Q: Calculate the amount of H2S in cubic meters measured at 49°C and at a pressure of 0.2 barG, which ma...
A: Hydrogen sulphide (H2S) can be prepared by action of FeS with dilute hydrochloric acid (HCl). The eq...
Q: A 100.0 mL100.0 mL solution of 0.0200 M Fe3+0.0200 M Fe3+ in 1 M HClO41 M HClO4 is titrated with 0.1...
A: The balanced titration is as follows : The two half reactions that occur at the indicator electrode...
Q: write a structural formula for compound. Q.) 3-Chloro-1,2-propanediol
A: The given name for organic compound is 3-Chloro-1,2-propanediol. The root name "propane" indicates t...
Q: Determine the number of moles (of molecules or formula units) in sample. 55.98 g CF2Cl2
A:
Q: Consider the reaction of solid P₄ and chlorine gas to form gaseous phosphorus trichloride. The balan...
A: The reaction given is P₄(s) + 6 Cl₂ (g) → 4 PCl₃ (g)
Q: The pathway for nicotinic acid requires a series of highly unfavorable reactions. Which other alkalo...
A:
Q: Show how you might bring about the following conversions. For any conversion involving more than one...
A:
Q: Show how alcohol or diol can be prepared from an alkene. Q.)1,2-Hexanediol
A: Oxidation Reaction is a reaction of carbon compounds with oxygen. Basically, oxygen atom is added in...
Q: Zinc(s) (MW=65.38), with a mass of 0.228 g, was reacted with 20.00 mL of NaClO (MW=74.44) according ...
A: The species normality can be explained as the species concentration in which the algebraic ratio bet...
Q: If 5.00 x 102 calories of heat are absorbed by 100.0 g H2O initially at 20.3 °C, what is the final t...
A:
Q: Three large proteins are ionized at the pH at which an electrical FFF separation is carried out. ...
A: Field-flow fractionation, (FFF) is a type of separation technique in which field is applied to a f...
Q: Reaction: N2O (g) -> 2N2 (g) + O2 (g) Performed at a temperature of 575 ° C across platinum in a ...
A: Since you have asked multiple questions, we will solve the first question for you. If youwant any sp...
Q: When carrying out this two step procedure (bromination and subsequent elimination) on cyclopentene, ...
A: The product of given reaction surely could not be alkyne since it is strained one.
Q: What is the pH of a buffer solution that is 0.46 M chloroacetic acid and 0.29 M sodium chloroacetate...
A: Since chloroacetic acid is a monoprotic acid.
Q: Complete the following Molecular Orbital Diagram of NH3 by a) Using ↑ and V symbol to fill in electr...
A: NH3 Molecular orbital diagram: Electronic configuration of N7 =1S22S22P3 ...
Q: At 25oC, a 100mL solution is found to contain 20% by mass acetic acid (CH3COOH). The density of the ...
A: Given: Density of solution (ρs) = 1.05 g cm-3 Volume of solution initially (V1) = 50 mL Final volume...
Q: Calculate The solubility of Ba(IO3)2 in a solution prepared by mixing 200 mL of 0.00100 M Ba(NO3)2 w...
A: As you have posted multiple parts of the question and have not mentioned which part you want us to s...
Q: Name compound
A: Following points should be kept in mind while naming an organic compound: The parent chain should c...
Q: How many grams of sulfur would be needed to produce 23.4 g of H2O using the following balanced equat...
A: The reaction given is Since mass of H2O to be produced = 23.4 g And since mass = moles X molar mass...
Q: Suppose you were told that each reaction is a substitution reaction, but you were not told the mech...
A:
Q: 16.78
A: (a) Kb for ammonia is 1.8*10^-5 Kb for hydroxylamine is 1.1*10^-8
Q: When does reduction process occurs?
A: The reduction reaction may be explained as the process in which species gained electrons and the oxy...
Q: Carbonic acid, H2CO3, can be found in a wide variety of body fluids (from dissolved CO2). (a) Calcul...
A: The Bronsted-Lowry acid-base theory was purposed by Bronsted and Lowery is called Bronsted-Lowry aci...
Q: Using the given starting material, how would you prepare the target molecule? (Hint: two different r...
