Q: (а) (b) OH ОН OH or OH or ČI В `NO2 A NO2 ČH3 A B
A:
Q: Select the best answer that numbers the compounds below from most to least acidic [1 = most acidic,…
A: For more acidic in nature , -I effect should be more Polarization of H+ will be more
Q: Referring to the molecule below, which atom is the most acidic H bonded to? * 15 13 но 14 12 11 16…
A: An acid is a compound which dissociate to give hydrogen ion on dissolving in water. Higher the…
Q: Draw the conjugate base for the acid, HC≡CH. Draw all hydrogens. Remember to include charges and…
A: Conjugate base of any acid is formed when the acid donates 1 proton in the solution.
Q: H2C CH2 H2C-
A: The methylene group attached to the electron-withdrawing carbony group is called the active…
Q: complete the equation for the reaction between each Lewis acid-base pair. In each equation, label…
A: INTRODUCTION: Lewis acid is defined as the species that accepts a pair of electrons. Lewis base is…
Q: A. H. H3C- E C- H3C H. B.
A: Acidity of a proton in a molecule depends on various factors like hybridization, Electronegativity,…
Q: ank the indicated atoms from the most (1) to the least (3) basic. ONH NH2 B A
A: We have to identify the order of basicity. More unstable the anion is , more it will be basic.
Q: Which of the following is not an acid
A: Given group of answer choices HBr , HCl , HI , HNO3 are acid because they gives H+ ions in water.
Q: molecule conjugate base of molecule 1 : H. Click and drag to start drawing a structure. H HH H…
A: Lewis structure is a method of of representing bonding pattern in a molecule. it represent how…
Q: Which proton is the most acidic proton in the molecule below? (2) H2 H3C `C `H (4) H2 (1) (3) 3 O 1
A:
Q: HO OH HO
A: Acidic protons leaves the negative part behind. If it is stable the proton will be highly acidic or…
Q: Which indication of relative acid strengths is INCORRECT? Select one: O HCI > HF O H₂SO4 > H₂SO3 O…
A:
Q: Zーエ
A: Piar.1 : Explanation: In both the compounds loan pair on nitrogen atom is responsible for…
Q: Choose the strongest base. OH3C-NH₂ NH₂ H₂C- O NH₂ NO₂ NH₂
A: Solution Correct option is A) H3C-NH2
Q: In the following compounds, tell how many acidic “a-H's" there are in each. do le H. 2.
A: α-H's are the hydrogen atoms bonded to the alpha carbon atom, alpha carbon atoms are the adjacent…
Q: N. DN H-N F E OF
A: When loan pair of nitrogen not involved in delocalization (resonance) then it shows more basic…
Q: 5. Rank the highlighted protons from most acidic to least acidic. А. Ha ÓH most acidic least acidic…
A:
Q: Which of the following is not an acid? Group of answer choices HBr HCl NH3 HI HNO3
A: According to Bronsted -Lowry theory of acids and bases, an acid is a substance which can donate H+…
Q: Referring to the molecule below, which atom is the most acidic H bonded to? C21 C2 O24 O15 C6
A: Acidity of a proton depends on the stability of its conjugate base. Greater is the stability of…
Q: OH OH i) ii) ČH3 NO2 C A B
A:
Q: proton rank Ha на CN OH
A:
Q: What nitrogen base is shown here? NH2 ALureeil ZI
A:
Q: Which arrow is pointing to the most atom with the most acidic proton? A D вс E
A:
Q: ELOH 'BUOH OEt 1 3 4 2.
A:
Q: Select the best answer that numbers the compounds below from most to least acidic [1 = most acidic,…
A:
Q: How many acidic hydrogens are present in the molecule shown below? None 2 4 3 1
A: We have to calculate the number of acidic hydrogen.
Q: Classify these compounds as acid, base, salt, or other. Drag the appropriate items to their…
A: The given compounds can be classified as acid ,base ,salt and others (hydrocarbon) as follows.
Q: Rank the following in order of increasing basicity. The first in your list should be the least…
A: OH- is a conjugate base of H2O. F- is a conjugate base of HF. Cl- is a conjugate base of HCl.
Q: Using the data in the table, which of the conjugate bases below is the strongest base?…
A: The relationship between the Kb of conjugate base and Ka of acid is given by where Kw = ionisation…
Q: Rank the following compounds from most acidic to least. Rank from most acidic to least acidic. To…
A: We have to rearrange the given organic compounds from most acidic to least acidic
Q: HCIO4 + Hz5 O4 )NH3 + H3 PO4 > NH4 +Hz PO 3. CH3 0 t NH3 > CHz OH + NH +HCO,
A: According to bronsted Lowry theory- A conjugate acid base pair are those which differ by only one…
Q: Which of the following acids is the strongest acid? HNO2 Ka = 4.5x10-4…
A: The strongest acid can be identified as Based on Ka values which acid has more Ka value means that…
Q: Which of the following is NOT a Lewis base: O a. CH3SH Ob. NH3 O c. BF3 Od. H₂O
A: we have to select the molecule which is not a Lewis base
Q: Which species are Lewis acids?a. BBr3b. CH3CH2OHc. (CH3)3C+d. Br-
A: A chemical compound or ionic species is considered as Lewis acid only if it is an…
Q: Choose the desired base to react with those molecules below: `NH2 Choose... + СООН Choose.. + OH…
A: Given bases are, NaH KOH NaHCO3 BuLi
Q: For each of the following pairs, predict the strongest acid: H20 or H2S: [ Select] HBr or HI: […
A: H2S is stronger acid than H2O because SH bond is weaker than OH bond so easily dissociates and gives…
Q: Arrange the following ions from the most basic to the least basic. yo (1) (II) (III) (IV) zo
A: Arrange the following ions from the most basic to the least basic .
Q: a. .OH b. C.
A: This is basic concept of the basicity. Here lone pairs of an atom attacks on H+ and gets protonated…
Q: OH (d) OH „NH3 „NH, NH3 (e) O,N H,CO gas phase (f) .N' H.
A: Answer to sub-parts d, e, f
Q: Label each reactant and product in this reaction as a Brønsted acid or base. HCN + NH, CN + NH,…
A:
Q: Chemistry
A:
Q: F H2 .C HO Ho
A:
Q: Which of the following species is the strongest base? Multiple Choice HO H2N" CH3COO cr
A: A solution is said to be strong base , if it easily accepts H+ (protons ) in its solution.
Q: Classify each of the following acids as strong or weak. Drag the appropriate items to their…
A: The classification of acids are given below
Q: hydrogen: CH, (c) NH, OH HO SH HO ot "Is (a) ) NH, OH HO, HO
A: Acidity is define as tendency to release H+ in the solution. When H+ is removed then negative…
Q: E. Choose the More Acidic for Each of the Following Pairs and Explain why! NH4 NH3 +) HO. OH2 CH3…
A:
Q: Write “most" under the most basic compound. Write “least" under the least basic compound. Locts NHz…
A:
Q: 1- Select the one which is neither an acid nor base а) CHCOОН b) НCI c) KCI d) CH3OH
A: Acid is species which donates proton Base is species which accepts proton
Q: Consider the structure of a base. Modify the structure to draw the conjugate acid. Add or delete…
A: An acid is substance which can accept pair of electrons whereas base is substance which can donate…
8
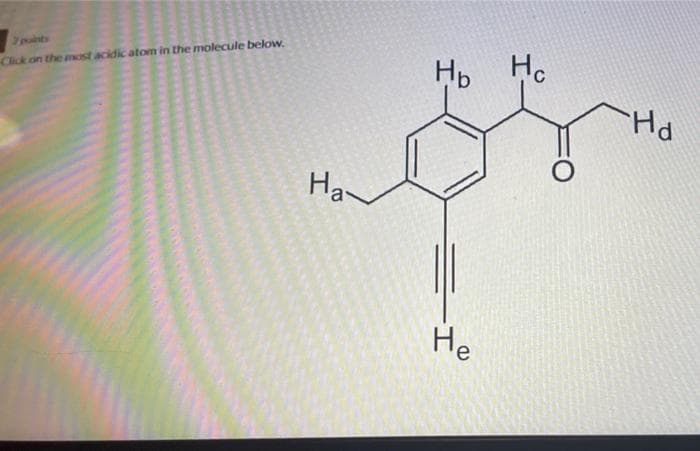
Step by step
Solved in 2 steps with 1 images

- Summarize the relationship between pKa and acid strength by completing the following sentences: a. The higher the pKa of an acid, the stronger or weaker the acid. b. The lower the pKa of an acid, the stronger or weaker the acid.Consider the following bases: a. For each base above, circle the atom/atoms with the highest PE (will release the most P.E.when a lone pair on this atom combines with an H+ ) b. Rank the bases 1 (highest P.E./strongest base) to 7 (lowest PE/weakest base), and explainyour reasoning.Choose the 3 most basic nitrogen atoms in the next molecule. Order them from more basic to least basic like this: __ > ___ > ___

