ck A piece of dry ice (solid carbon dioxide) with a mass of 27.5 g sublimes (converts from solid to gas) into a large balloon. esc trol ~ ^ Mother to Son & pdf 1 7 Q A 2 option Z 2 W S X #1 3 H command 980 23 P E D $ 4 C Assuming that all of the carbon dioxide ends up in the balloon, what is the volume of the balloon at 18°C and a pressure of 756 mmHg ? Express your answer to three significant figures. ▸ View Available Hint(s) ΕΧΕΙ ΑΣΦ R V= Provide Feedback Submit F % 5 V T G 6 4 Ĉ O MacBook Air B Y & 7 H ** N ? . 8 J L 641 F 1 M ( 9 K 149 O < H Y O L d www P command > I www [ ? option I . Next Show All 1
ck A piece of dry ice (solid carbon dioxide) with a mass of 27.5 g sublimes (converts from solid to gas) into a large balloon. esc trol ~ ^ Mother to Son & pdf 1 7 Q A 2 option Z 2 W S X #1 3 H command 980 23 P E D $ 4 C Assuming that all of the carbon dioxide ends up in the balloon, what is the volume of the balloon at 18°C and a pressure of 756 mmHg ? Express your answer to three significant figures. ▸ View Available Hint(s) ΕΧΕΙ ΑΣΦ R V= Provide Feedback Submit F % 5 V T G 6 4 Ĉ O MacBook Air B Y & 7 H ** N ? . 8 J L 641 F 1 M ( 9 K 149 O < H Y O L d www P command > I www [ ? option I . Next Show All 1
Introductory Chemistry: A Foundation
8th Edition
ISBN:9781285199030
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter13: Gases
Section: Chapter Questions
Problem 94QAP
Related questions
Question
Transcribed Image Text:ck
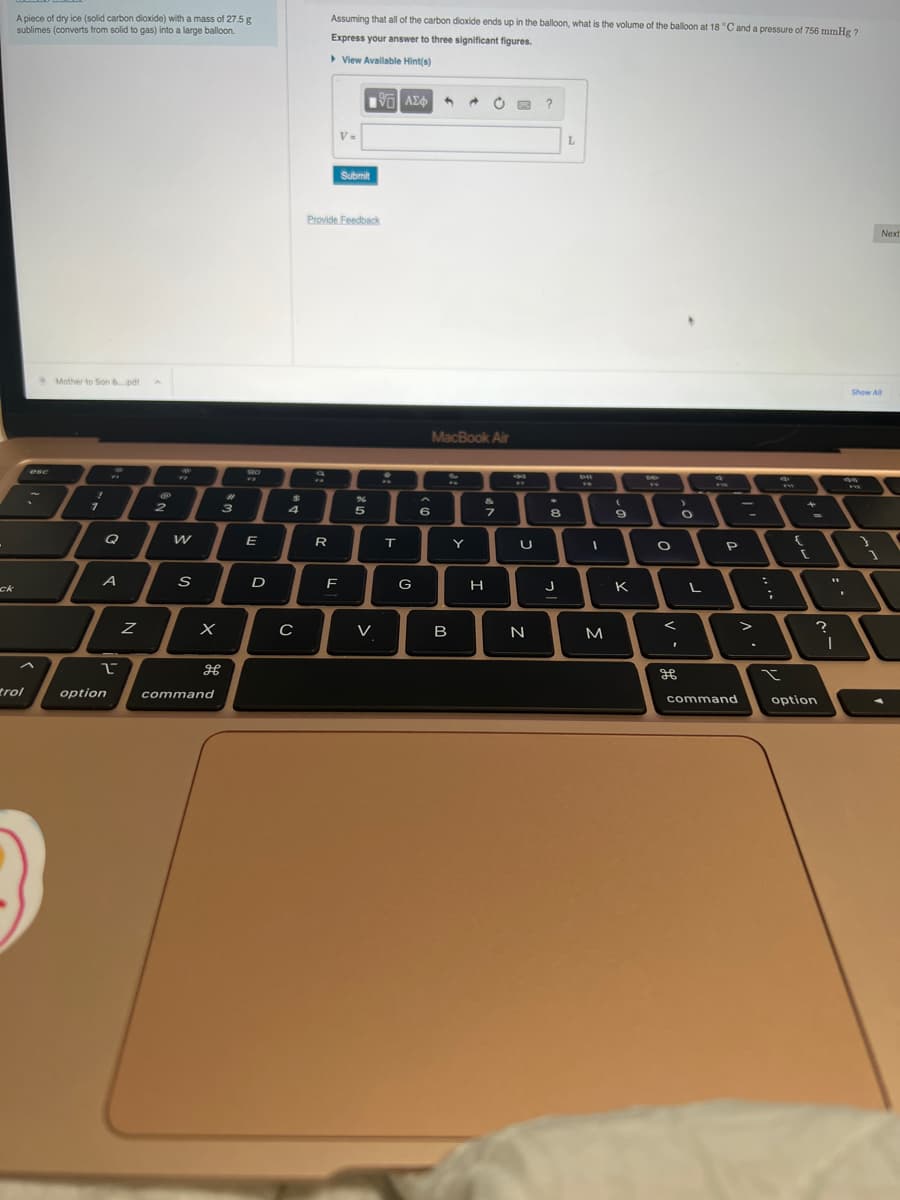
A piece of dry ice (solid carbon dioxide) with a mass of 27.5 g
sublimes (converts from solid to gas) into a large balloon.
trol
esc
"
Mother to Son &...pdf
1
7
Q
A
2
option
Z
2
Ww
S
x
#
3
H
command
880
E
D
$
4
C
Assuming that all of the carbon dioxide ends up in the balloon, what is the volume of the balloon at 18 °C and a pressure of 756 mmHg ?
Express your answer to three significant figures.
▸ View Available Hint(s)
R
V=
F
ΕΧΕΙ ΑΣΦ
Provide Feedback
Submit
%
5
V
T
G
6
4 ^ O
MacBook Air
B
Y
&
7
H
W
U
N
?
8
J
L
6-1
F
1
M
(
9
K
O
<
H
Y
O
L
P
command
>
:
;
I
+
[
option
?
I
39
.
Show All
Next
1
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781285199030
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:
9781285853918
Author:
H. Stephen Stoker
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:
9780534420123
Author:
Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:
Cengage Learning