Q: 1. Which of the following lists contains common heteroatoms found in organic molecules? A) N, O, S, ...
A: Note: According to our guidelines we are supposed to answer only one question
Q: 53. Which atomic orbitals overlap to form the C-H o bonding molecular orbitals of ethylene, H2C=CH2?...
A: It's a multiple question type. The above mentioned questions are explained below :
Q: Which is true about the chemical equation for photosynthesis? A.carbon dioxide is produced B.oxyge...
A:
Q: How many grams of boric acid should be used in compounding the following prescription? Report to the...
A: Ans (0.5/100) ×60= 0.3 g of Proparacaine hydrochloride (2.5/100) ×60= 1.5 g of pilocarpine hydr...
Q: Gaseous ethane (CH,CH, reacts with gaseous oxygen gas (0,) to produce gaseous carbon dioxide (CO,) a...
A: Given : Mass of ethane = 27.66g Mass of Oxygen gas = 141.2g
Q: possible Own be. но, OH a. two b. three c. eight d. nine
A: Number of stereoisomers possible for a compound = 2n where n is number of choral or asymmetric cente...
Q: 31. Rank the following compounds in order of increasing melting point, putting the compound with the...
A: 31. Melting point of a compound depends on the inter-molecular force. Greater is the inter-molecular...
Q: 1.Is the equation balanced? CH4 + O2 --> CO2 + H2O Yes or No
A: Here we have to say that the equation is balanced or not.
Q: 45. Which of the following is the appropriate conversion of (CH3)2CHOCH2CH2CH2OH to a skeletal struc...
A: Appropriate conversation of (CH3)2CHOCH2CH2CH2OH to a skeletal structure = ?
Q: QUESTION 3 EXPLAIN WHAT WOULD HAPPEN IN A DISTILLATION IF A STUDENT DID NOT ADD ANY BOILING STONES T...
A: Boiling Stones:- Boiling stone or boiling chips are tiny porous materials made of alumina, silicon c...
Q: Determine the % of acetic acid (MM = 60.05 g/mol) in a 2.00 mL vinegar sample that has a mass of 1.9...
A: Given: Volume of acetic acid solution = 2.00 mL> Molar mass of acetic acid = 60.05 g/mol. Mass of...
Q: b) Suppose 10.0g of potassium reacts with excess oxygen gas, and 10.5 g of K2O is produced. What is ...
A:
Q: Draw the following structure. 4-sec-Butyl-2-chlorononane cis-1-Bromo-2-ethylcyclopentane
A: Given :- 4-sec-Butyl-2-chlorononane cis-1-Bromo-2-ethylcyclopentane To draw :- Structure of given...
Q: . What is the coefficient of the oxygen molecule in the elephant toothpaste equation? 2 H2O2 --> 2 ...
A: The elephant toothpaste equation is the chemical reaction showing decomposition of H2O2 to H2O and O...
Q: Write the expression for the equilibrium constant Kp for the following reaction. Enclose pressures i...
A:
Q: (1) Identify each of the following half-reactions as either an oxidation half-reaction or a reductio...
A: Oxidation : loss of electrons Reduction : gain of electrons
Q: 20. Rank the following compounds in order of increasing strength of intermolecular forces, putting t...
A: Note: According to our guidelines we are supposed to answer only one question
Q: 40. What is the approximate bond angle for the C-C-N bond in acetonitrile, CH3CN? A) 90° B) 109.5° C...
A: To determine the bond angle between C-C-N , in acetonitrile, we would first draw its shape .
Q: What is the pressure in Pascal (Pa) of 2560 mmHg
A: Change pressure of 2560 mmHg to Pascal ?
Q: I need help with these question please
A: 1 inch = 2.54 cm 1 cm = 10 mm 1 mm = 10-3 m
Q: 25. Which of the following compounds has the highest boiling point? HO OH OH IV A) I B) II C) III D)...
A: Boiling point:The temperature at which the vapor pressure of a liquid becomes equal to the atmospher...
Q: 35. Which of the following concepts can be used to explain the difference in acidity between acetyle...
A: Acidity increases with increase in s character Acidity increases with the group that exerts -I effec...
Q: How to calculate theoretical mass percentage of water in a solution
A: Here we are aksed how to calculate theoretical mass percentage of water in a solution.
Q: Which of the following is a Lewis acid but not a Brønsted-Lowry acid? А) CH:ОН В) H-О С) СH3COОH D) ...
A: (40) Lewis acid can be defined as the species which can accept pair of electrons while Bronsted-Low...
Q: 8. Determine the half-life of an anti-hypertensive'drug if it appears to be eliminated from the body...
A:
Q: II. Von Weirman ratio. Fill in the Blanks With the given conditions during precipitation, conclude w...
A:
Q: A 50.0 mL solution of Ca(OH)2 with an unknown concentration was titrated with 0.340 M HNO3. To reach...
A: Given Volume of Ca( OH)2 solution ( V1 ) = 50 mL Molarity of HNO3 ( M2 ) = 0.340 M ...
Q: (Use S for Soluble and write the insoluble product's molecular formula if one forms) Stock Solution ...
A: We follow the solubility chart to find the solubility of any salt. According to the solubility chart...
Q: Starting on the left, label each of the following molecules as cis or trans. ÇI H3C Br Br
A: Interpretation: We have to label cis and trans .
Q: 43. Which of the following is the appropriate conversion of (CH3)4C to a skeletal structure? II II I...
A: The first broblem is an example of line notation representation of a hydrocarbon where each single b...
Q: 1.0 mol of an ideal gas with CV,m=3R/2 are transformed from an initial state T= 685K and P= 1.40bar ...
A:
Q: Consider the enthalpy diagram of an exothermic reaction. Where are the reactants in relation to the ...
A: The diagramme is given below.
Q: 925r38? Which is the balanced nuclear equation for beta decay, 2Sr38? 92Sr38 --> 93Y39 + e1 O 92Sr38...
A: The beta particle is °e-1 The beta decay results in increase in atomic number by 1 whereas the ayom...
Q: 31. Which of the following statements explain why H2O is a stronger acid than CH4? A) H2O can form h...
A:
Q: 2. Which of the following statements about a Brønsted-Lowry base is true? A) The net charge may be z...
A: Introduction: The Bronsted-Lowry theory of acids and bases was the further modification of the Arrhe...
Q: Which of the following molecules contain the same functional groups? CH;CH(NH2)CH; CH;CH2CH2NH2 CH:C...
A: Note: As per our guidelines, we are supposed to answer only one question when multiple questions are...
Q: The end point ( or equivalence point ) your acid / base titration between HCl and NaOH occurred when...
A: For an acid-base reaction the equivalence point is the moles of acid and the moles of base would ne...
Q: 11. Which of the following statements correctly describes the typical number of bonds for carbon, ni...
A: The electronic configuration of carbon is 1s2 2s2 2p2. The electronic configuration of nitrogen is ...
Q: To form a solution, the solute must Question 9 options: must evenly dissolve the solvent. be evenly ...
A: We know that a solution is a homogenous mixture consisting of a solute dissolved into a solvent. The...
Q: 49. What is the hybridization for each of the indicated atoms in the following compound? H2C CH3 I I...
A: 49) Answer : ( I ) carbon center have three sigma bond so it's 3sigma = s + p + p mixing of orbital...
Q: Ten grams of water and ten grams of iron are exposed to the sun side by side for exactly one hour. A...
A: Given: Mass of water = mass of iron = 10 g. And time passed = 1 hour.
Q: True or False The three states of matter can be interconverted without changing the substance's com...
A:
Q: <HW Chapter 5 - Assignment 2 For Practice 5.6 - Enhanced - with Feedback Part A Enter an equation fo...
A:
Q: Show how these pairs of elements combine into octet-rule molecular compounds, as in the first exampl...
A:
Q: A Lewis för Is shown below, nowever, its formal charges are not minimized. Starting from this struct...
A:
Q: 33. Which of the following intermolecular forces would not form between similar molecules of the str...
A: 33. n- butanol is having OH group. Therefore it has hydrogen bonding. n-butanol is having C-O bond. ...
Q: Quantum chemical calculations (Natural Resonance Theory) give a 1.7% weighting to the resonance form...
A:
Q: II. Von Weirm an ratio. Fill in the Blanks With the given conditions during precipitation, conclude ...
A: van-Weirman ratio is expressed as: Relative supersaturation=Q-SS (1)where,Q: Concen...
Q: Write a net ionic equation for the reaction that occurs when aqueous solutions of potassium hydroxid...
A:
Q: Rank the following atoms in order of decreasing electronegativity, putting the most electronegative ...
A:
is this chiral, achiral, meso? explain
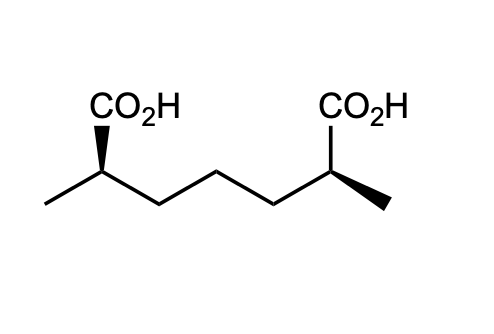
Step by step
Solved in 2 steps with 1 images


