Q: Rank the elements by effective nuclear charge, Zelf, for a valence electron. Highest Z Lowest Ze Ans...
A:
Q: Answer the following pls
A:
Q: The titration below was performed by placing 10.00 mL solution in an Erlenmeyer flask and titrating ...
A: The given volume of the basic solution present in the Erlenmeyer flask (V1) = 10.00 ml Molarity of t...
Q: this problem because we are going to orally solve virtually later. atleast I will be going to study ...
A: For maximum products , reaction must move towards product side. Means reaction should be Spontaneou...
Q: Write the electron configuration for the following element (short-hand notation is acceptable) Cr
A: Atomic number of Cr ( Chromium ) = 24
Q: The lattice energy of cesium iodide is the energy required for the following reaction. CsI(s) → Cs+(...
A: CsI(s) → Cs+(g) + I−(g) ΔHrxn = ΔHlattice Equation 1: 2 Cs(s) + I2(g) → 2 CsI(s) ΔH1 ...
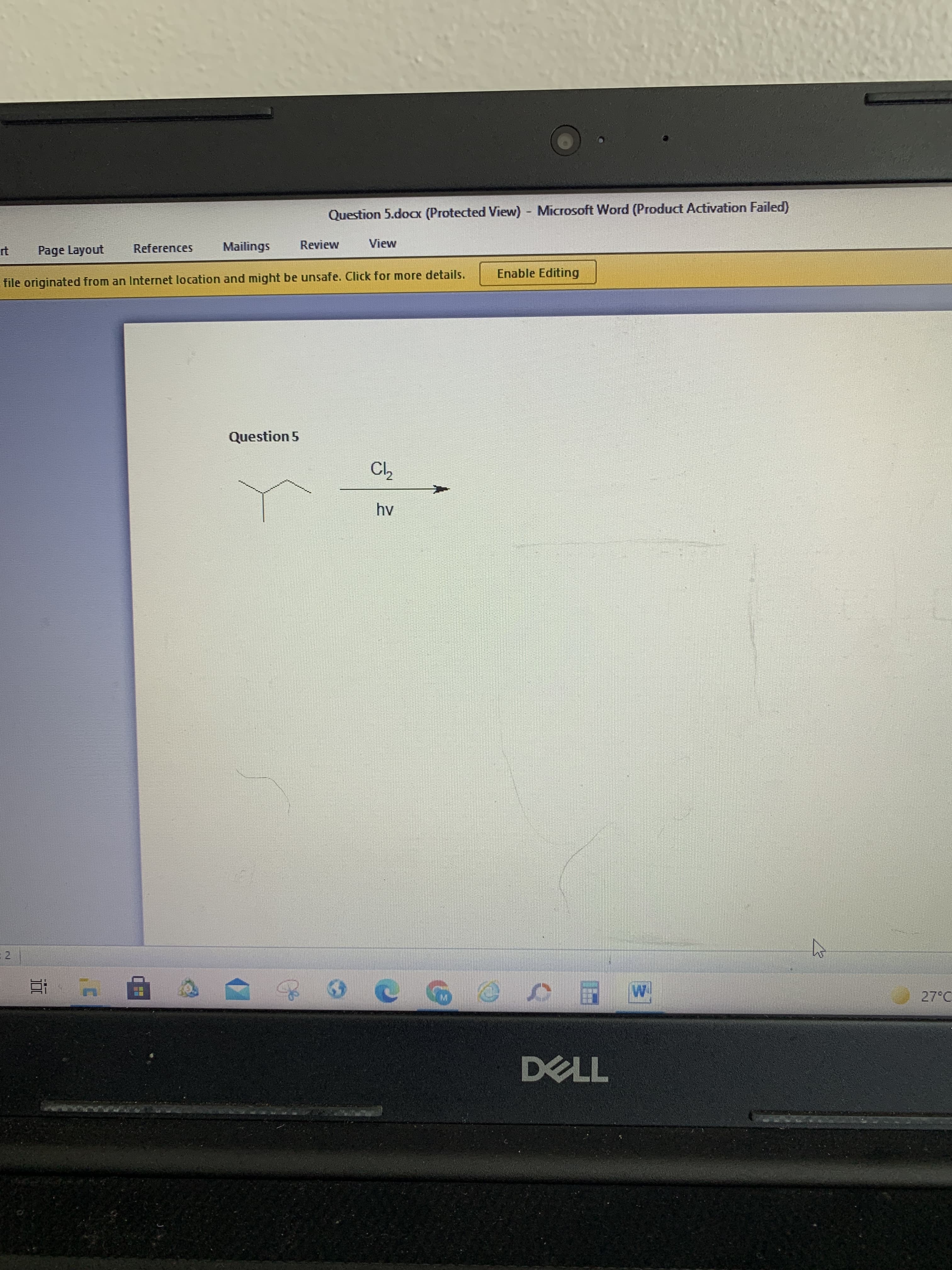
Q: Which of the following is a second chain propagation step in the free radical chlorination of ethane...
A:
Q: Stress - Strain Behaviour Formula A cylindrical metal specimen 15.0 mm in diameter and 150 mm Tensil...
A:
Q: Sorbic acid (HC6H7O2, pKa = 4.77) is widely used in the food industry as a preservative. For exampl...
A:
Q: Write the ground state electron configuration of Ca using the noble-gas shorthand notation. electron...
A:
Q: Choose and explain which element has the greater value for the following properties: i. electron af...
A: 1. F has higher electron affinity than that of Na atom this is because electron affinity increases f...
Q: Solve for the value of x in the following combustion reaction of a particular alkane in the presence...
A:
Q: How much heat (in kJ) is evolved in the complete combustion of 2.043 g C4H10? * 13 C4H10 (g) + 02(g)...
A:
Q: estion 5 of 9 Determine which diagram best represents the ionic compound K, SO, dissolved in water. ...
A: We have given that Determine which diagram best represent the ionic compound K2SO4 dissolved in wat...
Q: Sodium chloride, the table salt (NaCl) is added in cooking to enhance the flavor of the food. When 1...
A:
Q: The following product is expected from the reaction of 3-heptyne with sodium metal in liquid ammonia...
A:
Q: O CH2 CH, CH, CH;-CH-CH, CH3-CH-CH,-CH2 CH=CH, CH, CH,-CH-CH-CH, II II IV V.
A: I is a primary carbocation. It is stabilized by +I effect of one alkyl group and 1 hyperconjugable a...
Q: Why permanganate has 5 peaks in its UV-Visible spectrum between 600-500 nm?
A: Mn is an d block element having d orbitals and ligand oxygen is there and there occurs ligand to me...
Q: For each of the trials I-IV in Part I of the experiment, determine which reactant was limiting and d...
A: Temperature of water = 24oC = 24 + 273.15 K = 297.15 K Atmospheric pressure, Patm= 729 torr Vapour p...
Q: Batteries are charged and discharged due to the concentration of the solutions inside. Calculate the...
A:
Q: 4. Each of the following alkyl bromides gives only one product in via an E2 mechanism. Provide the m...
A:
Q: H H2SO4 heat
A: (3) Alkene on reaction with hydronium ion produces more stable carbonation by the nucleophilic atta...
Q: Select the ionic compound that is insoluble in water. Select one: O A. MgS O B. KBr OC. NazCO; O D. ...
A:
Q: I Review I Constants SSED THIS? Read Section 6.6 ages 228 - 235) ; Watch KCV 6.6, IWE 6.10. volume f...
A:
Q: What are the Ions present in: 1) (NH4)2CO3 2) Zn(OH)2 3)Ba(NO3)2 4) PbS 5) Zn2(PO4)3 6) Hg2I2 7) PbS...
A: We have to predict the ions present in the following ionic compounds 1) (NH4)2CO3 2) Zn(OH)2 3)...
Q: What are the major properties of carboxylic acids? Discuss these properties with respect to followin...
A:
Q: The preparation of the given compound below from acetylene and any other organic and inorganic reage...
A: Acetylene reacts with a base to form an acetylide anion which gives a nucleophilic substitution reac...
Q: CI CH3 но CH=CH, но CH3 (1) H,N CH, H. CH: CH,CH, HO
A: Classify the following compounds as E and Z structures as applicable:
Q: I. Expressions of Concentration 1. Calculate the sodium content, in terms of mEq, of 1 g of ampicill...
A: (1) Given that - Mass of ampicillin sodium = 1 g = 1000 mg Molar mass of ampicillin sodium, C6H18N...
Q: KClO3 in a 0.1862 g sample of an explosive was determined by the reaction with 50.00 mL of Fe2+ 0.01...
A: Given that - Mass of KClO3 sample = 0.1862 g Volume of Fe2+ Solution = 50.00 mL Molarity of Fe2+ ...
Q: The green light emitted by a stoplight has a wavelength of 511 nm. What is the frequency of this pho...
A:
Q: ? lings Review View Help Enable Edr Ss you need to edit, it's safer to stay in Protected View. Quest...
A:
Q: Zaitsev’s rule: states " The alkene formed in greatest amount is the one that corresponds to
A: The major minor alkene as product is determined by Zaitsev’s rule
Q: Examples of Analyzing Chemical Reactions
A: Analyzing Chemical Reactions- A chemical decomposition reaction or analysis reaction is one of the m...
Q: What volume will 0.850 moles of Kr occupy at STP?
A:
Q: Calculate the osmotic pressure in bar units glucose aqueous solution at 25.000C with a concentration...
A: Temperature = 25.00°C ...Or because °C + 273 = k ...So 25.00 °C + 273 = 298 k Concentration = ...
Q: Tris(hydroxymethyl)aminomethane is a common base used to prepare buffers in biochemistry. Its Kb is...
A:
Q: The SDS's have__information sections that conform to the requirements of the GHS of Classification a...
A: MSDS or commonly known as material safety data sheet is a important requirement for every chemical s...
Q: Solve for the problem about titration. A 5.25mL volume of AgNO3 was used to completely precipitate ...
A:
Q: ugati base ionizes entirely, releasing OH- ion, which is a powerful proton acceptor. Expound these: ...
A:
Q: The titration on the right was performed by placing 10.00 mL solution in an Erlenmeyer flask and tit...
A: Indicators - Acid, base indicators are basically weak organic acids that change color when they ion...
Q: What is the major difference in set up between a simple distillation and a fractional distillation? ...
A: Distillation is a separation technique use to separate the liquid mixture on basis of their volatili...
Q: 2H2 CH,CH,CH,C=CCH3 Lindlar Pd Give the structure of the ommited in the reaction|
A:
Q: How much heat is required to warm 10.0 g of ice, initially at-10.0°C, to steam at 110.0°C? (specific...
A: First of all, let's see what values have been given in the question, Mass of ice = 10.0 g Initial te...
Q: The SN2 reaction can be best carried out with A. Primary alkyl halide B. Secondary alkyl halide ...
A: The SN2 reaction can be best carried out with --
Q: Is the reaction a redox reaction; "yes" or "no"
A:
Q: Solve for the value of x in the following combustion reaction of a particular alkane in the presence...
A: The alkane reacts with oxygen to form carbon dioxide and water. The given combustion reaction is as ...
Q: Methanol(CH3OH) has a melting point of -96.7°C and a boiling point of 64.7°C. Its heat of fusion and...
A: We are provided different physical data for methanol.
Q: You are working in my lab. I ask you to make 0.250 L of H3PO4 (density is 1.651 g/cm³). I give you s...
A: Note: Phosphorus triiodide (PI3) actually reacts with water (H2O) to form phosphorous acid, H3PO3 wh...
Q: 20. A gas mixture contains 30.0% CH4 and 70.0% Xe by mass. If the total pressure of this mixture is ...
A: Partial Pressure of a gas in mixture is determined by - P(partial) = Ptotal* mole fraction Mole fra...

Step by step
Solved in 6 steps with 6 images

- Can you make a suggestion for a research topic about disaster risk? or any topic that can be modified and innovated that is still non-exist up to this present. The prototype also is simple and can be made at home.https://www.webassign.net/blb12/a-table-c.pdf ^ | Just copy and paste this in the search bar into googleAccording to Riippi et al. (1998), briefly discuss the effects of increased compression force to the following tablet properties: Disintegration time Surface profiles (as seen on SEM photographs)
- When working with volatile chemicals, such as flammable solvents the best way to control exposures is to: A. Work on the open benchtop as laboratory ventilation is sufficient to remove vaporsB. Work in a chemical exhaust hood (fume hood) as it provides vapor and splash protection when the sash is in the proper positionC. Conduct all of your work outdoorsD. It doesn't really matter where you work because most exposures are insignificantGive typed full explanation not a single word hand written otherwise leave it High-pressure liquid chromatography (HPLC) is a method used in chemistry and biochemistry to purify chemical substances. The pressures used in this procedure range from around 500 kilopascals (500,000 Pa ) to about 60,000 kPa (60,000,000 Pa ). It is often convenient to know the pressure in torr. If an HPLC procedure is running at a pressure of 3.17×108 Pa , what is its running pressure in torr? Express the pressure numerically in torr.Chemistry help asap!
- Is number 11 a true or false statement and why? this is a non graded practice worksheetUse this video to answer the questions: https://www.youtube.com/watch?v=BocfSXdbaU0The goal is to convince an investor to invest in your company so that you can build a factory to make ester compounds. For example, you could make hexyl pentanoate, and sell it for use in perfumes and make the investor money. You must come up with a compound that you can make and sell. You can use your textbooks and theinternet as resources. You can also access the link: https://jameskennedymonash.wordpress.com/2013/12/13/infographic-table-of-estersand-their-smells/ to look at an infographic of the ester compounds responsible for the smell of different fruits, perfumes and flowers. At the end of your research, you must have the compound table filled out
- Imagine printing a graft with a 3D printer using marine-derived collagen. collagen must be isolated first from fish. Afterwards, Tissue Scaffolding should be created with 3D Printing. then Cytotoxicity Test should be done. Cartilage Regeneration in Chondrocyte Culture. This collagen is obtained by using fish waste. for example, fish skins from restaurants or fish waste and skins from factories that produce canned tuna. question: How should methodological considerations/engineering standards be● Relevant engineering standards. Please explain clearly and you can make some commentsWhat is the wedge and dash notation of C2H6O?Issue 1Emulsifying, gelling, stabilizing or thickening agents are controlled in Canada. They must be legally declared in the ingredient lists according to the law. Question 1 options:TrueFakeJustify your answer. Issue 3Several branches of the food industry use pigskin to produce gelatin. Pork gelatin remains the most affordable and widespread way to thicken and stabilize food products. It can be found in cosmetics, candies, marshmallows, yogurt and margarine. TrueFakeJustify your answer. Issue 5There are several alternatives to using animals in the production of gelatin, such as agar-agar made from seaweed, pea or corn starch or pectin from different fruits. However, the action of this gelatin is not as effective as that of animal gelatin. TrueFakeJustify your answer.

