Q: It is believed that two carbon-12 nuclei can react in the core of a supergiant star to form…
A: Two carbon-12 nuclei reacts with each other to form sodium-23 and hydrogen-1 . We have to calculate…
Q: Isomerism and Stereochemistry A. Identify the configuration of the double bond. B. Redraw the…
A: Given structure is : A. Identify the configuration of the double bond. B. Redraw the structure and…
Q: ČI
A:
Q: E°cell = +1.47 V for the galvanic cell V(s) | V2+(1 M) || Cu2+(1 M) | Cu(s) Determine the value of…
A:
Q: Without reference to any data tables, choose which member of following pairs has the lesser…
A: (27) H2O(l) or H2O(s) B has the lesser amount of entropy.
Q: Mass of aspirin used is .090 g the absorbance for the final solution was .304 nm Slope is 1708…
A: Given: Mass of aspirin used = 0.090 g The absorbance for the final solution was = 0.304 Slope is…
Q: Draw the predominant form of this amino acid at pH = 12.
A: Calculate the isoelectric point (pl) for the given amino acid At pI the amino acid exist as…
Q: Calculate the pH of a 0.500 M aqueous solution of NH3. The Kp of NH3 is 1.77 x 10-5 O 8.95 O 11.47 O…
A: Given: Molarity of aqueous solution of NH3 = 0.500 M Kb of NH3 = 1.77×10-5
Q: Write the simplest formula of the oxide. Write the balanced equation for the reaction. Include the…
A:
Q: these two reactions: Reaction 1: 2OF2(g)⟶O2(g)+2 F2(g)ΔH=−49.4 kJ Reaction 2:…
A: ->A reaction is spontaneous if ∆G<0 ->A reaction is non spontaneous if ∆G>0
Q: (c) Predict the product(s) for the following reaction with supporting mechanism. i) SeO, Aq. dioxane…
A: This ragents are oxidizing agents.Oxidation takes place with DDQ and SeO2.
Q: Neglecting any effects caused by volume changes, what would you expect about the ionic strength of…
A: Solution- Ionic strength- The total of the molalities of each type of ion present multiplied by the…
Q: final volume
A:
Q: Which one of the following is the weakest acid? O HF (Ka = 6.8 × 10-4) HCIO (Ka = 3.0 x 10-8) O HNO2…
A: Lower the value of acid dissociation constant (Ka), lower the concentration of H+ ions. Hence, we…
Q: Calculate the pH of a solution that is (This problem requires values in your textbook's specific use…
A:
Q: t functional group.
A:
Q: Apply the Bronsted theory of acids and bases to the following reaction: N3-(aq) + H2O( l ) ⇆ HN3(aq)…
A: We have to predict the correct option as per bronsted acid base theory.
Q: Kiko, a Chem 31.1 student, accidentally shuffled two test tubes with different compounds on his way…
A: We have to determine the compounds A and B.
Q: Write TRUE if the BOLD word/phrase makes the statement correct. Otherwise, write the correct…
A: KMnO4 is strong oxidizing agent and it is purple in color. It can oxidize alcohols to carboxylic…
Q: (b) What is E-factor?
A: E - factor is used in Green chemistry which is a ratio of total mass of waste and mass of product.…
Q: Give the major product that is formed when the primary hydroxyl group of the following…
A: We have to predict the major product.
Q: Which of the following is not a theory for the process of sweetness? a. AH-BX b. GBX c.…
A: To explain the chemistry behind the process of sweetness, there have been various theories…
Q: (a) One amino acid that is likely to have a net negative charge at pH 8. (b) One amino acid that may…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: 3. (Stoichiometry + LR/ER) The thermite reaction, used for welding iron, is the reaction of Fe;O4…
A:
Q: 7. The heat capacity of N; as a function of temperature is shown below: Cp.m 28.883 -0.157T +0.808T…
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for…
Q: (c) Complete the following reactions. CHO „NO2 в CHO C Br 25°C NH D + E 90°C 合
A:
Q: Which of the following could DECREASE the amount of the recovered crystals in the purification by…
A:
Q: Thermodynamics and Catalysis Part B- Arrange the reaction energy diagrams based upon the rates of…
A: Activation energy: When the reactant molecules convert into the products, they required the minimum…
Q: It is suggested that the decomposition of nitrogen pentoxide dissolved in an organic solvent is a…
A: A question based on kinetics of reaction in solution that is to be accomplished.
