Q: What is the solubility of PbF₂ in a solution that contains 0.0550 M F⁻ ions? (Ksp of PbF₂ is 3.60 × ...
A: The solubility of any salt is defined as the how much the given solute can be dissolved completely i...
Q: How to define appearance of substance?
A: The physical appearance of a substance is its colour, size, shape, boiling point, melting point, tas...
Q: How many covalent bonds? How many types of covalent bonds forming your double bond? What are the typ...
A: As per our guidelines we can only solve first three sub-parts. Please resubmit the other questions a...
Q: Which statement about chemical equilibrium is not true? O The same equilibrium state can be attained...
A: The reaction rates of the forward and backward reactions are generally equal. All reactant and prod...
Q: Which anions were present in the student’s unknown? Explain.
A: In the first step, an unknown solution is first acidified with Nitric acid (HNO3) which generates bu...
Q: 1. Octane, C8H18, has 18 different constitutional or chain isomers. One of them, isooctane, is used ...
A: C. n-Octane will have a higher boiling point than 3,3‑dimethylhexane, because it branches less than...
Q: The reaction of (CH3)3CBR with hydroxide ion proceeds with the formation of (CH3);COH. (CH3);CBr(ag)...
A:
Q: 8a) Show the mechanism for the radical reaction of pentene 3-methylhexane with Br2 in the presence o...
A:
Q:
A: Given that, w/v % = 0.85% M.Wt = 58.5 g mol-1 Normality = ?
Q: Write the balanced NET ionic equation for the reaction when nickel (II) iodide and aluminum sulfate ...
A:
Q: What are the signs of AH°, AS" and AG° for the reaction of dissociation of N2 moiecuie to two nitrog...
A: Since there is triple bond between two N atom hence breaking of triple bond is quite difficult requi...
Q: might yield. Writing the reaction mechanism is different from writing the overall balanced equation....
A: Detail mechanistic pathway is given below
Q: An analytical chemist is titrating 52.7 mL of a 0.3500M solution of methylamine (CH, NH,) with a 0.3...
A:
Q: Why is neutralization between an acid and a base exothermic reaction
A: Neutralization reaction:
Q: volume of the gas if the temperature is Ne gas occupies a volume of Ne gas occupies a volume r volum...
A: Given- V1=355 ml T1=345K T2=300K V2=?
Q: Enter your answer in the provided box. The atomic masses of the two stable isotopes of copper, Cu (6...
A: Calculation of average atomic mass of cu:
Q: There are two steps in the extraction of copper metal from chalcocite, a copper ore. In the first st...
A: Chemical reactions can be added, subtracted, or multiplied like an algebraic equation.
Q: The oxidation of ammonia into nitric oxide is an exothermic process. 4NH,(g) + 5O,(g) AH n = -904.4 ...
A:
Q: Predict whether the solution NH,CN (aq) is acidic, basic, or neutral, and explain the basis of your ...
A: NH4CN is a salt which is made by neutralisation reaction between an acid and a base.
Q: Give 5 examples of Nonpolar and Polar Solvents.
A: Non-polar Solvents : Non-polar solvents are that compound which do not have a permanent dipole mome...
Q: Carbon dioxide and water react to form methanol and oxygen, like this: 2 CO,(9)+4 H,0(9)→2CH,OH(1)+3...
A:
Q: 9- Calculate the molar concentration of HNO3 (63.0 g/mol) in a solution that has a specific gravity ...
A: Molarity is a concentration term for a solution. The molarity of a given solution is defined as the ...
Q: le answer for each question and fill in the corresponding bubble on your Scantron form. 1. A solutio...
A: Polar compound are those compounds which have polar bond which arises due to difference in electrone...
Q: After performing an experiment on density. You found out that the actual density and the calculated ...
A: The experiment performed was the determination of density of a substance.
Q: NH3(9) + 502(g)-4NO(g) 6H20(g), vas carried out at a certain emperature. A system was prepared with ...
A:
Q: Given the following two reactions with their enthalpy changes (AH): N2(g) + 3 H2(g)-2 NH3(g) AH1 = -...
A: The Hess's law states that the total enthalpy change during a complete chemical reaction is the same...
