CO(g) +2 H2 (9) 2 CH3OH(g) K = 2.2 x 104 at 298 k %3D A stoichiometric mixture of CO(9) and H2 (g) was allowed to react in two different 2.0 L rigid containers at a constant temperature of 298 K. The reaction is represented by the equation above. Diagram 1 represents the uncatalyzed reaction and diagram 2 represents the catalyzed reaction one hour after the reactants were mixed. Which of the following correctly explains the experimental results represented in the particle diagrams? S Although the reaction is thermodynamically favorable because AG° < 0 based on the value of K, only the catalyzed reaction could proceed in one hour because its reactant molecules had a A higher average kinetic energy. Although the reaction is thermodynamically favorable because AG° < 0 based on the value of K, only the catalyzed reaction could proceed in one hour because it has a lower activation- B energy reaction pathway. The reaction is not thermodynamically favorable because AG° > 0 based on the value of K, but the addition of a catalyst improved the orientation of the reactants during collisions, allowing the catalyzed reaction to proceed in one hour. The reaction is not thermodynamically favorable because AG° > 0 based on the value of K, but the catalyzed reaction could proceed in one hour because it has a lower AH and a higher AS.
CO(g) +2 H2 (9) 2 CH3OH(g) K = 2.2 x 104 at 298 k %3D A stoichiometric mixture of CO(9) and H2 (g) was allowed to react in two different 2.0 L rigid containers at a constant temperature of 298 K. The reaction is represented by the equation above. Diagram 1 represents the uncatalyzed reaction and diagram 2 represents the catalyzed reaction one hour after the reactants were mixed. Which of the following correctly explains the experimental results represented in the particle diagrams? S Although the reaction is thermodynamically favorable because AG° < 0 based on the value of K, only the catalyzed reaction could proceed in one hour because its reactant molecules had a A higher average kinetic energy. Although the reaction is thermodynamically favorable because AG° < 0 based on the value of K, only the catalyzed reaction could proceed in one hour because it has a lower activation- B energy reaction pathway. The reaction is not thermodynamically favorable because AG° > 0 based on the value of K, but the addition of a catalyst improved the orientation of the reactants during collisions, allowing the catalyzed reaction to proceed in one hour. The reaction is not thermodynamically favorable because AG° > 0 based on the value of K, but the catalyzed reaction could proceed in one hour because it has a lower AH and a higher AS.
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter12: Chemical Equilibrium
Section: Chapter Questions
Problem 113QRT
Related questions
Question
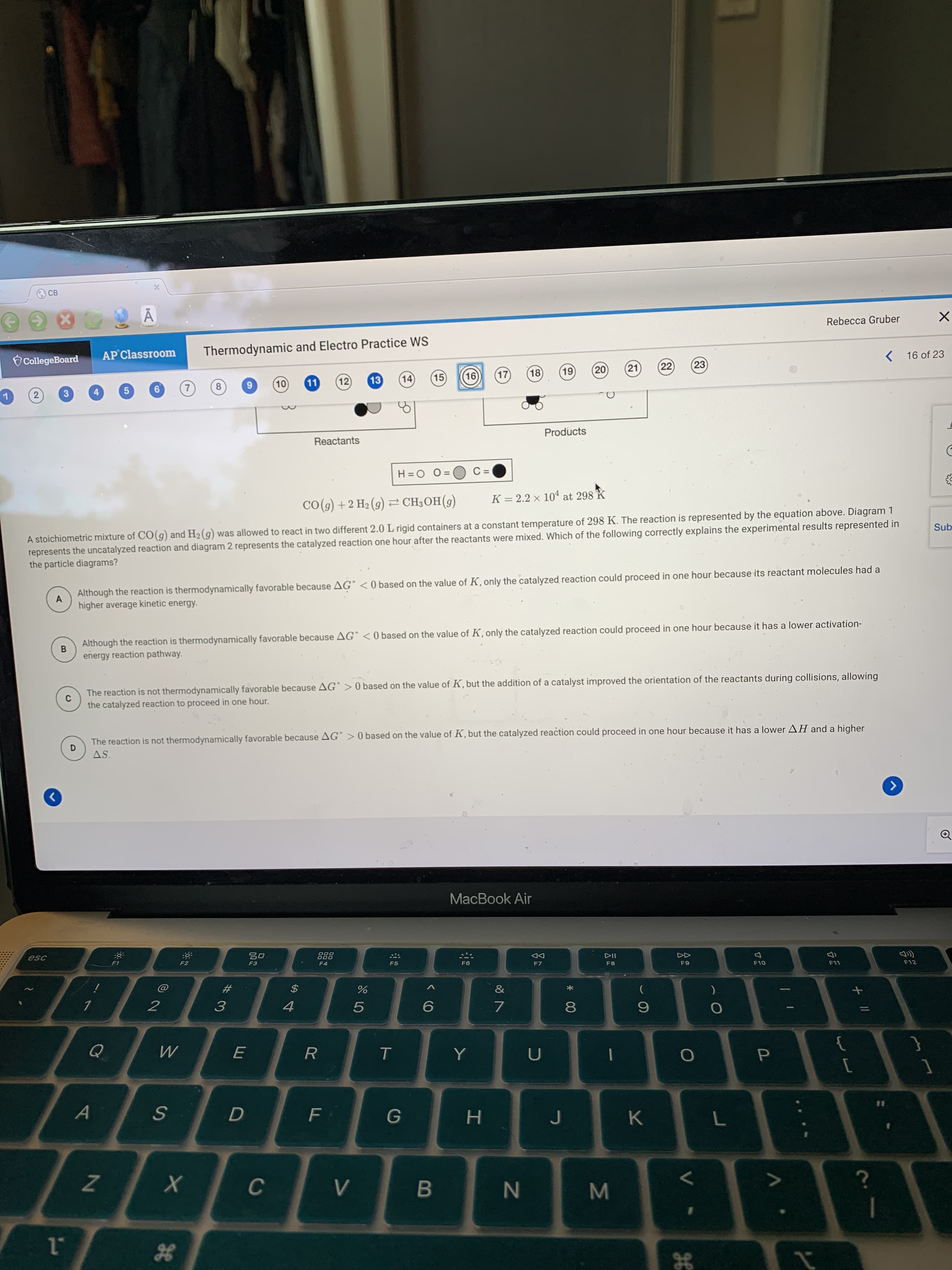
Transcribed Image Text:CO(g) +2 H2 (9) 2 CH3OH(g)
K = 2.2 x 104 at 298 k
%3D
A stoichiometric mixture of CO(9) and H2 (g) was allowed to react in two different 2.0 L rigid containers at a constant temperature of 298 K. The reaction is represented by the equation above. Diagram 1
represents the uncatalyzed reaction and diagram 2 represents the catalyzed reaction one hour after the reactants were mixed. Which of the following correctly explains the experimental results represented in
the particle diagrams?
S
Although the reaction is thermodynamically favorable because AG° < 0 based on the value of K, only the catalyzed reaction could proceed in one hour because its reactant molecules had a
A
higher average kinetic energy.
Although the reaction is thermodynamically favorable because AG° < 0 based on the value of K, only the catalyzed reaction could proceed in one hour because it has a lower activation-
B
energy reaction pathway.
The reaction is not thermodynamically favorable because AG° > 0 based on the value of K, but the addition of a catalyst improved the orientation of the reactants during collisions, allowing
the catalyzed reaction to proceed in one hour.
The reaction is not thermodynamically favorable because AG° > 0 based on the value of K, but the catalyzed reaction could proceed in one hour because it has a lower AH and a higher
AS.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning