Q: The decomposition of N,O, follows first-order kinetics based on the reaction below. 2N,O,(g) → O,g) ...
A: Given: The rate constant K is 3.46×10-5/s.
Q: The isotope 226Ra has a half-life for the radioactive decay of 1600 years. How long will it take the...
A:
Q: Consider two aqueous solutions of nitrous acid (HNO,). Solution A has a concentration of [HNO-] = 0....
A: Percent of ionization depends on the concentration of acid. It increases with dilution.
Q: 17. What is a family or group of elements?
A: A family or group of elements has to be defined.
Q: Draw the structures of the fi ve constitutional isomers of molecular formula C 5H 10 that contain a ...
A: The structures of the five constitutional isomers of molecular formula C5H10 that contain a double b...
Q: A chemical reaction in a bomb calorimeter evolves 3.15 kJ of heat. If the temperature of the calorim...
A: From given data q = 3.15 kJ = 3150 J T1= 19.19°C = 292.19 K T2= 22.03°C = 295.03 K Formula used C=...
Q: 14
A: Answer :- Below I have explained step by step solution Hope this will really gonna help you !!!!! ...
Q: i. Make the definition of "molality", "molarity" "mole fraction". Explain how we could calculate the...
A: Density = mass / Volume Number of moles = mass / molar mass
Q: Nitric acid HNO3 is ___________________________ in water. Question options: inso...
A: Nitric acid (HNO3) is a strong mineral acid. It dissociates completely into hydrogen ion and nitrate...
Q: Calculate AS° for C3H4(g) + 2 H2(g) → C3H8(g). Substance S° (J/mol·K) C3H4 (g) 266.9 H2 (g) 130.6 C3...
A: # We can make use of the standard entropy of each of reactants and products to calculate the change...
Q: Use the values provided below to calculate the predicted isoelectric point, pl, value for aspartame ...
A: Isoelectric point: Isoelectric point of a amino acid is the pH at which amino acid has a neutral ch...
Q: 6. Visualize a substance with the following points on the phase diagram: a triple point at 0.5 atm a...
A: Since you have posted a question with multiple sub-parts, we will solve first three subparts for you...
Q: Determine the geometry about each interior atom in each mol- ecule and sketch the molecule. (Skeleta...
A: Determine the geometry of each interior atom in each molecule and sketch the molecule. (a) CH3OH (H3...
Q: DThe ing Ig of bacy mass. Calculate thle dose in milligrams fo a 155 lb person. (1 15-453.599) recom...
A: (1) Following information given in the question - Recommended dose of drug = 8.00 mg/kg Mass of the ...
Q: Identify the functional groups present in each of the following molecules: (a) methionine CH3SCH2CH2...
A: Functional groups affect the chemical properties of a molecule.
Q: A rigid 8.20 L flask contains a mixture of H: gas, O; gas, and Ar gas. Within the flask, there are 2...
A: With the help of Ideal gas equation and partial pressure calculate the answer
Q: Which molecule has polar bonds but is non-polar? SF4 BF3 NH3 H2O
A: 1. Molecule is polar if resultant dipolemoment is not zero. 2. Bond is polar if two electronegative ...
Q: When spheres of radius r are packed in a body-centered cubic arrangement, they occupy 68.0% of the a...
A: Volume of the sphere is given by, VS=43πr3 where r is the radius of the atom. Volume of the unit cel...
Q: Sodium biphosphate, used in medicine as a saline laxative prior to colonoscopy and as a stabilizer i...
A: NaOH + H3PO4 ------->
Q: What is the identity of L? CrO42- AsO43- PO43- Hg2+
A: Qualitative inorganic analysis involves the detection of the anions and cations present in acompound...
Q: How can the Tyndall effect be used to distinguishbetween a colloid and a solution? Why?
A: The scattering of the light by the particles that are suspended in the medium is called as Tyndall e...
Q: A naturally occurring sample of an element contains only twoisotopes. The first isotope has a mass o...
A: % of first isotope = 60.11 % Mass of first isotope = 68.9255 amu Mass of second isotope = 70.9247 am...
Q: Each of the following substances is a liquid at -50°C. Place these liquids in order of increasing va...
A: Vapor pressure is a measure of the tendency of a material to change into the gaseous or vapor state,...
Q: N O,→2NDE * ½ Oz is . 2 Ng Og = NOg t NO, 3. beo NOtO,t NOz NO,+ NO3 NO t NOg 2. ky >2ND2 Appy stead...
A: Rate of reaction represents the change of concentration of a reactant or a product with respect to t...
Q: At room temperature, table sugar has a solubility of 200. g in 100 mL of water. In boiling water, so...
A: Saturated solution is the one when a certain amount of solute dissolved in a solvent. Unsaturated so...
Q: At 20 g block of copper is in contact with a 100 g block of iron. After an extended time, the blocks...
A: Given: Mass of copper, mc=20 g Temperature change, ∆T=(T-400)°C Specific heat, Cc=0.384 J/g ∙ °C Mas...
Q: Identify the correct electron domain geometry for XeF4
A: To find: The correct electron domain geometry for XeF4
Q: A gas mixture contains one-third methane by volume (recall what that means in terms of mole%) and th...
A:
Q: Write the following numbers in standard notation. Use a terminal decimal point when needed:(a) 6.500...
A: Hello. Since the question contains more than three sub-parts, the first three sub-parts shall be onl...
Q: Write structural formulas for all the constitutionally isomeric alcohols of molecular formula C5H12O...
A: To find: structural formulas for all the constitutionally isomeric alcohols of molecular formula C5H...
Q: o chem - please show stereochemistry and break down each step as much as possible
A: In this reaction, as a tertiary carbocation is formed, it will follow SN1 reaction pathway.
Q: When a solution is diluted, the concentration of the solution increases. Question options: ...
A: We know that Dilution refers to the process of adding additional solvent to a solution to decrease i...
Q: What volume of 0.300M H2SO4 is required to exactly neutralize 200mL of 0.500M NaOH? 2NaOH + H2SO4 ( ...
A: Given: Molarity of H2SO4 M1 = 0.300M Molarity of NaOH M2= 0.500M Volume of NaOH V2= 200 mL To find: ...
Q: How many kilojoules per molof ammonium nitrate are released when acold pack is activated?
A: A cold pack is a device that consists of two bags, one containing water, inside a bag containing am...
Q: Describe how molecular formulas and structural formulas areused to represent the compositions of mol...
A: Molecular formulas and structural formulas are used to represent the compositions of molecules that ...
Q: Assign E or Z for each double bond in the following: H3C CO2H CH2CI CH3
A: Alkene of the type abC=Cxy (a, b, x and y are substituents) show geometrical isomerism but it cannot...
Q: Chemistry Question
A: Dear student I have given answer to your question in the image format.
Q: The Kf for the formation of the B(CN)42- is 7.4 × 1039. Calculate the concentration of B2+ at equili...
A: The reaction for the formation of the complex is: B2+ + 4CN- → BCN42-
Q: I keep getting this wrong
A: Since you have asked multiple questions, we will solve the first question for you. If you want any s...
Q: An organic liquid is either methyl alcoholethyl alcohol or a mixture of the two.A 0.220 g sample of ...
A: The combustion reaction of methyl alcohol and ethyl alcohol:
Q: Calculate the percent by mass of a solution made from 12 g NaCl (the solute) and 64 g water.
A: Interpretation - The percent by mass of a solution made from 12 g NaCl (the solute) and 64 g water i...
Q: Consider the following system at equilibrium at 698 K:H2(g) + I2(g) 2HI(g)When some HI(g) is added t...
A: We have Le-chatelier principle as per which every system tends to undo the effect imposed on it to r...
Q: What is a Fischer projection?
A: Any organic compound can be represented in two dimensions and in three dimensions all well. Th...
Q: A 1.25-g gas sample occupies 663 mL at 25°C and 1.00 atm. Whatis the molar mass of the gas?a) 0.258 ...
A: Given details, P = 1 atm V = 0.663 L T = 298 K R = 0.0821 Latm/molK To calculate the molar mass of g...
Q: Answer ONLY number 2 (letter a, b and c )
A: 2. C3H8 (g) + 3 H2O (l) -----> 3CO (g) + 7 H2 (g) Given : Rate of formatio...
Q: The two hydrophilic monomers shown below can be copolymerized in an alternating fashion in aqueous s...
A: Radical reactions are initiated by radical chain initiator such as tert-butyl hydroperoxide, AIBN et...
Q: Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free en...
A: The relationship between the potential of an electrochemical cell and ΔG° is given as- The overall...
Q: How are conversions involved with volume?
A: The quantities which come across during the scientific studies are named as physical quantities. The...
Q: organic chem
A:
Q: Which diene could not participate in a Diels-Alder reaction?
A: The diene molecule must be flexible to adopt the s-cis conformation to involve in the Diels-Alder re...
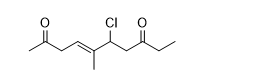
Name the molecule given below its open structure according to the IUPAC system.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images


