 types of RNA and list their roles in making proteins
- A) Draw the structure and give the name of a nucleotide made of G + ribose. B) Write the complementary base sequence for the matching strand in the DNA section shown below.5’ – C T G T A T A C G T T A – 3’ Please answer both partswrite the difference between DNA and RNA. Also compare the function of DNA and RNA . ( please write in your own words , do not copy from chegg bartleby )Complete the table below to show the differences between a DNA molecule and an RNA molecule. Parameter DNA RNA Nitrogenous Bases Number of strand/s Sugar group
- Draw and label a diagram showing four DNA nucleotides, linked together in two strands. PLEASE DRAW ON PAPERIn the double helix of DNA, the ____________ bases of one strand exhibit complementary base pairing with the bases of the other strand.The backbone of DNA is held together by ____________ bonds. Question 3 options: N-glycosidic Peptide Hydrogen Phosphodiester