Q: Aqueous hydrochloric acid (HCI) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium ...
A: Given, Mass of HCl = 1.8 g Mass of NaOH = 3.0 g What is the theoretical yield of NaCl when NaOH r...
Q: Which of the following statements is TRUE about elimination tests for anions? of Select one: SCN' in...
A: Here we have to write about correct statement for elimination tests for anions. Here no option is co...
Q: Please answer fast There is a 20 cm-1 distance between two consecutive lines in the microwave spect...
A: The solution of the question is given below:
Q: a solution is made by dissolving 13.5g of glucose in 0.100kg of water. What is the masspercentage of...
A: The mass of solute in gram present in 100 g solution is known as the mass percent concentration of a...
Q: Given a solution of 0.1 M Ag+ and O.2M of Pb2+, (Ksp for silver oxalate = 3.5x10^-11 and ksp for lea...
A: Solubility product Ksp is the product of ionic concentration required to precipitate any insoluble s...
Q: Iron and water react to form iron(III) oxide and hydrogen, like this: 2 Fe(s)+3 H,O(g)→Fe,O3(s)+3 H,...
A: The equilibrium constant of a chemical reaction is the value of its reaction quotient at chemical eq...
Q: A solution has [Fe2+] of 0.0200 M. What concentration of sulfide is needed before precipitation begi...
A:
Q: What is the mass percent of an aqueous sodium carbonate solution in which the mole fraction of Na2CO...
A: Solution - According to the question - Given - Moles of Na2CO3 = =mole fraction * moles of solution ...
Q: 1.You need to prepare 1L of a 5xTE stock solution (1x TE buffer:...
A: Q1) Volume of buffer solution = 1 L (1000 mL) Conc. of buffer solution to be prepar...
Q: 1. Aluminum metal (Al) and hydrogen chloride (HCI) react to form aluminum chloride (AICI3) and hydro...
A: Given, Al and HCl react to form AlCl3 and H2 gas , then A. What is the balanced reaction = ? B. Ho...
Q: Consider the reaction: H + ¿n → 2¾H + ¿n + _}H. Calculate the energy change for this reaction. Is er...
A: Energy relesed in a nuclear reaction is, E= E=Δm×931MV
Q: Given a solution of 0.1 M Ag+ and 0.2M of Pb2+, (Ksp for silver oxalate = 3.5x10^-11 and ksp for lea...
A: More soluble precipitate is the one having higher solubility product value. Here lead oxalate is the...
Q: 4.00 g of a certain Compound X, known to be made of carbon, hydrogen and perhaps oxygen, and to have...
A:
Q: Calculate the approximate length of a PVA strand in centimeters, assuming that each monomer unit con...
A: The O-C-C bond point in PVA chain is three-sided plannar, bringing about a bond point of 120°. Nonet...
Q: An analytical chemist is titrating 185.8 mL of a 1.200M solution of benzoic acid (HC,H,CO,) with a 1...
A:
Q: Ô https://app.101edu.co Question 3 of 24 A substance which is cyclic, conjugated, and has 4n + 2 pi ...
A: Compound is wheather aromatic, antiaromatic or non aromatic is obeyed by Huckel rule.
Q: 6) Using arrows to represent electrons, Construct the orbital diagram of the Cr and Cr3+ ion. Cr Cr3...
A: The question is regarding electronic configuration of elements. General trends of size and electron ...
Q: Give the product(s), including all stereoisomers, of the reactions below. Is the product mirture opt...
A:
Q: Aqueous hydrobromic acid (HBr) reacts with solid sodium hydroxide (NaOH) to produce aqueous sodium b...
A:
Q: For the following balanced reaction: 2NA + 2H2O = 2NaOH +H2 Which would create more moles of NaOH: ...
A:
Q: 5. The product(s) of the following reaction Mow CH2-CH2 excess HBr CH2 is/are: heat Od CH2 MAD (A Io...
A: HBr dissociate and formed H+ and Br-,where H+ act as a electrophile and Br- act as a nucleopile
Q: Calculate the amount of phycocyanin in Sample 1 in mg where A620=0.211 and A650=0.086, taking into...
A: Phycocyanin is a protein-pigment complex that belongs to the family of phycobiliprotein. It is a wat...
Q: Calculate the enthalpy change for the dissolution of (NH4)2SO4(s) using the enthalpy of formation da...
A:
Q: Give the IUPAC name. Use the IUPAC 1993 recommendations for organic nomenclature.
A: Given:- To find:- The IUPAC nomenclature of the above compound.
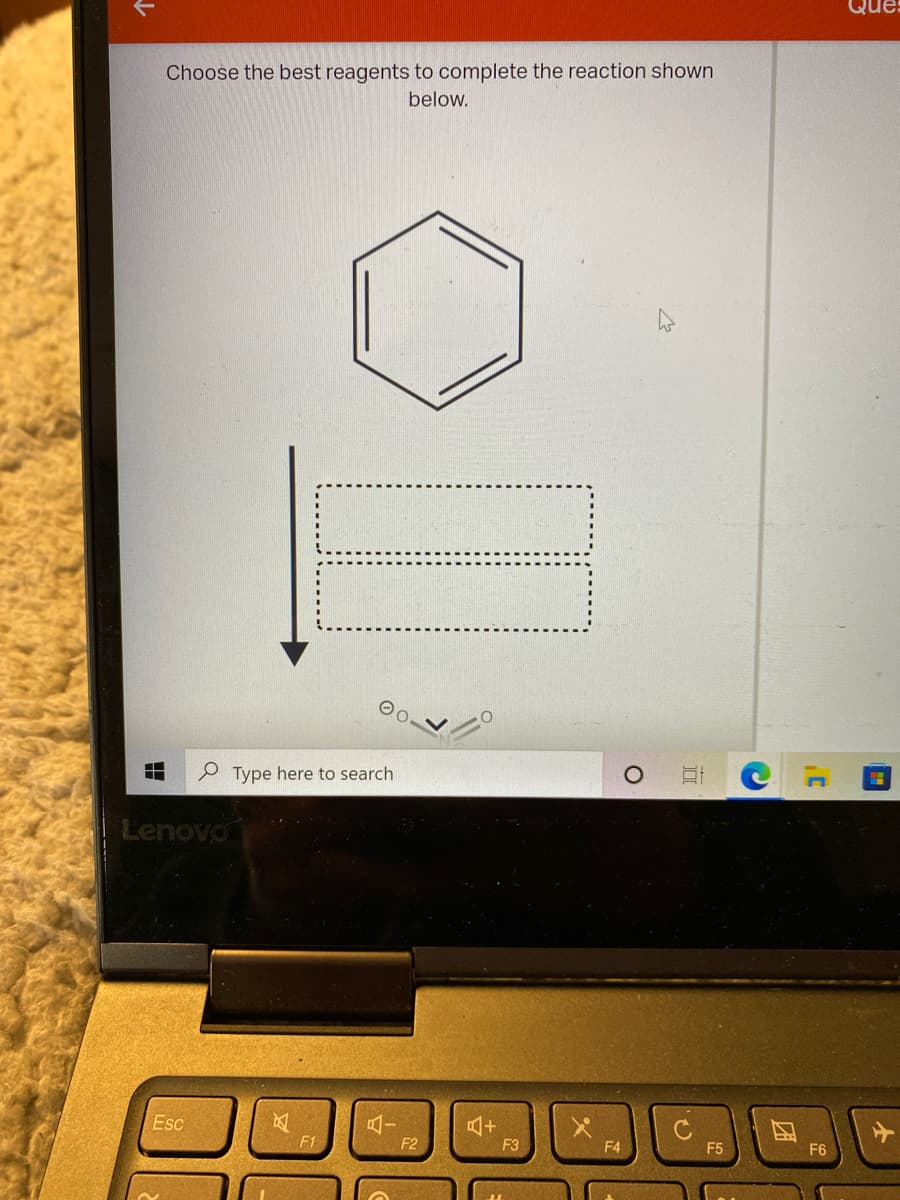
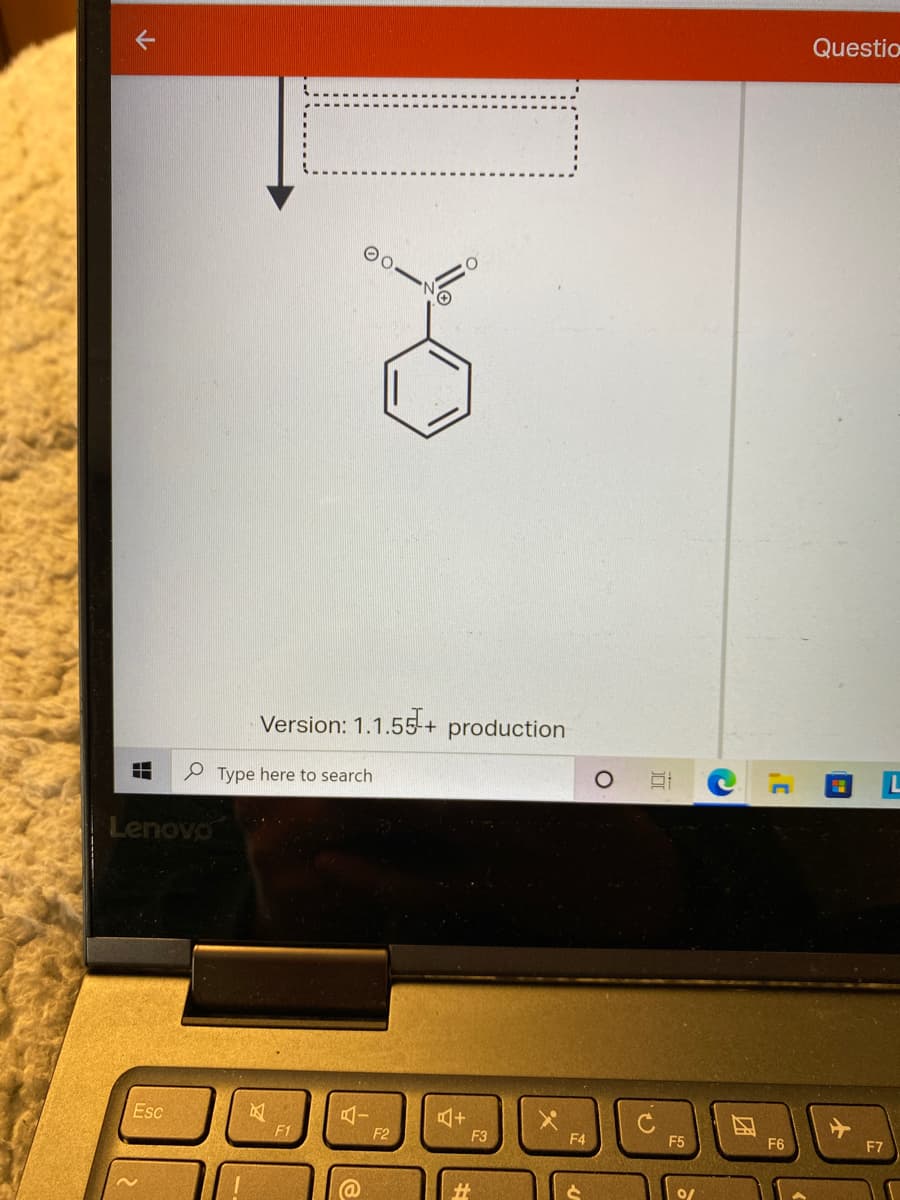
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Supply synthetic routes to the following molecules using the indicated starting materialsFor SN1 Explain the order in which 2o (secondary) alkyl halides reacted (fastest to slowest) and explain why. 2o (secondary) compounds listed are: (see picture) 2-chlorobutane 2-bromobutane bromobenzene bromocyclopentane bromocyclohexane Base your explanations on the following considerations: the nature of leaving group, the effect of structure, steric hindrance and any other feature. Be sure to explain which alkyl halides did not react and whyGive the products of the reaction given below and state which reaction willproceed faster?
- Under second-order conditions (strong base/nucleophile), SN2 and E2 reactions may occur simultaneously and compete witheach other. Show what products might be expected from the reaction of 2-bromo-3-methylbutane (a moderately hindered 2°alkyl halide) with sodium ethoxide.provide the starting material using the product on the right. also provide the reagentsGive the missing reactants, reagent(s), conditions, and/or major product for the reactions below-