A:
Q: Carbonate ion, Hydroxide ion, Carbon monoxide, phosphorus trifluoride
A: Since you have asked multiple questions with multiple subparts, we will solve the first question and...
Q: Show the product(s) with the correct stereochemistry, if applicable. -CH2CH3 1. ВНз, THF 2. Н2О2, ОН...
A:
Q: Which of the following Lewis structures would be an expansion to the octet rule? Select one: a. N...
A: The Lewis structure or molecular skeleton is also known as electron dot structure. It is the complet...
Q: Trimethylamine, (CH3)3N, is a gas with a fishy, ammonia–like odor. An aqueous solution that is 0.29 ...
A: The ionisation reaction of (CH3)3N is (CH3)3N (aq) + H2O (l) ------> (CH3)3NH+ (aq) + OH-
Q: Describe how to prepare 50.0 ml of a 3.00M hydrochloric acid solution using 12.0M HCl.
A: Assuming V mL of 12 M HCl solution is used to make 50 mL of 3.00 M HCl solution. Since moles of HCl ...
Q: The pH of a household ammonia solution is 11.75. Calculate the mass of ammonia needed in a 2.50 L s...
A:
Q: A pipet is used to transfer 5.00 mL of a 1.25 M stock solution in flask “S” to a 25.00-mL volumetric...
A: In the given problem, the molarity of the serially diluted solution can be calculated by using the r...
Q: What is the mechanism for this reaction?
A:
Q: Chemistry Question
A:
Q: A sugar crystal contains approximately 1.8x1017 sucrose (C12H22O11) molecules. What is its mass in m...
A:
Q: How can we separate the components of a compound?
A: Compound refers to those substances that are formed by the combination of elements in a certain spec...
Q: Consider a solution that initially is 0.050 M with respect to Ag(NH3)2+. The Kf of the complex ion i...
A: Hi, since there are multiple sub-parts posted we will provide you with answers for the first three s...
Q: Propose a mechanism to account for this acid-catalyzed dehydration. OH H,SO, + H,O heat (racemic)
A:
Q: a piece of metal weighing 10.0 g at an initial temperature of 87.3 °C was placed into 100.0 g of wat...
A:
Q: What is [OH-] in a solution with [H3O+] = 2.0x10-4M
A: The relationship between pH and pOH is given by pH + pOH = 14
Q: Solutions lons pH meter Water + 7.00 но", он 3 Strong acid А , н + 2.00 Weak acid + 4.50 A , H 0 3 S...
A: Since pH = -log[H+ ] = -log[H3O+ ] where [H+ ] = concentration of H+ ions [H3O+ ] = concentration o...
Q: Propose a mechanism for this reaction. CICH,CH,OH Na,CO, H,0 H,C-CH,
A: Given reaction,
Q: Suppose you were told that each reaction is a substitution reaction, but you were not told the mech...
A: Given:
Q: A buffer solution is made that is 0.307 M in H2CO3 and 0.307 M in KHCO3 . If Ka for H2CO3 is 4.20x10...
A: Given concentration of H2CO3 is 0.307 M and concentration of KHCO3 is 0.307 M. Acid dissociation co...
Q: d. Provide the overall balanced equation for this galvanic cell. e. Write the line notation for the ...
A: Given:
Q: Chemistry Question
A: The given compounds are represented as follows:
Q: Carbonic acid, H2CO3, can be found in a wide variety of body fluids (from dissolved CO2). (a) Calcul...
A: The ionization of carbonic acid is as follows, a) The first dissociation constant is represented as...
Q: (b) Using appropriate chemical reactions for illustration, show how calcium present as...
A: chemical reactions for illustration, calcium present as the dissolved HCO3- salt in water is easier...
Q: Write the formula for ionic compound. Rubidium hydrogen sulfate
A: The given ionic compound name is Rubidium hydrogen sulfate
Q: Water, Sulfate ion, & Ammonium
A: Compound H2O: H atom is a group 1 element has 1 valence electron and O atom is a group 16 element ha...
Q: Ammonium iodide dissociates reversibly to ammonia and hydrogen iodide. NH4I(s) ⇌ NH3(g) + HI(g) At 4...
A:
Select the member of each pair that undergoes SN1 solvolysis in aqueous ethanol
more rapidly
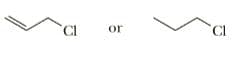
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images


