Q: If you have a reaction where OH⁻ acts as a base, what would be the conjugate acid? A. O^2- B. NaOH…
A: General concept.
Q: 1. Arrange the following compounds in order of increasing acidity (start with the most acidic). ÇOOH…
A: Carboxylic acids are Organic compounds with acidic nature. Alcohols are generally neutral but…
Q: I CH;O- is a stronger base than CH;OH A) True B) False True
A:
Q: Draw the products of each proton transfer reaction. Label the acid and base in the starting…
A: Interpretation - The products of each proton transfer reaction are to be explained. Concept…
Q: + NAOH A OH
A: If the conjugate ion is more stable then the compound will be more acidic.
Q: Rank the conjugate bases of each group of acids in order of increasing basicity. a.NH3, H2O, CH4…
A: (a)
Q: Complete each acid-base reaction and predict whether the position of equilibrium lies toward the…
A:
Q: 1 Label each reactant and product in this reaction as a Brønsted acid or base.…
A: Interpretation- To label each reactant and product as Brønsted acid or base in the given reaction -…
Q: Label each reactant and product in this reaction as a Brønsted acid or base. CH, OH + OH- CH,O¯ +…
A: According to Bronsted concept: An acid is defined as a substance which has a tendency to donate a…
Q: What is the product of the following reaction?
A: The molecule that has lone pair of electron on it acts as a nucleophile as it is electron rich and…
Q: What is the product of this reaction? NaOH Br
A: In the given reaction, NaOH is a good base, and Br is a good leaving group. So, here elimination…
Q: What is the app. Keg of the following acid/base equilibrium? Na NaOCH3 CH3OH
A: Bronsted acid are those which can donate H+ ions in aqueous solutions. Similarly Bronsted base are…
Q: 2. Name the acid and its conjugate base (in the forward reaction) in the equation below: a) CH3NH₂ +…
A: Acid donates a proton or hydrogen ion in aqueous solution and base accepts a hydrogen ion in aqueous…
Q: What is one set of conjugate acid/base pairs in this reaction? CH3COOH(aq) +…
A: We have to choose the one set of conjugate acid/base pairs in this reaction. CH3COOH(aq) +…
Q: An aqueous solution of CH3NH3NO3 will be Group of answer choices A. basic, because of the…
A: Since the salt given CH3NH3NO3 is made from the acid HNO3 and base CH3NH2. And we know that the salt…
Q: Draw the products of each acid–base reaction. Indicate whether equilibrium favors the reactants or…
A: Amines are denoted by the formula RNH2, where R can be an alkyl or aryl group. The presence of lone…
Q: In the following reaction, what compound is the conjugate base? HBr + NH3 →NH4+ + Br−
A: In this question, we have to find out the correct answer of given problem by the help of conjugate…
Q: If OH⁻ acts as a base in a reaction, what would be its conjugate acid? A. OH⁻ B. NaOH C.O2-…
A: conjugate base is formed when an acid donates a proton. conjugate acid is formed when a base accepts…
Q: Complete the following acid-base reactions.
A: The given reaction is,
Q: CH3 o 11 HgC-C-C Ha C-C CH3
A:
Q: Identify the base in the following non-aqueous reaction POCl3 + Cl- → POCl4-
A: The base in non aqueous medium is given below
Q: HCN (aq) + SO4^2- (aq) --> CN^- (aq) + HSO4^- (aq) Predict whether you would expect the reaction to…
A: According to Bronsted Lowry concept, acid is a substance that gives proton and base is a substance…
Q: List the following compounds in order from strongest acid to weakest acid: CH4 CH3COOH CH3OH CHC,OH
A: An acid is a compound that loses proton or accepts an electron. However, a base will be just…
Q: Which two species act as bases in the acid-base reaction shown below? H2SO4 HNO3 H2NO3 HSO4 + + 1 2…
A: The given acid-base reaction is a Bronsted-Lowry acid-base reaction.
Q: CH3O- is a stronger base than CH3OH. * False O True
A: We have to predict whether statement is true or not.
Q: Determine whether the following conjugates can undergo hydrolysis. 1. ClO4- 2. CH3NH3+
A: The given conjugates whether they undergo hydrolysis or not has to be determined.
Q: 10. Which is the conjugate acid in the following reaction? :CH3 H20 CH4 Но + II II IV А) І B) II C)…
A:
Q: Which of the following is the correct use of arrow-pushing for this reaction? o-H :H + O-H H + Fo:
A: In the given reaction, the nucleophile hydride ion will abstract the proton from the given alcohol.
Q: Which of the following bases would you use to accomplish the reaction shown below? Br • NH3 • T-BUQK…
A: Organic reaction mechanisms
Q: product reagents 1 Acidic KMNO4 HO,
A: As per the rules, only the first question can be answered. The product mentioned for question 1 is…
Q: Ethanol can act as either a Lewis acid or a Lewis base (donates or accepts a pair of electrons).…
A: Ethanol is an alcohol with two carbon atoms bearing an alcoholic group with a molecular formula…
Q: Complete the following reactions: CH,CH,I (1) NaOEt, ELOH
A: Given reaction is : Complete the reaction and give the major product = ?
Q: Draw the products of each acid-base reaction. a. + NaOH OH bmou
A: Dear student since you have asked multiple questions but according to guidelines we will solve 1st…
Q: Show the mecanism of the following reactions NaOH но 2 Br Br2, H30" H 1. NaOEt/ ELOH 2. На
A: In this question, we will showing the mechanism for all three reactions by using curved arrows. You…
Q: Draw the products of the following acid base reactions and indicate whether products or reactants…
A: Since you have posted a question with multiple sub-parts, we will solve first three subpartsfor you.…
Q: 5. Identify the Bronsted acid and the Bronsted base in both sides in each reaction below: a. CH,OH +…
A: Bronsted acid is the substance which donates protons in the reactions. And Bronsted base is the…
Q: Which of the following compounds is the weakest acid? A) CH3CH3 CH3OH CH3CI CH3CN
A: “Since you have asked multiple question, we will solve the first question for you. If you want any…
Q: Complete the folowing reactions. If no reaction occurs, write "No reaction" a) CH -Ç H-CH, -COOH…
A: Acids possess an ability to liberate H+ ions in solution. When they are treated with strong metals,…
Q: What is the major product of the following reaction?
A: Inorganic chemistry, Cyanide is used to increase the carbon numbers in a molecule. It is very toxic…
Q: What is the major product of this reaction? NaOH
A:
Q: Label each reactant and product in this reaction as a Bronsted acid or base. CH3OH + OH- ---->…
A: In the neutralization reaction, the species donates a proton H+ is called Bronsted acid and the…
Q: Correctly identify the products of the following general acid base reaction [ H2X Z(OH)3 → products…
A: According to Lewis theory, acid is the one which accepts the electrons and base is the one which…
Q: 17. Which of the following will most readily underg an Sn2 reaction with NaOH? Br а. Br b. Br с. Br…
A: The question is based on the concept of organic reaction mechanism. we have to identify the reagent…
Q: Complete each acid-base reaction and predict whether the position of equilibrium lies toward the…
A: To find: Reactions of given acid-base. Details about the reactions. The reaction favors towards…
Q: Without looking them up, rank the following compounds in decreasing order of acidity.These examples…
A: The given compounds have to arranged in the decreasing order of acidity.
Q: 1) Draw the acid/base products of the following reactions: HO. NaOH HCI NH2 NaOH HCI HO
A: Acids give H+ ion to form its conjugate base. Bases accept H+ ion to form its conjugate acid. They…
Q: Which of the following reactants is a base? H2O(1) + CH3CH2NH2(aq) → CH3CH2NH3*(aq) + OH (aq) O a.…
A: The substance which donates H+ is an acid or which accepts H+ is a base.
Q: Mg(OH)2 is a strong base. True or false
A:
Q: Draw the products of each acid–base reaction. Indicate whether equilibrium favors the reactants or…
A: The products of each acid-base reaction has to be given.
Q: Which member of each pair is the stronger acid or base? (a) HClO or HClO3 (b) HCl or CH3COOH…
A: given a) HClO or HClO3 (b) HCl or CH3COOH (c) NaOH or CH3NH2
Can you help me complete this acid-base reaction and determine the reaction direction?
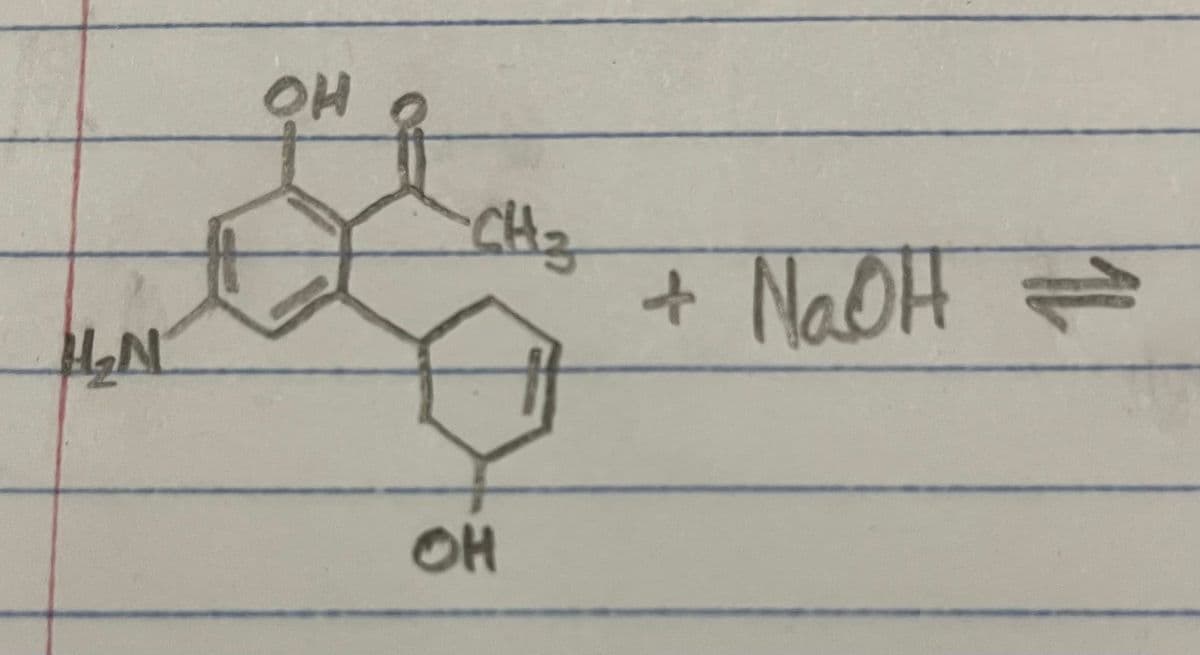
Step by step
Solved in 2 steps with 1 images