Q: The diasteromers of 4-t-butylcyclohexanol that you make can be distinguished by the splitting…
A: Given that The diasteromers of 4-t-butylcyclohexanol We have to distinguish by the splitting…
Q: ए नैमट म्रभ (लाCOTकनीना ड ड.25K10-4M the abseved the cett potentiat at जीाट विडव्ण्ववं जेकए 298 k…
A: The step wise solution is as follows -
Q: How many grams of Kr are in a 3.54 L cylinder at 58.5 °C and 5.22 atm? mass:
A:
Q: CH3 5. Which type of spectroscopy (IR, 'H NMR, and/or "C NMR) would be good to distinguish between…
A: The best spectroscopic methods to differentiate between these two compounds are 1H and 13C NMR,…
Q: 1. Examine Model 1. a. Write the theoretical chemical reaction that is used in the trials of Model…
A:
Q: 8. (Calorimetry - Determination of specific heat) A 5.1-gram piece of gold jewelry is removed from…
A:
Q: Question 2 23.79g of solid sodium hydroxide, NAOH is added to 87.5 ml of a 1.97M solution of…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: (a) Arrange the following compounds according to their reactivity towards electrophile. Justify your…
A: We must know that more aromatic a compound is, more stable it will be and thus less reactive. A…
Q: What ion would stronium replace in hydroxylapatite?
A: The effect of partial substitution of Ca2+ in hydroxyapatite by Sr2+ on bone mineral content (BMC)…
Q: The structure below is a ____________. cerebroside monoglycosyl ceramide…
A: Cerebroside is a glycosphingolipid found in muscle cells.
Q: Calculate ΔmixG and Δmix S for the formation of air containing 1 mol of gas by mixing nitrogen and…
A:
Q: A mixture of 8.05 moles of A, 10.36 moles of B, and 27.16 moles of C is placed in a one-liter…
A:
Q: The Ka of acetic acid (HC2H302) is 1.8 x 10-5. What is the pOH at 25.0 °C of an aqueous solution…
A:
Q: The graph below is a potential energy diagram for the hypothetical reaction: + B → C + D 100 90 80…
A: ->∆H = H(product)-H(reactant) If ∆H>0 => Endothermic If ∆H<0 => Exothermic ->An…
Q: Which of the following compounds is not an amino acid?
A:
Q: Exercise 28. Competing SN and E reactions. Predict whether the following reactions will proceed via…
A: In E2 elimination reaction leaving group and proton must be antiperiplanar to each other...
Q: The pH of a 0.55 M aqueous solution of hypobromous acid, HB1O, at 25.0 °C is 4.48. What is the…
A: The percentage of ionization of HBrO can be calculated as follows
Q: What are the molar concentrations of acetic acid (CH3COOH) and sodium acetate (CH3COONa) in an…
A:
Q: BlOHle OH Urea, air
A:
Q: 4. (a) Complete the following reactions and provide mechanism of the reactions involved. -CO2Et CN…
A:
Q: A 1.0 x 10-2 M aqueous solution of Ca(OH)2 at 25.0 °C has a pOH of O 12.30 1.70 O 2.00 O 7.00 12.00
A: pOH of Ca(OH)2 can be calculated as follows
Redraw the structure of the given compound with all the chiral centers in R configuration.
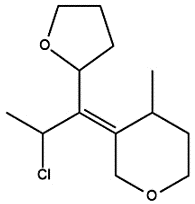
Step by step
Solved in 2 steps with 1 images


