Q: B. Fats and Oils. Consider the triglyceride FAT 1. Is FAT a simple or mixed TAG? CH,O-C–CHJỊCH, CHI…
A: Here we have to predict whether the given fat is simple or mixed fat and different products formed…
Q: ow much heat in kcal must be added to 0.79 kg of water at room temperature (20°C) to raise its…
A: We know that Q = m × c × ∆ T m = mass of water = 0.79 kg c = specific heat of water = 1 K cal / kg…
Q: 314,636.2 Joules is equal to how many calories? 75.2 kcal 75.2 cal 1316440.4 cal 1,316,440.4 kcal
A: 1 cal = 4.184 Joules. Using this unit conversion, we will calculate number of calories in given…
Q: What is the product of the following reaction? + H. НО add slowly
A: Aldol Condensation : When an ketone which has free α-Hydrogens in α-Carbon , which in presence of…
Q: Bicycling 10 miles per hour expends 290 Cal/h. How many chocolate chip cookies that contain 50. Cal…
A: Calculate the total amount of calories required during a 1.5 hr ride. Calories required = 290 cal1…
Q: If C + O2 CO2 + 90kcal H2 + 02 H20+ 60kcal CH4 + 202 - CO2 +2H2O+ 210 kcal Then the possible heat of…
A:
Q: Calculate the number of JOules in product A and product B fat (9) carb(g) protem(g) 3.9 Product A…
A: Given that a question product A and product B Value of product A and Product B is a gram and convert…
Q: СООН NH CI CI diclofenac
A: Soln Beilstein test is given by halo compounds since given compound have Cl group thus it…
Q: H2 Lindlar's Catalyst H.
A: A mixture of Palladium and calcium carbonate (or) barium sulfate poisoned with quinoline is known as…
Q: 5.1.3 Calculate the number of calories in 723.01 J.a) 172.80 cal d) 3025 calb) 172.8 cal e) 0.173…
A: The number of calories in 723.01 J has to be calculated.
Q: The final ratio of the CH3COOH and CH3COOK
A: pKa of acetic acid solution = 6.38The final pH of the buffer solution = 5.79We have to calculate…
Q: Dr. Andrews has some cookies in her office that contain 5.0 g fat, 20.0 carbohdryates, and 2.0 g of…
A: The total energy per serving of cookies in kcal is;
Q: H2 Pd/C H2 Lindlar's catalyst Na NH3
A: Product of the following reactions are
Q: Does toasting bread change the amount of calories in the bread?
A: Toasting of bread: Generally speaking toasting of bread is a chemical change. Addition of heat…
Q: Part A one stalk of celery that heats 205 g water from 23.2 °C to 33.7 °C q = kcal • Part B a waffle…
A:
Q: QUESTION 35 For problems 35-40, answer the questions about the 2-step reaction sequence shown below…
A: Ans Nucleophilic
Q: A) Br₂ Pd/C B) Br₂ C) H₂ reaction. Heat Pd/C Ο.Α. Α OB. B OC.C OD.D OE.E A Moving to another…
A: In presence of heat or sunlight there occur free radical bromination . ->Because free radical…
Q: You have two reactions A+B---> 2C. K1 C----> 1/2A+1/2B. K2 How are K1 and K2 related?
A:
Q: Calculate the number of joules in Product A, and enter your answer in Canvas. Show all work on your…
A: Energy Values for Substances (KJ/g)
Q: 2Na + BaCl2 Ba + 2NaCl is what type of reaction? 55>
A: Types of Reactions:- Combination Reactions - In this type of reaction, two or more elements or…
Q: `H + НО add slowly
A: The given reaction is a base catalyzed crossed aldol condensation reaction, in which two different…
Q: reaction 2H2 + O2 ---> 2H2O estimate heat of reaction in Kcal with these bond dissasociations O-H…
A: During a reaction heat can be absorbed or released. If heat is released during the reaction it is…
Q: pr the balanced equation shown below, how many moles of SOs will react with .7727 moles of CaHsS? he…
A:
Q: 60 F 50 40 kcal/mol 30 20 10 course of rx The products have more potential energy than the reactants…
A: The statement written is true or false ?
Q: Nutrition Facts sevings per container Senving sle 2 pleces pig Use the 4-9-4 Method to sening…
A: Energy of carbohydrates = 4 Cal/g Energy of fat = 9 Cal/g Energy of proteins = 4 Cal/g
Q: Convert -40. kcal/mol to kJ/mol using the fact that 1kcal = 4.184 kJ. Show work using dimensional…
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: MCQ 48: Catalysts speed up reactions but when equilibrium is reached it has A. huge effect B. no…
A: Given options are,
Q: convert 123 J to calories 515 cal 34.2 cal 443 cal 29.4 cal
A: Given 123 J
Q: (-85.8 predicted) 36 kcal (-57.2 predicted) resonance energy energy 21.8 kcal resonance energy -57.4…
A: The question deals with resonance energy which is released during the delocalization of electrons…
Q: Fermentation is a well established technology for conversion of sugar from crops into ethanol.…
A: Ethanol can made by a process known as fermentation. In that process, sugar from plant or crops, is…
Q: H*, H20 OH (а) OH (b) OH (с) (d) (е) no reaction
A: The given reaction is a hydration of alkene to give the corresponding alcohol. The mechanism of this…
Q: Convert -40. kcal/mol to kJ/mol using the fact that 1kcal = 4.184 kJ. Show work using dimensional…
A:
Q: One gram of alcohol provides 7 Calories. Calculate the number of Calories in a 5 fluid ozoz glass…
A: Given : Amount of calories in 1 g alcohol = 7 Cal And amount of alcohol in 5 fluid oz wine = 12.1 g…
Q: oue:-complete ithe given reactions with the mecessaryftarting material or greduct.? Naocts NaoHas…
A:
Q: G A 1 9. 4. 3. Free energy (kcal/mol)
A: Given graph of free enegy To find slowest step.
Q: но МСРВА Но но H30* step 2, step 1
A: mCPBA always reacts with electron rich olefin and forms a epoxide.
Q: classify the following reactions a) addiction b) elimination c) replacement d) rearreglo e)…
A: Since you have posted question with multiple sub-parts, we are entitled to answer the first 3 only.…
Q: Using dimensional analysis, convert 2.5 x 10-9 large calories to erg.
A: Given information: 2.5×10-94.3×10-5 atmosphere
Q: A milk-based dessert contains 15g carbohydrate, 6g fat, and 3g protein per 100g. What is the energy…
A:
Q: Draw the remaining product of the reaction. heat Hс — сн—С—сHz—ҫ—он ČH3
A:
Q: raw the missing reactant for the following reaction 1.(CH3CH2)2CuLi 2.H30*
A: Applying concept of organic synthesis.
Q: IDENTIFY THE NUMBER OF ATOMS (CL, C, H, and F) in the Refrigerant Hexachloroethane
A:
Q: In the reaction: CH4 + 2 O2 → CO2 + 2 H2O The experimental ?H is -213 kcal/mol. How much heat is…
A: Already given that for this reaction heat released during reaction is = -213kcal/mol Hear one mole…
Q: complete the reaction : 2Al + Fe2O3 =
A: The chemical equation between Aluminium and ferric oxide is given in step two.
Q: 9) When 40.0g NaoH and 40.0g tedO4 ané mued and 9)When limetin reacted which As thes limitang…
A: In a chemical reaction, limiting reagent is reactant species that is completely consumed in the…
Q: A system absorbs 1000 kcal of heat while doing 500 kcal of work. Calculate the AEsystem- -500 kcal…
A: Relationship between ∆E , q and w is ∆E = q + w Expression of work (w) : wsystem < 0 if system…
Q: 35. What is the maximum mass of carbohydrate in a 6-oz serving of diet soda that contains less…
A: Given: Average number of Calories for carbohydrate =4.1 Calorie/g
Q: The delta H value for the reaction 2S02 + 02 ------------2SO3 O a. -46.3 kcals b. -46.9 kcals O c.…
A:
Q: A) H2
A: Dear student I have given answer to your question in the image format.
Q: Absorbed from the intestines into the blood, nitrites (NO2-) interact with the hemoglobin of the…
A: The number of moles is a ratio of the mass of a substance to the molar mass of the substance.…

Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- How would the molecular orbitals of cyclopenta-1,3-diene, the carbocation, carbanion, and the radical differ? Drawings would be appreciated! Thank you in advance.Predict the products of halogenations, oxidations, reductions, and cleavages of alkenes, including the orientation (regiochemistry) and the stereochemistry of the reaction.Which is more stable - a phenyl radical, [C6H5]•, or a benzyl radical, [C6H5CH2]• - and why?
- Determine all of the products obtained from the addition of HCl to the 1,3-diene. Once determined, draw a mechanism that accounts for the formation of every product. Then, Identify and account for the formation of the major adduct/or adducts under these conditions assuming that the reaction is conducted under thermodynamic conditions.Provide Examples of dehydrohalogenation of dihalides to afford alkynes ?Predict the products of additions, oxidations, reductions, and cleavages of alkynes, including the orientation of the reaction (regiochemistry) and the stereochemistry.
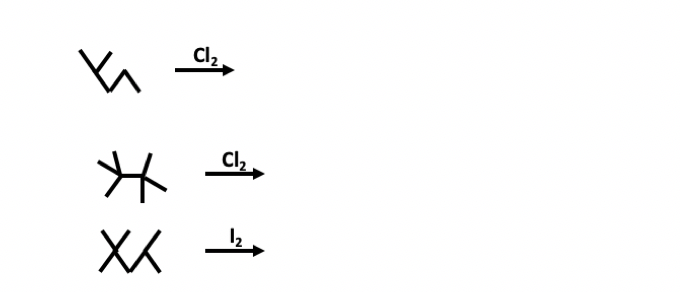
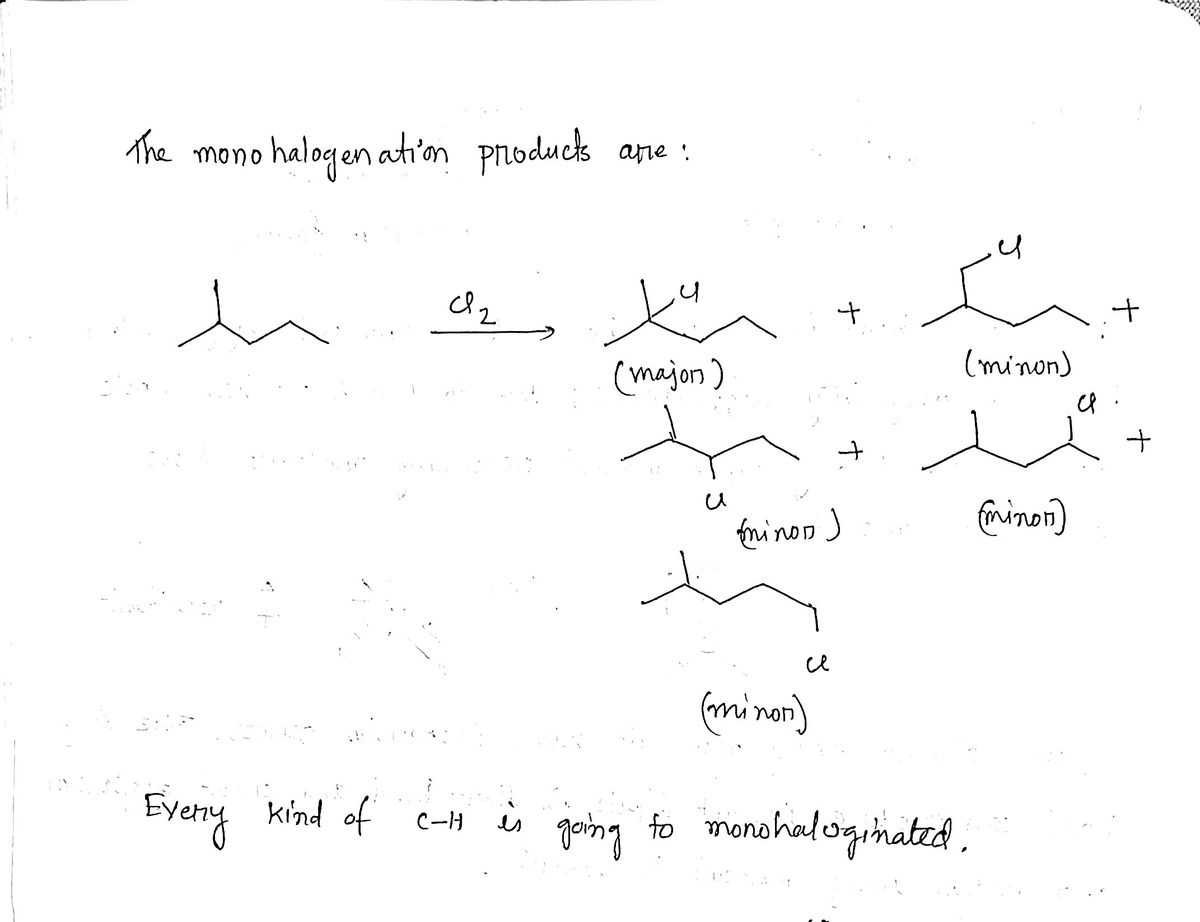
- Free-radical chlorination of hexane gives very poor yields of 1-chlorohexane, while cyclohexane can be converted to chlorocyclohexane in good yield. How do you account for this difference?Treatment of the diene shown with excess ozone followed by a reductive work-up produces.predict the product of each of the following reactions. Mono-halogenated products only for any radical halogenation reactions.
- Predict the product(s) of the following Diels-Alder reaction. Include stereochemistry if appropriate.Predict the product of the following Diels-Alder reaction. Make sure to indicate stereochemistry whereverapplicable.Uemura and coworkers studied a time dependent Diels-Alder reaction which first formed the endo product as the major organic product and with time produced the exo product (J. Org. Chem. 2018, 83, 9300−9304). Show the endo and exo product for the reaction below. Which is the thermodynamic product and which is the kinetic product? Explain your reasoning.





