Use the values provided below to calculate the predicted isoelectric point, pl, value for aspartame HO. HO. OH, NH2 NH2 phenylalanine aspartic acid pKa1 a-COOH = 1.83 pKa2 a-NH3* = 9.13 pКа1 а-СООН %3D1.88 pKa2 a-NH3* = 9.60 %3D pKa3 side chain-COOH = 3.65
Use the values provided below to calculate the predicted isoelectric point, pl, value for aspartame HO. HO. OH, NH2 NH2 phenylalanine aspartic acid pKa1 a-COOH = 1.83 pKa2 a-NH3* = 9.13 pКа1 а-СООН %3D1.88 pKa2 a-NH3* = 9.60 %3D pKa3 side chain-COOH = 3.65
Chapter26: Biomolecules: Amino Acids, Peptides, And Proteins
Section26.SE: Something Extra
Problem 50AP: The -helical parts of myoglobin and other proteins stop whenever a proline residue is encountered in...
Related questions
Question
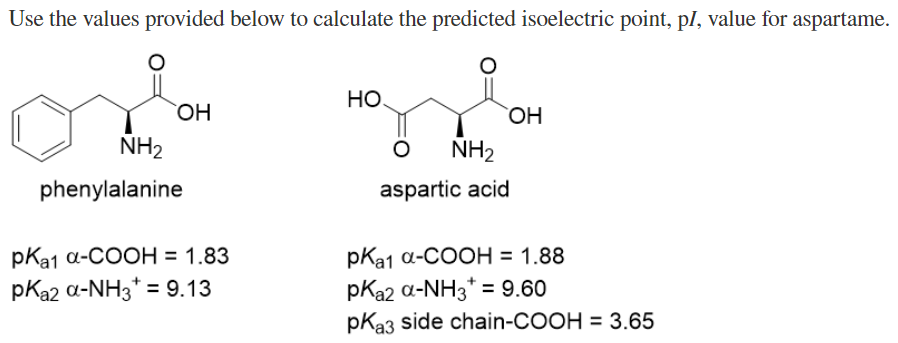
Transcribed Image Text:Use the values provided below to calculate the predicted isoelectric point, pl, value for aspartame.
HO
NH2
NH2
phenylalanine
aspartic acid
pКа1 а-СООН%3D 1.83
pKa2 a-NH3* = 9.13
pКa1 а-СООН %3D 1.88
pKa2 a-NH3* = 9.60
pKa3 side chain-COOH = 3.65
%3D
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning