Q: A chemistry graduate student is studying the rate of this reaction: NH,OH (aq) → NH, (aq)+H,O(aq) He…
A: The concentration-time data for a reaction is given.
Q: MW is 98 1HNMR: 1.7 quintet, 1.8 quintet, 2.3 triplet What molecule is this?
A:
Q: Which product is formed as a result of the decarboxylation of the following molecule?
A: Decarboxylation- Removal of CO2 on heating or in the presence of NaOH+CaO (soda lime) from…
Q: The vapor pressure of diethyl ether (ether) is 463.6 mm Hg at 25 °C. How many grams of estrogen…
A:
Q: c) i) d) e) a) h) This reaction was monitored for 2000 seconds starting with an initial H₂O2…
A: The concentration-time data of decomposition of H2O2 reaction is given.
Q: E.g. 1 R 4-C-OH I с E One H+ Formic acid, HCHO2, is a monoprotic acid. In a 0.100 M solution of…
A:
Q: NOBrNO+ Br₂ ½½/2 is second order in NOBr. In one experiment, when the initial concentration of NOBr…
A: In second order reaction sum of exponent in corresponding rate law of reactions is equal to 2
Q: 6. For the following molecule, provide the number of peaks you expect in each ¹H NMR signal due to…
A: NMR spectroscopy is an analytical technique that is used in structural elucidation of a compound.…
Q: 9. Give two names for each of the following: CH3CH₂ H₂C a. CH3CH2 C-CHCHCH3 CH3 b. CH3 C-CHCH₂CH3
A: Here we can see that we have an epoxide group as a…
Q: 3 As a pharmacist you are given ZnCl₂ (0.7%) Phenylephrine (0.1%) and Boric acid (1.1%) with E value…
A: When comparing a solution's solute concentration to that of the reference solution, which is often…
Q: 2 molecules of A come together to form an AA dimer. The rate of binding is slightly less than…
A: Dimerization of molecule ARate of forward reaction, i.e Binding or dimerization A-A= 2X106…
Q: Part E. Label the following molecule as R- or S- configuration H H3C CH3 C Br Н CH₂OH H НО II. CH3 Н…
A: For the chiral carbon i.e. the carbon atom having four different groups attached to itself.If the…
Q: 6. What alkene should be used to synthesize the following alkyl bromides? a) b) H3C H3C Br CH 3 CH3…
A:
Q: Arrange the following compounds in order of decreasing reactivity for a nucleophilic substitution…
A: Order pf reactivity towards nucleophile
Q: A 0.02 N HCI solution is titrated with Na2CO3 (106 g/mol) and 20 mL are spent in the titration. How…
A:
Q: Org. Chemistry - CANNOT be hand-drawn! 2,6-dimethyloct-2-ene Functional Groups illustrate the…
A: A question based on introduction to organic chemistry. The question is to be answered based on…
Q: Draw the organic product you would expect to isolate from the nucleophilic substitution reaction…
A: Tertiary alkyl halide undergo SN1 substitution reaction.Primary alkyl halide undergo SN2…
Q: 8. Draw the ions in the mass spectrum of methylcyclohexane that correspond to the m/z peaks of 98…
A: The mass spectra of a compound gives the information of different possible fragments present in the…
Q: 12.30 What diene and dienophile are needed to prepare each compound by a Diels-Alder reaction?
A: In Diels-Alder reaction, a dienophile reacts with a diene to yield a cyclic adduct. The reaction…
Q: What is the pH of a 0.350 M solution of lithium citrate? (K, Citric Acid = 7.4 x 104)
A: [lithium citrate] = c = 0.350 MKa = 7.4 x 10-4
Q: Please type out your answer, cannot be hand-drawn
A: Enantiomers: Enantiomers are non-superimposable mirror images of each other. They possess the same…
Q: Which of the following factors primarily dictates the distribution of terrestrial biomes on Earth?…
A: Climate factors like temperature and precipitation are the main determinants of where terrestrial…
Q: Pick the IUPAC name of the moelucle shown below. H H₂CO H O 4-methoxy-2.3 epoxyhexane O2.3…
A: The IUPAC is used to determine the name of the compound based on different functional group present…
Q: A student separates colored compounds A and B (shown in grey in the figure below) using column…
A: A question based on analytical process. Based on the Column chromatography and the polarity index,…
Q: 43.8 mL of 0.151 M NaOH was needed to neutralize 50.0 mL of an H2SO4 solution. What is the…
A:
Q: CH₂CH₂CH₂CH₂ CH,CCH₂CH, I CH₂CH₂CH₂
A:
Q: Calculate the ΔG for the vaporisation of mercury at 24oC using ΔH = 61.0 kJ mol -1and ΔS = 89.0 J…
A:
Q: Calculate the solubility of PbF2 in water at 25 °C. You'll find K sp Round your answer to 2…
A:
Q: Write the molecular, total ionic, and net ionic equation for the following precipitation reaction of…
A: We have to write the molecular, ionic and net ionic equation for the given reaction.
Q: 10 Thu €13 c13dec 5 13C NMR (red coupled, blue decoupled) 200 8 (ppm) 100 8 (ppm) dable 60 IR…
A: The 1H spectrum of the molecule with MW weight =148The chemical shifts are given as1.9ppm doublet,…
Q: A standard of solution was put through appropriate dilutions to give the concentrations of glucose…
A: Straight line equation can be written as:traight line equation can be written as:Using Linear…
Q: Regarding the solvent eluent used for TLC analysis, which of the following are correct? (select all…
A: Given Solvent eluent used TLC
Q: 7) 4.43 Identify each of the following disubstituted cyclohexanes as either a cis or a trans isomer.…
A: “Since you have posted multiple questions with multiple sub parts, we will provide the solution only…
Q: Using the balanced equation: 2C₂H6S(g) +902(g) → 4CO2(g) +2SO₂(g) + 6H₂O(g) and the diagram below,…
A: From the given diagram:The number of units of C2H6S = 3The number of units of O2 = 9
Q: Use the following information to answer the questions. Electroplating is a widely used process. One…
A: The number of moles of a compound deposited is calculated by the charge deposited during the…
Q: 4) What is the relationship between the structures shown below? CH₂Br CH3 H and A) not isomers B)…
A: ->Isomers are compounds having same molecular formula.-> If spatial arrangement different then…
Q: Determine the oxidation state and d configuration of the iridium metals and the bond order for the…
A: Iridium belongs to group-9 of the d-block in the periodic table. Electron configuration : [Xe] 4f14…
Q: Which of the following best describes the experimental conditions under which an alkane can be…
A: Halogenation of alkanes such as Chlorination and Bromination takes place via free radical mechanism…
Q: Ideal solution composition of A and B liquids at 80°C is 100 (for A) and 600 (for B) mmHg,…
A: Composition of solutionLiquid A, PA = 100 mmHgLiquid B, PB = 600 mmHg
Q: IN What is the IUPAC name of the following compound? NH₂ Submit Answer indes Q Search BOLES [Review…
A: To write IUPAC Nomenclature of a given organic compound, first we select the longest carbon chain .…
Q: Consider the bonding between K* and O². The type of bonding that exists between these entities is:…
A: K has atomic number 19 means it has 19 electrons its electronic configuration is 1s22s22p63s23p6…
Q: Consider the following equilibrium: 2NH₂ (g) N₂(g) + 3H₂(g) AG = 34. kJ Now suppose a reaction…
A: Given reaction is the formation reaction of Ammonia and we have to find out the direction of…
Q: A certain metal M forms a soluble sulfate salt MsO4. Suppose the left half 4' and cell of a galvanic…
A: Left electrode will be positive because at left electrode reduction will takes place and in galvanic…
Q: is it appropriate to use an air condenser in this experiment?
A: The appropriate reason for using an air condenser in this experiment is to prevent water from…
Q: 16.At the midpoint of a titration curve for valine A. The concentration of a conjugate base is equal…
A: At the midpoint of a titration curve for valine:A. The concentration of a conjugate base is equal to…
Q: Consider the molecule shown below. a. Which of the two nitrogens is more basic (1 or 2) b. What…
A: Based on number of bonding and lone pair of electrons, it hybridization will change. In case, around…
Q: 11. A certain reaction has an activation energy of E₂ = 7.0 kJ/mol. The rate of this reaction at…
A: it lowers the activation energy required for the reaction to occur. This leads to an increase in the…
Q: Would you expect a solution of ammonium chloride (NH CI) to be acidic or basic when added to water?…
A: When NH4Cl is hydrolysis with H20 it gives NH4OH and H+Here actually products of hydrolysis are…
Q: Identify the product(s) of the following reaction: bf H₂O
A:
Q: What mass of Cu(s) can be formed from 2.00 g of Cu2O(s) and 2.00 g of H2(g) by the following…
A:
C2.
Subject:- Chemistry
please identify the chiral centers and determine the sterechemistry of a compound that contains a cycolhexane
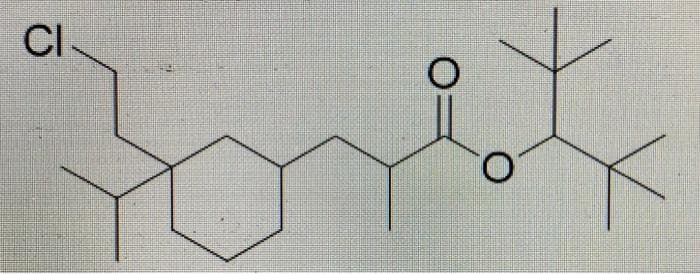
Step by step
Solved in 3 steps with 1 images


